Modulators of methyl modifying enzymes, compositions and uses thereof
Albrecht , et al.
U.S. patent number RE47,428 [Application Number 15/878,663] was granted by the patent office on 2019-06-11 for modulators of methyl modifying enzymes, compositions and uses thereof. This patent grant is currently assigned to Constellation Pharmaceuticals, Inc.. The grantee listed for this patent is Constellation Pharmaceuticals, Inc.. Invention is credited to Brian K. Albrecht, James Edmund Audia, Les A. Dakin, Victor S. Gehling, Jean-Christophe Harmange, Christopher G. Nasveschuk, Rishi G. Vaswani.








View All Diagrams
| United States Patent | RE47,428 |
| Albrecht , et al. | June 11, 2019 |
Modulators of methyl modifying enzymes, compositions and uses thereof
Abstract
Agents for modulating methyl modifying enzymes, compositions and uses thereof are provided herein.
| Inventors: | Albrecht; Brian K. (Cambridge, MA), Audia; James Edmund (Cambridge, MA), Dakin; Les A. (Natick, MA), Gehling; Victor S. (Somerville, MA), Harmange; Jean-Christophe (Andover, MA), Nasveschuk; Christopher G. (Stoneham, MA), Vaswani; Rishi G. (Lexington, MA) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | Constellation Pharmaceuticals,
Inc. (Cambridge, MA) |
||||||||||
| Family ID: | 48948171 | ||||||||||
| Appl. No.: | 15/878,663 | ||||||||||
| Filed: | January 24, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | Issue Date | ||
|---|---|---|---|---|---|
| 14766632 | |||||
| PCT/US2014/015706 | Feb 11, 2014 | ||||
| Reissue of: | 14839273 | Aug 28, 2015 | 9469646 | Oct 18, 2016 | |
Foreign Application Priority Data
| Feb 11, 2013 [WO] | PCT/US2013/025639 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 13/12 (20180101); C07D 401/14 (20130101); A61P 43/00 (20180101); C07D 471/04 (20130101); C07D 401/12 (20130101); A61K 31/4545 (20130101); C07D 409/06 (20130101); A61P 13/08 (20180101); C07D 409/06 (20130101); A61P 25/00 (20180101); C07D 401/14 (20130101); A61P 35/00 (20180101); C07D 417/04 (20130101); A61P 15/00 (20180101); C07D 417/14 (20130101); A61P 17/00 (20180101); A61P 11/00 (20180101); A61P 1/04 (20180101); A61P 1/16 (20180101); C07D 405/14 (20130101); A61P 13/10 (20180101); C07D 487/04 (20130101); A61K 31/4545 (20130101); C07D 417/14 (20130101); C07D 471/04 (20130101); C07D 405/14 (20130101); C07D 417/04 (20130101); C07D 487/04 (20130101); C07D 401/12 (20130101) |
| Current International Class: | C07D 401/12 (20060101); C07D 417/04 (20060101); C07D 417/14 (20060101); C07D 405/14 (20060101); C07D 471/04 (20060101); C07D 409/06 (20060101); A61K 31/4545 (20060101); C07D 401/14 (20060101); C07D 487/04 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 4738971 | April 1988 | Eriksoo et al. |
| 5308854 | May 1994 | Hoffman, Jr. et al. |
| 7838520 | November 2010 | Delorme et al. |
| 8410088 | April 2013 | Kuntz et al. |
| 8536179 | September 2013 | Miller |
| 8846935 | September 2014 | Duquenne |
| 9051269 | June 2015 | Albrecht et al. |
| 9085583 | July 2015 | Albrecht |
| 9206128 | December 2015 | Albrecht et al. |
| 9371331 | June 2016 | Albrecht et al. |
| 9409865 | August 2016 | Albrecht et al. |
| 9469646 | October 2016 | Albrecht et al. |
| 9745305 | August 2017 | Albrecht et al. |
| 10016405 | July 2018 | Albrecht |
| 2003/0207875 | November 2003 | Gymer et al. |
| 2003/0229081 | December 2003 | Maduskuie |
| 2004/0186138 | September 2004 | Annoura et al. |
| 2005/0266473 | December 2005 | Zhang et al. |
| 2006/0035938 | February 2006 | Bladh et al. |
| 2007/0155744 | July 2007 | Jones et al. |
| 2008/0027050 | January 2008 | Terauchi et al. |
| 2008/0227826 | September 2008 | Frechette et al. |
| 2008/0280917 | November 2008 | Albrecht et al. |
| 2009/0029991 | January 2009 | Stokes et al. |
| 2009/0075833 | March 2009 | Chinnaiyan et al. |
| 2009/0270361 | October 2009 | Ito et al. |
| 2010/0069630 | March 2010 | Lee et al. |
| 2010/0222420 | September 2010 | Chinnaiyan et al. |
| 2010/0261743 | October 2010 | Londregan et al. |
| 2010/0298270 | November 2010 | Keana et al. |
| 2011/0105509 | May 2011 | Kaila et al. |
| 2011/0212946 | September 2011 | Barrow et al. |
| 2012/0071418 | March 2012 | Copeland et al. |
| 2012/0264734 | October 2012 | Kuntz et al. |
| 2013/0040906 | February 2013 | Kuntz et al. |
| 2013/0230511 | September 2013 | Heymach et al. |
| 2013/0310379 | November 2013 | Albrecht et al. |
| 2014/0107122 | April 2014 | Kuntz et al. |
| 2014/0142083 | May 2014 | Kuntz et al. |
| 2015/0259351 | September 2015 | Albrecht et al. |
| 2015/0376190 | December 2015 | Albrecht et al. |
| 2016/0185757 | June 2016 | Albrecht et al. |
| 2016/0333016 | November 2016 | Albrecht et al. |
| 2003/020722 | Mar 2003 | WO | |||
| 2003/079986 | Oct 2003 | WO | |||
| 2007/011626 | Jan 2007 | WO | |||
| 2007/014838 | Feb 2007 | WO | |||
| 2007/067968 | Jun 2007 | WO | |||
| 2009/006577 | Jan 2009 | WO | |||
| 2009/087285 | Jul 2009 | WO | |||
| 2009/153721 | Dec 2009 | WO | |||
| 2011/131741 | Oct 2011 | WO | |||
| 2011/140324 | Nov 2011 | WO | |||
| 2011/140325 | Nov 2011 | WO | |||
| 2012/005805 | Jan 2012 | WO | |||
| 2012/024543 | Feb 2012 | WO | |||
| 2012/051492 | Apr 2012 | WO | |||
| 2012/068589 | May 2012 | WO | |||
| 2012/075080 | Jun 2012 | WO | |||
| 2012/115885 | Aug 2012 | WO | |||
| 2012/118812 | Sep 2012 | WO | |||
| 2013/039988 | Mar 2013 | WO | |||
| 2013/049770 | Apr 2013 | WO | |||
| 2013/067296 | May 2013 | WO | |||
| 2013/067300 | May 2013 | WO | |||
| 2013/067302 | May 2013 | WO | |||
| 2013/075083 | May 2013 | WO | |||
| 2013/075084 | May 2013 | WO | |||
| 2013/078320 | May 2013 | WO | |||
| 2013/120104 | Aug 2013 | WO | |||
| 2013/138361 | Sep 2013 | WO | |||
| 2013/155317 | Oct 2013 | WO | |||
| 2013/155464 | Oct 2013 | WO | |||
| 2013/173441 | Nov 2013 | WO | |||
| 2014/049488 | Apr 2014 | WO | |||
| 2014/062720 | Apr 2014 | WO | |||
| 2014/062733 | Apr 2014 | WO | |||
| 2014/071109 | May 2014 | WO | |||
| 2014/077784 | May 2014 | WO | |||
| 2014/085666 | Jun 2014 | WO | |||
| 2014/092905 | Jun 2014 | WO | |||
| 2014/097041 | Jun 2014 | WO | |||
| 2014/100080 | Jun 2014 | WO | |||
