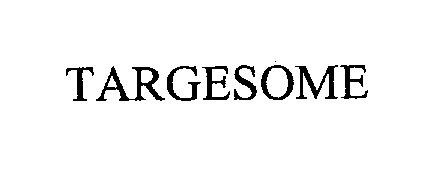TARGESOME
Targesome, Inc.
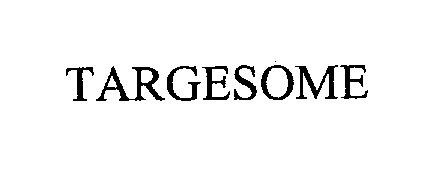
Mark For: TARGESOME™ trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 76191699 |
| Mark Literal Elements | TARGESOME |
| Mark Drawing Type | 1 - TYPESET WORD(S) /LETTER(S) /NUMBER(S) |
| Mark Type | TRADEMARK |
| Standard Character Claim | No |
| Register | PRINCIPAL |
| Current Location | FILE REPOSITORY (FRANCONIA) 2004-07-15 |
| Basis | 1(b) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | No |
| Intent To Use | Yes |
| Filed ITU | Yes |
| 44D Filed | No |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Attorney Name | THOMAS D. BRATSCHUN |
| Attorney Docket Number | 39032-24001 |
| Law Office Assigned | M60 |
| Employee Name | FRENCH, CURTIS W |
Timeline
| 2001-01-04 | Application Filed |
| 2003-03-04 | Published |
| 2003-03-04 | Published for Opposition |
| 2004-05-28 | Abandon |
| 2004-05-28 | Status: Abandoned because no Statement of Use or Extension Request timely filed after Notice of Allowance was issued. To view all docume |
| 2004-05-28 | Status: Dead/Abandoned |
| 2004-07-15 | Location: FILE REPOSITORY (FRANCONIA) |
| 2018-07-08 | Transaction Date |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 4030 Fabian Way Palo Alto, CALIFORNIA UNITED STATES 94303 |
| Legal Entity Type | Corporation |
| Legal Entity State | DELAWARE |
Documents
| IMAGE/TIFF | 2001-01-04 | |
| IMAGE/TIFF | 2001-01-04 | |
| IMAGE/TIFF | 2002-12-18 | |
| APPLICATION/XML | 2003-02-12 | |
| APPLICATION/XML | 2003-05-16 | |
| 2003-11-24 | ||
| 2003-12-05 | ||
| 2004-07-09 |
Attorney of Record
Good, Services, and Codes
(ABANDONED)
IC 005. US 006 018 044 046 051 052. G & S: Pharmaceutical preparations that aid in the transport and delivery of diagnostic, therapeutic and imaging agents for the treatment of tumor metastasis, solid tumor growth, osteoporosis, Paget's disease, humoral hyperglycemia of malignancy, angiogenesis, including tumor angiogenesis, retinopathy, macular degeneration, arthritis, including rheumatoid arthritis, periodontal disease, psoriasis and smooth muscle cell migration, diseases associated with neovascular growth, namely, solid tumors, blood borne tumors, leukemia, tumor metastastasis, benign tumors, acoustic neuromas, neurofibromas, trachomas, and pyogenic granulomas, rheumatoid arthritis, psoriasis, chronic inflammation, ocular angiogenic diseases, diabetic retinopathy, retinopathy of prematurity, macular degeneration, corneal graft rejection, neovascular glaucoma, retrolental fibroplasia, rubeosis, arteriovenous malformations, ischemic limb angiogenesis, Osler-Webber Syndrome, myocardial angiogenesis, plaque neovascularization, telangiectasia, hemophiliac joints, angiofibroma, and wound granulation, diseases of excessive or abnormal stimulation of endothelial cells, namely, intestinal adhesions, atherosclerosis, restenosis, scleroderma, and hypertrophic scars(ABANDONED)
IC 010. US 026 039 044. G & S: Medical devices, namely, polymerized nanoparticles for the transport and delivery of diagnostic, therapeutic and imaging agents
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| International Codes: | 10 |
| U.S. Codes: | 026,039,044 |
| Type Code | Type |
|---|---|
| GS0051 | Pharmaceutical preparations that aid in the transport and delivery of diagnostic, therapeutic and imaging agents for the treatment of tumor metastasis, solid tumor growth, osteoporosis, Paget's disease, humoral hyperglycemia of malignancy, angiogenesis, including tumor angiogenesis, retinopathy, macular degeneration, arthritis, including rheumatoid arthritis, periodontal disease, psoriasis and smooth muscle cell migration, diseases associated with neovascular growth, namely, solid tumors, blood borne tumors, leukemia, tumor metastastasis, benign tumors, acoustic neuromas, neurofibromas, trachomas, and pyogenic granulomas, rheumatoid arthritis, psoriasis, chronic inflammation, ocular angiogenic diseases, diabetic retinopathy, retinopathy of prematurity, macular degeneration, corneal graft rejection, neovascular glaucoma, retrolental fibroplasia, rubeosis, arteriovenous malformations, ischemic limb angiogenesis, Osler-Webber Syndrome, myocardial angiogenesis, plaque neovascularization, telangiectasia, hemophiliac joints, angiofibroma, and wound granulation, diseases of excessive or abnormal stimulation of endothelial cells, namely, intestinal adhesions, atherosclerosis, restenosis, scleroderma, and hypertrophic scars |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| ABANDONMENT - NO USE STATEMENT FILED | 2004-07-09 | |
| SOU EXTENSION 1 GRANTED | 2003-12-04 | |
| TEAS EXTENSION RECEIVED | 2003-11-24 | |
| SOU EXTENSION 1 FILED | 2003-11-24 | |
| NOA MAILED - SOU REQUIRED FROM APPLICANT | 2003-05-27 | |
| PUBLISHED FOR OPPOSITION | 2003-03-04 | |
| NOTICE OF PUBLICATION | 2003-02-12 | |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2002-12-23 | |
| EXAMINERS AMENDMENT MAILED | 2002-12-20 | |
| PREVIOUS ALLOWANCE COUNT WITHDRAWN | 2002-12-19 | |
| TEAS CHANGE OF CORRESPONDENCE RECEIVED | 2002-12-18 | |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2002-12-11 | |
| EXAMINERS AMENDMENT MAILED | 2002-12-06 | |
| ASSIGNED TO EXAMINER | 2002-12-05 | 76419 |
| PREVIOUS ALLOWANCE COUNT WITHDRAWN | 2002-10-08 | |
| ASSIGNED TO EXAMINER | 2002-10-08 | 78190 |
| ASSIGNED TO EXAMINER | 2002-10-07 | 76419 |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2002-09-27 | |
| ASSIGNED TO EXAMINER | 2002-09-09 | 78190 |
| REINSTATED | 2002-09-06 | |
| PETITION TO REVIVE-RECEIVED | 2002-06-10 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2002-06-06 | |
| ABANDONMENT - FAILURE TO RESPOND OR LATE RESPONSE | 2002-05-27 | |
| NON-FINAL ACTION MAILED | 2001-05-24 | |
| ASSIGNED TO EXAMINER | 2001-05-02 | 78190 |
| ASSIGNED TO EXAMINER | 2001-04-27 | 73363 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.