| 2015-200650 | Dec 2015 | WO | |||
| 2015/200650 | Dec 2015 | WO | |||
Other References
|
Vazquez, Optimization of personalized therapies for anticancer treatment. BMC Syst Biol. Apr. 12, 2013;7:31. 11 pages. cited by applicant . Amatangelo et al., Three-dimensional culture sensitizes epithelial ovarian cancer cells to EZH2 methyltransferase inhibition. Cell Cycle. Jul. 1, 2013;12(13):2113-9. cited by applicant . Extended European Search Report for Application No. 13746186.9, dated Aug. 5, 2015. 6 pages. cited by applicant . Fiskus et al., Combined epigenetic therapy with the histone methyltransferase EZH2 inhibitor 3-deazaneplanocin A and the histone deacetylase inhibitor panobinostat against human AML cells. Blood. Sep. 24, 2009;114(13):2733-43. cited by applicant . Fiskus et al., Histone deacetylase inhibitors deplete enhancer of zeste 2 and associated polycomb repressive complex 2 proteins in human acute leukemia cells. Mol Cancer Ther. Dec. 2006;5(12):3096-104. cited by applicant . International Search Report for Application No. PCT/US2013/025639, dated May 8, 2013, 9 pages. cited by applicant . Knutson et al., Selective inhibition of EZH2 by EPZ-6438 leads to potent antitumor activity in EZH2-mutant non-Hodgkin lymphoma. Mol Cancer Ther. Apr. 2014;13(4):842-54. cited by applicant . Knutson et al., A selective inhibitor of EZH2 blocks H3K27 methylation and kills mutant lymphoma cells. Nat Chem Biol. Nov. 2012;8(11):890-6. cited by applicant . Knutson et al., Durable tumor regression in genetically altered malignant rhabdoid tumors by inhibition of methyltransferase EZH2. Proc Natl Acad Sci U S A. May 7, 2013;110(19):7922-7. cited by applicant . Konze et al., An orally bioavailable chemical probe of the Lysine Methyltransferases EZH2 and EZH1. ACS Chem Biol. 2013;8(6)1324-34. cited by applicant . McCabe et al., EZH2 inhibition as a therapeutic strategy for lymphoma with EZH2-activating mutations. Nature. Dec. 6, 2012;492(7427):108-12. cited by applicant . Qi et al., Selective inhibition of Ezh2 by a small molecule inhibitor blocks tumor cells proliferation. Proc Natl Acad Sci U S A. Dec. 26, 2012;109(52):21360-5. cited by applicant . Spannhoff et al., The emerging therapeutic potential of histone methyltransferase and demethylase inhibitors. ChemMedChem. Oct. 2009;4(10):1568-82. cited by applicant . Van Aller et al., Long residence time inhibition of EZH2 in activated polycomb repressive complex 2. ACS Chem Biol. Mar. 21, 2014;9(3):622-9. cited by applicant . Woo et al., Biological evaluation of tanshindiols as EZH2 histone methyltransferase inhibitors. Bioorg Med Chem Lett. Jun. 1, 2014;24(11):2486-92. cited by applicant . Yap et al., Somatic mutations at EZH2 Y641 act dominantly through a mechanism of selectively altered PRC2 catalytic activity, to increase H3K27 trimethylation. Blood. Feb. 24, 2011;117(8):2451-9. cited by applicant . Verma et al., Identification of Potent, Selective, Cell-Active Inhibitors of the Histone Lysine Methyltransferase EZH2. ACS Med Chem Lett. Oct. 19, 2012;3(12):1091-6. cited by applicant . Abreu et al., DNA methylation: a promising target for the twenty-first century. Expert Opin Ther Targets. Aug. 2008;12(8):1035-47. cited by applicant . Gillet et al., The clinical relevance of cancer cell lines. J Natl Cancer Inst. Apr. 3, 2013;105(7):452-8. cited by applicant . Gura, Systems for identifying new drugs are often faulty. Science. Nov. 7, 1997;278(5340):1041-2. cited by applicant . Ito et al., A medium-term rat liver bioassay for rapid in vivo detection of carcinogenic potential of chemicals. Cancer Sci. Jan. 2003;94(1):3-8. cited by applicant . Johnson et al., Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials. Br J Cancer. May 18, 2001;84(10):1424-31. cited by applicant . National Cancer Institute. Seer Training Modules. Cancer Classification. Retrieved online at: http://training.seer.cancer.gov/module_ase/unit3_categories2_by_histology- .html. 3 pages. (2012). cited by applicant . Simone, Introduction. Omenn, Cancer Prevention. Part XIV, Oncology. Cecil Textbook of Medicine. 20th Edition, vol. 1. J. Claude Bennett (Ed.). W.B. Saunders Company. pp. 1004-1010. (1966). cited by applicant . STN registry database compound 322425-80-9 (entered STN on Feb. 20, 2001). cited by applicant . STN registry database compound 950111-40-7 from Chemical Library Supplier Enamine (entered STN on Oct. 10, 2007). cited by applicant . STN registry database compound 1002886-67-0 from the ZINC (Soichet Laboratory) (entered STN on Feb. 12, 2008). cited by applicant . Venkatesh et al., Role of the development scientist in compound lead selection and optimization. J Pharm Sci. Feb. 2000;89(2):145-54. cited by applicant . Williams et al., Foye's Principles of Medicinal Chemistry, 5th edition, pp. 50 and 59-61, 2002. cited by applicant . U.S. Appl. No. 13/988,180, filed Aug. 5, 2013, (National Stage of PCT/US2011/061740). cited by applicant . U.S. Appl. No. 14/358,558, filed May 15, 2014, U.S. Pat. No. 9,206,128, (National Stage of PCT/US2012/065796). cited by applicant . U.S. Appl. No. 14/358,455, filed May 15, 2014, U.S. Pat. No. 9,051,269, (National Stage of PCT/US2012/065797). cited by applicant . U.S. Appl. No. 14/707,874, filed May 8, 2015, U.S. Pat. No. 9,409,865, (continuation of U.S. Appl. No. 14/358,455). cited by applicant . U.S. Appl. No. 14/839,273, filed Aug. 28, 2015, U.S. Pat. No. 9,469,646. cited by applicant . U.S. Appl. No. 14/377,214, filed Aug. 7, 2014, U.S. Pat. No. 9,085,583, (National Stage of PCT/US2013/025639). cited by applicant . U.S. Appl. No. 14/661,797, filed Mar. 18, 2015, U.S. Pat. No. 9,371,331, (continuation of U.S. Appl. No. 14/377,214). cited by applicant . U.S. Appl. No. 15/155,749, filed May 16, 2016, (continuation of U.S. Appl. No. 14/661,797). cited by applicant . U.S. Appl. No. 14/769,471, filed Aug. 21, 2015, U.S. Pat. No. 9,745,305. cited by applicant . U.S. Appl. No. 14/911,343, filed Feb. 10, 2016, (National Stage of PCT/US2014/051201). cited by applicant . CAS Registry No. 1061629-12-6, (2015). cited by applicant . CAS Registry No. 1100242-53-2, (2015). cited by applicant . CAS Registry No. 1118826-71-3, (2015). cited by applicant . CAS Registry No. 1269034-31-2, (2015). cited by applicant . CAS Registry No. 1269036-62-4, (2015). cited by applicant . CAS Registry No. 1278089-62-5, (2015). cited by applicant . CAS Registry No. 1290560-58-5, (2015). cited by applicant . Copending U.S. Appl. No. 14/766,632, filed Aug. 7, 2015, entitled "Modulators of Methyl Modifying Enzymes, Compositions and Uses Thereof". cited by applicant . Copending U.S. Appl. No. 14/661,797, filed Mar. 18, 2015, entitled "Modulators of Methyl Modifying Enzymes, Compositions and Uses Thereof". cited by applicant . U.S. Appl. No. 14/661,797, filed Mar. 18, 2015. cited by applicant . Alexei Vazquez, "Optimization of Personalized Therapies for Anticancer Treatment," BMC Systems Biology, 2013, 7:31, 11 pages, http://www.biomedcentral.com/1752-0509/7/31. cited by applicant . Amatangelo et al., "Three-Dimensional Culture Sensitizes Epithelial Ovarian Cancer Cells to EZH2 Methyltransferase Inhibition," Cell Cycle, 12(13), 2013, 2113-2119. cited by applicant . Extended European Search Report issued in European Application No. 13746186.9, dated Aug. 5, 2015. 6 pages. cited by applicant . Fiskus, et al., "Combined Epigenetic Therapy with the Histone Methyltransferase EZH2 Inhibitor 3-Deazaneplanocin A and the Histone Deacetylase Inhibitor Panobinostat Against Human AML Cells," Blood, Sep. 24, 2009, 114:13, pp. 2733-2743. cited by applicant . Fiskus, et al., "Histone Deacetylase Inhibitors Deplete Enhancer of Zeste 2 and Associated Polycomb Repressive complex 2 Proteins in Human Acute Leukemia Cells," Molecular Cancer Therapeutics, 2006;5:3096-3104. cited by applicant . International Search Report, International Application No. PCT/US2013/025639, International Filing Date Feb. 11, 2013, Mailed May 8, 2013, 9 pages. cited by applicant . Knutson et al., "Selective Inhibition of EZH2 by EPZ-6438 Leads to Potent Antitumor Activity in EZH2-Mutant Non-Hodgkin Lymphoma," Molecular Cancer Therapeutics, 13(4), 2014, 842-854. cited by applicant . Knutson, et al., "A Selective Inhibitor of EZH2 Blocks H3K27 Methylation and Kills Mutant Lymphoma Cells," Nature Chemical Biology, 8(11), 2012, 890-896. cited by applicant . Knutson, et al., "Durable Tumor Regression in Genetically Altered Malignant Rhabdoid Tumors by Inhibition of Methyltransferase EZH2," Proceedings of the National Academy of Sciences of the United States of America, 110(19), 2013, 7922-7927. cited by applicant . Konze, et al., "An Orally Bioavailable Chemical Probe of the Lysine Methyltransferases EZH2 and EZH1," ACS Chemical Biology, (2013), 8(6), 1324-1334, CAPLUS, DOI: 10.1021/cb400133j. cited by applicant . McCabe, et al., "EZH2 Inhibition as a Therapeutic Strategy for Lymphoma with EZH2-Activating Mutations," Nature, 492(7427), 2012, 108-112. cited by applicant . PubChem Compound Summary for CID 40170690, May 30, 2009, 2 pages. cited by applicant . PubChem Compound Summary for CID 50961558, Mar. 29, 2011, 2 pages. cited by applicant . PubChem Compound Summary for CID 6918837, Jul. 28, 2006, 2 pages. cited by applicant . PubChem Compound Summary for CID 73087, Aug. 1, 2005, 2 pages. cited by applicant . Qi, et al., "Selective Inhibition of Ezh2 by a Small Molecule Inhibitor Blocks Tumor Cells Proliferation," Proceedings of the National Academy of Sciences of the United States of America, 109(52), 2012, 21360-21365. cited by applicant . Spannhoff, et al., "The Emerging Therapeutic Potential of Histone Methyltransferase and Demethylase Inhibitors," Chem Med Chem, 2009, 4:1568-1582. cited by applicant . Van Aller, et al., "Long Residence Time Inhibition of EZH2 in Activated Polycomb Repressive Complex 2," ACS Chem. Biol., 9(3), 2014, 622-629. cited by applicant . Verma, et al., "Identification of Potent, Selective, Cell-Active Inhibitors of the Histone Lysine Methyltransferase EZH2," ACS Medicinal Chemistry Letters, 3(12), 2012, 1091-1096. cited by applicant . Woo, et al., "Biological Evaluation of Tanshindiols as EZH2 Histone Methyltransferase Inhibitors," Bioorganic & Medicinal Chemistry Letters, 24(11), 2014, 2486-2492. cited by applicant . Yap, et al., "Somatic mutations at EZH2 Y641 act dominantly through a mechanism of selectively altered PRC2 catalytic activity, to increase H3K27 trimethylation," Blood, vol. 117, No. 8, Feb. 24, 2011, pp. 2451-2459. cited by applicant . Registry, Sep. 29, 2011, RN: 1333889-30-7. cited by applicant . Registry, Sep. 7, 2011, RN 1329352-49-9. cited by applicant . Registry, Sep. 7, 2011, RN: 1329234-68-5. cited by applicant . Registry, Sep. 6, 2011, RN 1328976-87-9. cited by applicant . Registry, Sep. 5, 2011, RN: 1328462-28-7. cited by applicant . Registry, Sep. 4, 2011, RN: 1328132-30-4. cited by applicant . Registry, Sep. 2, 2011, RN: 1327055-57-1. cited by applicant . Registry, Sep. 1, 2011, RN: 1326727-17-6. cited by applicant . Registry, May 25, 2011, RN: 1300453-83-1. cited by applicant. |
Primary Examiner: Campell; Bruce R
Attorney, Agent or Firm: McCarter & English, LLP Davis; Steven G. DeGrazia; Michael J.
Parent Case Text
CROSS-REFERENCE TO RELATED APPLICATIONS
This application is a .Iadd.reissue application of U.S. Pat. No. 9,469,646, which is a .Iaddend.continuation of U.S. application Ser. No. 14/766,632, filed Aug. 7, 2015, which is a 35 U.S.C. .sctn.371 national stage filing of International Application No. PCT/US2014/015706, filed Feb. 11, 2014, which claims priority to International Application No. PCT/US2013/025639, filed Feb. 11, 2013.Iadd., the entire contents of each of which are incorporated by reference herein.Iaddend..
Claims
The invention claimed is:
1. A compound of the formula: ##STR00090## or a pharmaceutically acceptable salt thereof.
2. The compound of claim 1, wherein the compound is of the formula: ##STR00091## or a pharmaceutically acceptable salt thereof.
3. A pharmaceutical composition comprising a compound of the formula: ##STR00092## or a pharmaceutically acceptable salt thereof; and a pharmaceutically acceptable carrier.
4. The pharmaceutical composition of claim 3, wherein the compound is of the formula: ##STR00093## or a pharmaceutically acceptable salt thereof.
Description
BACKGROUND OF THE INVENTION
Eukaryotic chromatin is composed of macromolecular complexes called nucleosomes. A nucleosome has 147 base pairs of DNA wrapped around a protein octamer having two subunits of each of histone protein H2A, H2B, H3, and H4. Histone proteins are subject to post-translational modifications which in turn affect chromatin structure and gene expression. One type of post-translational modification found on histones is methylation of lysine and arginine residues. Histone methylation plays a critical role in the regulation of gene expression in eukaryotes. Methylation affects chromatin structure and has been linked to both activation and repression of transcription (Zhang and Reinberg, Genes Dev. 15:2343-2360, 2001). Enzymes that catalyze attachment and removal of methyl groups from histones are implicated in gene silencing, embryonic development, cell proliferation, and other processes.
SUMMARY OF THE INVENTION
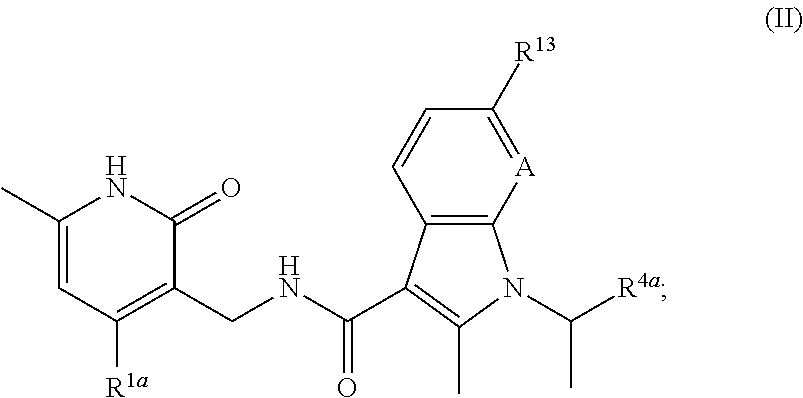
The present disclosure encompasses the recognition that methyl modifying enzymes are an attractive target for modulation, given their role in the regulation of diverse biological processes. It has now been found that compounds of this invention, and pharmaceutically acceptable compositions thereof, are effective as agents that stimulate activity of histone methyl modifying enzymes, including histone methylases and histone demethylases. Such compounds have the general formula II:
##STR00001## or a pharmaceutically acceptable salt thereof, wherein each variable is as defined herein.
Compounds of the present invention, and pharmaceutically acceptable compositions thereof, are useful for treating a variety of diseases, disorders or conditions, associated with a methyl modifying enzyme. Such diseases, disorders, or conditions include those described herein.
Compounds provided by this invention are also useful for the study of methyl modifying enzymes in biological and pathological phenomena; the study of intracellular signal transduction pathways mediated by methyl modifying enzymes and the comparative evaluation of new methyl modifying enzyme modulators.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1. Exemplary compounds of Formula II.
DETAILED DESCRIPTION OF CERTAIN EMBODIMENTS
1. General Description of Compounds of the Invention
In certain embodiments, the present invention provides a compound of Formula II:
##STR00002## or a pharmaceutically acceptable salt thereof, wherein:
A is CH or N;
R.sup.1a is selected from --C.sub.1-C.sub.2 alkyl and --O--(C.sub.1-C.sub.2 alkyl), wherein R.sup.1a is optionally substituted with one or more fluoro;
R.sup.4a is selected from --(C.sub.1-C.sub.4 alkylene)-O--(C.sub.1-C.sub.3 alkyl), 1-substituted-piperidin-4-yl, C.sub.3-C.sub.6 cycloalkyl optionally substituted with one or more fluoro, and tetrahydropyranyl; and
R.sup.13 is selected from hydrogen, halo, phenyl, pyridinyl, and --O--(C.sub.1-C.sub.4 alkyl).
2. Compounds and Definitions
Definitions of specific functional groups and chemical terms are described in more detail below. For purposes of this invention, the chemical elements are identified in accordance with the Periodic Table of the Elements, CAS version, Handbook of Chemistry and Physics, 75.sup.th Ed., inside cover, and specific functional groups are generally defined as described therein. Additionally, general principles of organic chemistry, as well as specific functional moieties and reactivity, are described in Organic Chemistry, Thomas Sorrell, University Science Books, Sausalito, 1999; Smith and March March's Advanced Organic Chemistry, 5.sup.th Edition, John Wiley & Sons, Inc., New York, 2001; Larock, Comprehensive Organic Transformations, VCH Publishers, Inc., New York, 1989; Carruthers, Some Modern Methods of Organic Synthesis, 3.sup.rd Edition, Cambridge University Press, Cambridge, 1987; the entire contents of each of which are incorporated herein by reference.
Unless otherwise stated, structures depicted herein are also meant to include all isomeric (e.g., enantiomeric, diastereomeric, and geometric (or conformational)) forms of the structure; for example, the R and S configurations for each asymmetric center, Z and E double bond isomers, and Z and E conformational isomers. Therefore, single stereochemical isomers as well as enantiomeric, diastereomeric, and geometric (or conformational) mixtures of the present compounds are within the scope of the invention. Unless otherwise stated, all tautomeric forms of the compounds of the invention are within the scope of the invention. Additionally, unless otherwise stated, structures depicted herein are also meant to include compounds that differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the present structures including the replacement of hydrogen by deuterium or tritium, or the replacement of a carbon by a .sup.13C- or .sup.14C-enriched carbon are within the scope of this invention. Such compounds are useful, for example, as analytical tools, as probes in biological assays, or as therapeutic agents in accordance with the present invention.
Where a particular enantiomer is preferred, it may, in some embodiments be provided substantially free of the corresponding enantiomer, and may also be referred to as "optically enriched." "Optically-enriched," as used herein, means that the compound is made up of a significantly greater proportion of one enantiomer. In certain embodiments the compound is made up of at least about 90% by weight of a preferred enantiomer. In other embodiments the compound is made up of at least about 95%, 98%, or 99% by weight of a preferred enantiomer. Preferred enantiomers may be isolated from racemic mixtures by any method known to those skilled in the art, including chiral high pressure liquid chromatography (HPLC) and the formation and crystallization of chiral salts or prepared by asymmetric syntheses. See, for example, Jacques et al., Enantiomers, Racemates and Resolutions (Wiley Interscience, New York, 1981); Wilen, et al., Tetrahedron 33:2725 (1977); Eliel, E. L. Stereochemistry of Carbon Compounds (McGraw-Hill, NY, 1962); Wilen, S. H. Tables of Resolving Agents and Optical Resolutions p. 268 (E. L. Eliel, Ed., Univ. of Notre Dame Press, Notre Dame, Ind. 1972).
A wavy bond
at a chiral center in a chemical structure is used to denote compounds of the invention that are optically pure, but whose optical rotation has not been determined. A straight bond at a chiral center indicates a racemic mixture although, as stated above, the invention also includes all possible isomeric forms of the racemate.
The terms "halo" and "halogen" as used herein refer to an atom selected from fluorine (fluoro, --F), chlorine (chloro, --Cl), bromine (bromo, --Br), and iodine (iodo, --I).
The term "alkylene" refers to a bivalent alkyl group. An "alkylene chain" is a polymethylene group, i.e., --(CH.sub.2).sub.n--, wherein n is a positive integer, preferably from 1 to 6, from 1 to 4, from 1 to 3, from 1 to 2, or from 2 to 3. A substituted alkylene chain is a polymethylene group in which one or more methylene hydrogen atoms are replaced with a substituent. Suitable substituents include those described below for a substituted aliphatic group.
The term "alkyl," as used herein, refers to a monovalent saturated, straight- or branched-chain hydrocarbon radical derived from an aliphatic moiety containing between one and six carbon atoms by removal of a single hydrogen atom. In some embodiments, alkyl contains 1-5 carbon atoms. In another embodiment, alkyl contains 1-4 carbon atoms. In still other embodiments, alkyl contains 1-3 carbon atoms. In yet another embodiment, alkyl contains 1-2 carbons. Examples of alkyl radicals include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, iso-butyl, sec-butyl, sec-pentyl, iso-pentyl, tert-butyl, n-pentyl, neopentyl, n-hexyl, sec-hexyl, and the like.
As described herein, compounds of the invention may contain "optionally substituted" moieties. In general, the term "substituted", whether preceded by the term "optionally" or not, means that one or more hydrogens of the designated moiety are replaced with a suitable substituent. Unless otherwise indicated, an "optionally substituted" group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be substituted with more than one substituent selected from a specified group, the substituent may be either the same or different at each position. Combinations of substituents envisioned under this invention are preferably those that result in the formation of stable or chemically feasible compounds. The term "stable", as used herein, refers to compounds that are not substantially altered when subjected to conditions to allow for their production, detection, and, in certain embodiments, their recovery, purification, and use for one or more of the purposes disclosed herein.
Suitable monovalent substituents on a substitutable carbon atom of an "optionally substituted" group are independently halogen; --(CH.sub.2).sub.0-4R.sup..smallcircle.; --(CH.sub.2).sub.0-4OR.sup..smallcircle.; --O--(CH.sub.2).sub.0-4C(O)OR.sup..smallcircle.; --(CH.sub.2).sub.0-4CH(OR.sup..smallcircle.).sub.2; --(CH.sub.2).sub.0-4SR.sup..smallcircle.; --(CH.sub.2).sub.0-4Ph, which may be substituted with R.sup..smallcircle.; --(CH.sub.2).sub.0-4O(CH.sub.2).sub.0-1Ph which may be substituted with R.sup..smallcircle.; --CH.dbd.CHPh, which may be substituted with R.sup..smallcircle.; --NO.sub.2; --CN; --N.sub.3; --(CH.sub.2).sub.0-4N(R.sup..smallcircle.).sub.2; --(CH.sub.2).sub.0-4N(R.sup..smallcircle.)C(O)R.sup..smallcircle.; --N(R.sup..smallcircle.)C(S)R.sup..smallcircle.; --(CH.sub.2).sub.0-4N(R.sup..smallcircle.)C(O)NR.sup..smallcircle..sub.2; --N(R.sup..smallcircle.)C(S)NR.sup..smallcircle..sub.2; --(CH.sub.2).sub.0-4N(R.sup..smallcircle.)C(O)OR.sup..smallcircle.; --N(R.sup..smallcircle.)N(R.sup..smallcircle.)C(O)R.sup..smallcircle.; --N(R.sup..smallcircle.)N(R.sup..smallcircle.)C(O)NR.sup..smallcircle..su- b.2; --N(R.sup..smallcircle.) N(R.sup..smallcircle.)C(O)OR.sup..smallcircle.; --(CH.sub.2).sub.0-4C(O)R.sup..smallcircle.; --C(S)R.sup..smallcircle.; --(CH.sub.2).sub.0-4C(O)OR.sup..smallcircle.; --(CH.sub.2).sub.0-4C(O)SR.sup..smallcircle.; --(CH.sub.2).sub.0-4C(O)OSiR.sup..smallcircle..sub.3; --(CH.sub.2).sub.0-4OC(O)R.sup..smallcircle.; --OC(O)(CH.sub.2).sub.0-4SR--; --SC(S)SR.sup..smallcircle.; --(CH.sub.2).sub.0-4SC(O)R.sup..smallcircle.; --(CH.sub.2).sub.0-4C(O)NR.sup..smallcircle..sub.2; --C(S)NR.sup..smallcircle..sub.2; --C(S)SR.sup..smallcircle.; --SC(S)SR.sup..smallcircle.; --(CH.sub.2).sub.0-4OC(O)NR.sup..smallcircle..sub.2; --C(O)N(OR.sup..smallcircle.) R.sup..smallcircle.; --C(O)C(O)R.sup..smallcircle.; --C(O)CH.sub.2C(O)R.sup..smallcircle.; --C(NOR.sup..smallcircle.)R.sup..smallcircle.; --(CH.sub.2).sub.0-4SSR.sup..smallcircle.; --(CH.sub.2).sub.0-4S(O).sub.2R.sup..smallcircle.; --(CH.sub.2).sub.0-4S(O).sub.2OR.sup..smallcircle.; --(CH.sub.2).sub.0-4OS(O).sub.2R.sup..smallcircle.; --S(O).sub.2NR.sup..smallcircle..sub.2; --(CH.sub.2).sub.0-4S(O)R.sup..smallcircle.; --N(R.sup..smallcircle.)S(O).sub.2NR.sup..smallcircle..sub.2; --N(R.sup..smallcircle.)S(O).sub.2R.sup..smallcircle.; --N(OR.sup..smallcircle.)R.sup..smallcircle.; --C(NH)NR.sup..smallcircle..sub.2; --P(O).sub.2R.sup..smallcircle.; --P(O)R.sup..smallcircle..sub.2; --OP(O)R.sup..smallcircle..sub.2; --OP(O)(OR.sup..smallcircle.).sub.2; --SiR.sup..smallcircle..sub.3; --(C.sub.1-4 straight or branched)alkylene)O--N(R.sup..smallcircle.).sub.2; or --(C.sub.1-4 straight or branched alkylene)C(O)O--N(R.sup..smallcircle.).sub.2, wherein each R.sup..smallcircle. may be substituted as defined below and is independently hydrogen, C.sub.1-6 aliphatic, --CH.sub.2Ph, --O(CH.sub.2).sub.0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R.sup..smallcircle., taken together with their intervening atom(s), form a 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, which may be substituted as defined below.
Suitable substituents on a substitutable nitrogen of an "optionally substituted" group include --R.sup..dagger., --NR.sup..dagger..sub.2, --C(O)R.sup..dagger., --C(O)OR.sup..dagger., --C(O)C(O)R.sup..dagger., --C(O)CH.sub.2C(O)R.sup..dagger., --S(O).sub.2R.sup..dagger., --S(O).sub.2NR.sup..dagger..sub.2, --C(S)NR.sup..dagger..sub.2, --C(NH)NR.sup..dagger..sub.2, or --N(R.sup..dagger.)S(O).sub.2R.sup..dagger.; wherein each R.sup..dagger. is independently hydrogen, C.sub.1-6 aliphatic which may be substituted as defined below, unsubstituted --OPh, or an unsubstituted 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur, or, notwithstanding the definition above, two independent occurrences of R.sup..dagger., taken together with their intervening atom(s) form an unsubstituted 3-12-membered saturated, partially unsaturated, or aryl mono- or bicyclic ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
Suitable substituents on the aliphatic group of R.sup..dagger. are independently halogen, --R.sup..cndot., -(halo R.sup..cndot.), --OH, --OR.sup..cndot., --O(halo R.sup..cndot.), --CN, --C(O)OH, --C(O)O R.sup..cndot., --NH.sub.2, --NHR.sup..cndot., --NR.sup..cndot..sub.2, or --NO.sub.2, wherein each R.sup..cndot. is unsubstituted or where preceded by "halo" is substituted only with one or more halogens, and is independently C.sub.1-4aliphatic, --CH.sub.2Ph, --O(CH.sub.2).sub.0-1Ph, or a 5-6-membered saturated, partially unsaturated, or aryl ring having 0-4 heteroatoms independently selected from nitrogen, oxygen, or sulfur.
As used herein, the term "inhibitor" is defined as a compound that binds to and/or inhibits a target S-adenosylmethionine (SAM) utilizing enzyme with measurable affinity. In certain embodiments, an inhibitor has an IC.sub.50 and/or binding constant of less about 50 .mu.M, less than about 1 .mu.M, less than about 500 nM, less than about 100 nM, or less than about 10 nM.
The terms "measurable affinity" and "measurably inhibit," as used herein, means a measurable change in activity of at least one SAM utilizing enzyme between a sample comprising a provided compound, or composition thereof, and at least one SAM dependent enzyme, and an equivalent sample comprising at least one SAM dependent enzyme, in the absence of said compound, or composition thereof.
3. Description of Exemplary Compounds
In some embodiments of Formula II, R.sup.1a is selected from --OCH.sub.3, --CH.sub.3, --OCHF.sub.2, and --CH.sub.2CH.sub.3.
In some embodiments of Formula II, R.sup.4a is selected from --CH.sub.2OCH.sub.3, --CH(CH.sub.3)OCH.sub.3, 4,4-difluorocyclohexyl, cyclopropyl, tetrayhyrdopyran-4-yl, 1-(t-butoxycarbonyl)-piperidin-4-yl, 1-(isobutoxycarbonyl)-piperidin-4-yl, 1-(isopropoxycarbonyl)-piperidin-4-yl, 1-(2-fluoroethyl)-piperidin-4-yl, 1-(2,2-difluoroethyl)-piperidin-4-yl, 1-(2,2,2-trifluoroethyl)-piperidin-4-yl, 1-(2-hydroxyisobutyl)-piperidin-4-yl, 1-(hydroxyisopropylcarbonyl)-piperidin-4-yl, 1-(ethoxycarbonylmethyl)-piperidin-4-yl, 1-(isopropylcarbonyl)-piperidin-4-yl, 1-methylpiperidin-4-yl, 1-(methylsulfonyl)-piperidin-4-yl, 1-(ethylsulfonyl)-piperidin-4-yl, 1-(isopropylsulfonyl)-piperidin-4-yl, 1-(phenyl)-piperidin-4-yl, 1-(oxetan-3-yl)piperidin-4-yl, 1-(pyridin-2-yl)-piperidin-4-yl, and 1-(pyrimidin-2-yl)-piperidin-4-yl.
In some embodiments of Formula II, R.sup.13 is selected from hydrogen, chloro, fluoro, --OCH(CH.sub.3).sub.2, phenyl, and pyridin-2-yl.
Exemplary compounds of Formula II are set forth in FIG. 1. In some cases two (or more) of the compounds in FIG. 1 having one (or more) wavy bonds will have the exact same structure. Because the wavy bond represents a chiral center of undetermined optical rotation, such compounds will be understood to be separate and distinct optical isomers of one another. FIG. 1 is annotated to indicate those sets of two or more compounds that have the same depicted structure, but are of different stereochemistry.
4. Uses, Formulation and Administration
Pharmaceutically Acceptable Compositions
According to another embodiment, the invention provides a composition comprising a compound of this invention or a pharmaceutically acceptable derivative thereof and a pharmaceutically acceptable carrier, adjuvant, or vehicle. The amount of compound in compositions of this invention is such that is effective to measurably modulate a histone methyl modifying enzyme, or a mutant thereof, in a biological sample or in a patient. In certain embodiments, the amount of compound in compositions of this invention is such that is effective to measurably modulate a histone methyl modifying enzyme, or a mutant thereof, in a biological sample or in a patient.
In certain embodiments, a composition of this invention is formulated for administration to a patient in need of such composition. In some embodiments, a composition of this invention is formulated for oral administration to a patient.
The term "patient," as used herein, means an animal, preferably a mammal, and most preferably a human.
The term "pharmaceutically acceptable carrier, adjuvant, or vehicle" refers to a non-toxic carrier, adjuvant, or vehicle that does not destroy the pharmacological activity of the compound with which it is formulated. Pharmaceutically acceptable carriers, adjuvants or vehicles that may be used in the compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylenepolyoxypropylene-block polymers, polyethylene glycol and wool fat.
A "pharmaceutically acceptable derivative" means any non-toxic salt, ester, salt of an ester or other derivative of a compound of this invention that, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention or an inhibitorily active metabolite or residue thereof.
Compositions of the present invention may be administered orally, parenterally, by inhalation spray, topically, rectally, nasally, buccally, vaginally or via an implanted reservoir. The term "parenteral" as used herein includes subcutaneous, intravenous, intramuscular, intra-articular, intra-synovial, intrasternal, intrathecal, intrahepatic, intralesional and intracranial injection or infusion techniques. Preferably, the compositions are administered orally, intraperitoneally or intravenously. Sterile injectable forms of the compositions of this invention may be aqueous or oleaginous suspension. These suspensions may be formulated according to techniques known in the art using suitable dispersing or wetting agents and suspending agents. The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example as a solution in 1,3-butanediol. Among the acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium.
For this purpose, any bland fixed oil may be employed including synthetic mono- or di-glycerides. Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions. These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant, such as carboxymethyl cellulose or similar dispersing agents that are commonly used in the formulation of pharmaceutically acceptable dosage forms including emulsions and suspensions. Other commonly used surfactants, such as Tweens, Spans and other emulsifying agents or bioavailability enhancers which are commonly used in the manufacture of pharmaceutically acceptable solid, liquid, or other dosage forms may also be used for the purposes of formulation.
Pharmaceutically acceptable compositions of this invention may be orally administered in any orally acceptable dosage form including, but not limited to, capsules, tablets, aqueous suspensions or solutions. In the case of tablets for oral use, carriers commonly used include lactose and corn starch. Lubricating agents, such as magnesium stearate, are also typically added. For oral administration in a capsule form, useful diluents include lactose and dried cornstarch. When aqueous suspensions are required for oral use, the active ingredient is combined with emulsifying and suspending agents. If desired, certain sweetening, flavoring or coloring agents may also be added.
Alternatively, pharmaceutically acceptable compositions of this invention may be administered in the form of suppositories for rectal administration. These can be prepared by mixing the agent with a suitable non-irritating excipient that is solid at room temperature but liquid at rectal temperature and therefore will melt in the rectum to release the drug. Such materials include cocoa butter, beeswax and polyethylene glycols.
Pharmaceutically acceptable compositions of this invention may also be administered topically, especially when the target of treatment includes areas or organs readily accessible by topical application, including diseases of the eye, the skin, or the lower intestinal tract. Suitable topical formulations are readily prepared for each of these areas or organs.
Topical application for the lower intestinal tract can be effected in a rectal suppository formulation (see above) or in a suitable enema formulation. Topically-transdermal patches may also be used.
For topical applications, provided pharmaceutically acceptable compositions may be formulated in a suitable ointment containing the active component suspended or dissolved in one or more carriers. Carriers for topical administration of compounds of this invention include, but are not limited to, mineral oil, liquid petrolatum, white petrolatum, propylene glycol, polyoxyethylene, polyoxypropylene compound, emulsifying wax and water. Alternatively, provided pharmaceutically acceptable compositions can be formulated in a suitable lotion or cream containing the active components suspended or dissolved in one or more pharmaceutically acceptable carriers. Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol and water.
For ophthalmic use, provided pharmaceutically acceptable compositions may be formulated as micronized suspensions in isotonic, pH adjusted sterile saline, or, preferably, as solutions in isotonic, pH adjusted sterile saline, either with or without a preservative such as benzylalkonium chloride. Alternatively, for ophthalmic uses, the pharmaceutically acceptable compositions may be formulated in an ointment such as petrolatum.
Pharmaceutically acceptable compositions of this invention may also be administered by nasal aerosol or inhalation. Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other conventional solubilizing or dispersing agents.
Most preferably, pharmaceutically acceptable compositions of this invention are formulated for oral administration. Such formulations may be administered with or without food. In some embodiments, pharmaceutically acceptable compositions of this invention are administered without food. In other embodiments, pharmaceutically acceptable compositions of this invention are administered with food.
The amount of compounds of the present invention that may be combined with the carrier materials to produce a composition in a single dosage form will vary depending upon the host treated and the particular mode of administration. Preferably, provided compositions should be formulated so that a dosage of between 0.01-100 mg/kg body weight/day of the inhibitor can be administered to a patient receiving these compositions.
It should also be understood that a specific dosage and treatment regimen for any particular patient will depend upon a variety of factors, including the activity of the specific compound employed, the age, body weight, general health, sex, diet, time of administration, rate of excretion, drug combination, and the judgment of the treating physician and the severity of the particular disease being treated. The amount of a compound of the present invention in the composition will also depend upon the particular compound in the composition.
Uses of Compounds and Pharmaceutically Acceptable Compositions
Compounds and compositions described herein are generally useful for the modulating of activity of one or more enzymes involved in epigenetic regulation.
Epigenetics is the study of heritable changes in gene expression caused by mechanisms other than changes in the underlying DNA sequence. Molecular mechanisms that play a role in epigenetic regulation include DNA methylation and chromatin/histone modifications. Histone methylation, in particular, is critical in many epigenetic phenomena.
Chromatin, the organized assemblage of nuclear DNA and histone proteins, is the basis for a multitude of vital nuclear processes including regulation of transcription, replication, DNA-damage repair and progression through the cell cycle. A number of factors, such as chromatin-modifying enzymes, have been identified that play an important role in maintaining the dynamic equilibrium of chromatin (Margueron, et al. (2005) Curr. Opin. Genet. Dev. 15:163-176).
Histones are the chief protein components of chromatin. They act as spools around which DNA winds, and they play a role in gene regulation. There are a total of six classes of histones (H1, H2A, H2B, H3, H4, and H5) organized into two super classes: core histones (H2A, H2B, H3, and H4) and linker histones (H1 and H5). The basic unit of chromatin is the nucleosome, which consists of about 147 base pairs of DNA wrapped around the histone octamer, consisting of two copies each of the core histones H2A, H2B, H3, and H4 (Luger, et al. (1997) Nature 389:251-260).
Histones, particularly residues of the amino termini of histones H3 and H4 and the amino and carboxyl termini of histones H2A, H2B and H1, are susceptible to a variety of post-translational modifications including acetylation, methylation, phosphorylation, ribosylation, sumoylation, ubiquitination, citrullination, deimination, and biotinylation. The core of histones H2A and H3 can also be modified. Histone modifications are integral to diverse biological processes such as gene regulation, DNA repair, and chromosome condensation.
The present disclosure provides compounds and compositions for modulating activity of histone methyl modifying enzymes. Histone methyl modifying enzymes are key regulators of cellular and developmental processes. Histone methyl modifying enzymes may be characterized as either histone methyl transferases or histone demethylases. Histone demethylase enzymes have modules that mediate binding to methylated residues. For example, multiple demethylases contain a Tudor domain (e.g., JMJD2C/GASC1) or a PHD domain (e.g., JARID1C/SMCX, PHF8).
The lysine specificities of many histone methyltransferases have been characterized. For example SET7/9, SMYD3, and MLL1-5 are specific for H3K4. SUV39H1, DIM-5, and G9a are specific for H3K9. SET8 is specific for H4K20.
DOT1 is an example of a non-SET domain containing histone methylase. DOT1 methylates H3 on lysine 79.
Just as histone methylases have been shown to regulate transcriptional activity, chromatin structure, and gene silencing, demethylases have also been discovered which impact gene expression. LSD1 was the first histone lysine demethylase to be characterized. This enzyme displays homology to FAD-dependent amine oxidases and acts as a transcriptional corepressor of neuronal genes (Shi et al., Cell 119:941-953, 2004). Additional demethylases defining separate demethylase families have been discovered, including JHDM1 (or KDM2), JHDM2 (or KDM3), JMJD2 (or KDM4), JARID (or KDM5), JMJD3 (or KDM6), and JMJD6 families (Lan et al., Curr. Opin. Cell Biol. 20(3):316-325, 2008).
Demethylases act on specific lysine residues within substrate sequences and discriminate between the degree of methylation present on a given residue. For example, LSD1 removes mono- or dimethyl-groups from H3K4. Members of the JARID1A-D family remove trimethyl groups from H3K4. UTX and JMJD3 demethylate H3K27, counteracting effects of EZH2 methylase activity. Substrate specificities of other demethylases have been characterized (see Shi, Nat. Rev. 8:829-833, 2007).
One class of histone methylases is characterized by the presence of a SET domain, named after proteins that share the domain, Su(var)3-9, enhancer of zeste [E(Z)], and trithorax. A SET domain includes about 130 amino acids. SET domain-containing methylase families include SUV39H1, SET1, SET2, EZH2, RIZ1, SMYD3, SUV4-20H1, SET7/9, and PR-SET7/SET8 families (reviewed in Dillon et al., Genome Biol. 6:227, 2005). Members of a family typically include similar sequence motifs in the vicinity of and within the SET domain. The human genome encodes over 50 SET domain-containing histone protein methylases, any of which can be used in an assay described herein.
EZH2 is an example of a human SET-domain containing methylase. EZH2 associates with EED (Embryonic Ectoderm Development) and SUZ12 (suppressor of zeste 12 homolog) to form a complex known as PRC2 (Polycomb Group Repressive Complex 2) having the ability to trimethylate histone H3 at lysine 27 (Cao and Zhang, Mol. Cell 15:57-67, 2004). PRC2 complexes can also include RBAP46 and RBAP48 subunits.
The oncogenic activities of EZH2 have been shown by a number of studies. In cell line experiments, over-expression of EZH2 induces cell invasion, growth in soft agar, and motility while knockdown of EZH2 inhibits cell proliferation and cell invasion (Kleer et al., 2003, Proc. Nat. Acad. Sci. USA 100:11606-11611; Varambally et al., (2002), "The polycomb group protein EZH2 is involved in progression of prostate cancer," Nature 419, 624-629). It has been shown that EZH2 represses the expression of several tumor supressors, including E-cadherin, DAB2IP and RUNX3 among others. In xenograft models, EZH2 knockdown inhibits tumor growth and metastasis. Recently, it has been shown that down modulation of EZH2 in murine models blocks prostate cancer metastasis (Min et al., "An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-kappaB," Nat Med. 2010 March; 16(3):286-94). EZH2 overexpression is associated with aggressiveness of certain cancers such as breast cancer (Kleer et al., Proc. Nat. Acad. Sci. USA 100:11606-11611, 2003). Recent studies also suggest that prostate cancer specific oncogenic fusion gene TMPRSS2-ERG induces repressive epigenetic programs via direct activation of EZH2 (Yu et al., "An Integrated Network of Androgen Receptor, Polycomb, and TMPRSS2-ERG Gene Fusions in Prostate Cancer Progression," Cancer Cell. 2010 May 18; 17(5):443-454).
In some embodiments, compounds of the present invention modulate the activity of one or more enzymes involved in epigenetic regulation. In some embodiments, compounds of the present invention modulate the activity of a histone methyl modifying enzyme, or a mutant thereof. In some embodiments, compounds of the present invention modulate EZH2 activity. In some embodiments, compounds of the present invention down-regulate or suppress the activity of EZH2. In some embodiments, compounds of the present invention are antagonists of EZH2 activity.
In some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with a histone methyl modifying enzyme. Accordingly, in some embodiments, the present invention provides a method of modulating a disease and/or disorder associated with a histone methyl modifying enzyme. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with a histone methyl modifying enzyme comprising the step of administering a compound or composition of Formula II.
In some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with overexpression of EZH2. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with overexpression of EZH2 comprising the step of administering a compound or composition of Formula II. In some embodiments, the above method additionally comprises the preliminary step of determining if the subject is overexpressing EZH2.
In some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with cellular proliferation. In some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with misregulation of cell cycle or DNA repair. In some embodiments, compounds and compositions of the present invention are useful in treating cancer. Exemplary types of cancer include breast cancer, prostate cancer, colon cancer, renal cell carcinoma, glioblastoma multiforme cancer, bladder cancer, melanoma, bronchial cancer, lymphoma and liver cancer.
The study of EZH2 deletions, missense and frameshift mutations suggest that EZH2 functions as a tumor suppressor in blood disorders such as myelodysplastic syndromes (MDS) and myeloid malignancies (Ernst et al., Nat Genet. 2010 August; 42(8):722-6; Nikoloski et al., Nat Genet. 2010 August; 42(8):665-7). Accordingly, in some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with the presence of a mutant form of EZH2. In some embodiments, compounds and compositions of the present invention are useful in treating diseases and/or disorders associated with the presence of Y641N EZH2. In some embodiment, the disease or disorder associated with the presence of a mutant form of EZH2 is a human B cell lymphoma. In some embodiments, the disease and/or disorder associated with the presence of Y641N EZH2 is follicular lymphoma or diffuse large-B-cell lymphoma. In some embodiments, compounds or compositions of the present invention are useful in treating blood disorders, such as myelodysplastic syndromes, leukemia, anemia and cytopenia. Sneeringer et al., "Coordinated activities of wild-type plus mutant EZH2 drive tumor-associated hypertrimethylation of lysine 27 on histone H3 (H3K27) in human B-cell lymphomas," Proceedings of the National Academy of Sciences, PNAS Early Edition published ahead of print on Nov. 15, 2010.
In some embodiments, the present invention provides a method of reducing the activity of EZH2 in a subject comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of reducing the activity of wide-type EZH2 in a subject comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of reducing the activity of a mutant form of EZH2 in a subject comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of reducing the activity of a mutant form of EZH2 in a subject comprising the step of administering a compound or composition of Formula II, wherein the mutant form of EZH2 is Y641N EZH2. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with EZH2 comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with wide-type EZH2 comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with a mutant form of EZH2 comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with a mutant form of EZH2 comprising the step of administering a compound or composition of Formula II, wherein the mutant form of EZH2 is Y641N EZH2. In some embodiments, the above method additionally comprises the preliminary step of determining if the subject is expressing a mutant form of EZH2, such as Y641N EZH2 In some embodiments, the present invention provides a method of reducing the activity of a mutant form of EZH2, such as Y641N EZH2, in a subject in need thereof comprising the step of administering a compound or composition of Formula II. In some embodiments, the present invention provides a method of treating a subject suffering from a disease and/or disorder associated with a mutant form of EZH2 comprising the step of administering a compound or composition of Formula II. In some embodiments, the above method additionally comprises the preliminary step of determining if the subject is expressing a mutant form of EZH2, such as Y641N EZH2. In some embodiments, that determination is made by determining if the subject has increased levels of histone H3 Lys-27-specific trimethylation (H3K27me3), as compared to a subject known not to express a mutant form of EZH2.
EQUIVALENTS
The representative examples that follow are intended to help illustrate the invention, and are not intended to, nor should they be construed to, limit the scope of the invention. Indeed, various modifications of the invention and many further embodiments thereof, in addition to those shown and described herein, will become apparent to those skilled in the art from the full contents of this document, including the examples that follow and the references to the scientific and patent literature cited herein. It should further be appreciated that the contents of those cited references are incorporated herein by reference to help illustrate the state of the art.
It will be appreciated that for compound preparations described herein, when reverse phase HPLC is used to purify a compound, a compound may exist as an acid addition salt. In some embodiments, a compound may exist as a formic acid or mono-, di-, or tri-trifluoroacetic acid salt.
It will further be appreciated that the present invention contemplates individual compounds described herein. Where individual compounds exemplified are isolated and/or characterized as a salt, for example, as a trifluoroacetic acid salt, the present invention contemplates a free base of the salt, as well as other pharmaceutically acceptable salts of the free base.
The following examples contain important additional information, exemplification and guidance that can be adapted to the practice of this invention in its various embodiments and the equivalents thereof.
Procedures for preparing the compounds exemplified below, as well as additional compounds/intermediates in the synthetic schemes can be found in International Application No. PCT/US2013/025639, the contents of which are incorporated herein by reference.
EXAMPLES
As depicted in the Examples below, in certain exemplary embodiments, compounds are prepared according to the following general procedures. It will be appreciated that, although the synthetic methods and Schemes depict the synthesis of certain compounds of the present invention, the following methods and other methods known to one of ordinary skill in the art can be applied to all compounds and subclasses and species of each of these compounds, as described herein.
Unless otherwise noted, all solvents, chemicals, and reagents were obtained commercially and used without purification. The .sup.1H NMR spectra were obtained in CDCl.sub.3, d.sub.6-DMSO, CD.sub.3OD, or d.sub.6-acetone at 25.degree. C. at 300 MHz on an OXFORD (Varian) with chemical shift (.delta., ppm) reported relative to TMS as an internal standard. HPLC-MS chromatograms and spectra were obtained with Shimadzu LCMS-2020 system. Chiral analysis and purification were obtained with Yilite P270.
Example 1
Synthesis of Compounds 327 and 346 and Related Compounds and Intermediates
The title compounds of this Example and other related compounds were prepared according to the following general scheme. In addition, where indicated, modifications of this scheme are disclosed for the synthesis of still additional related compounds of the invention and intermediates thereof.
##STR00003## ##STR00004##
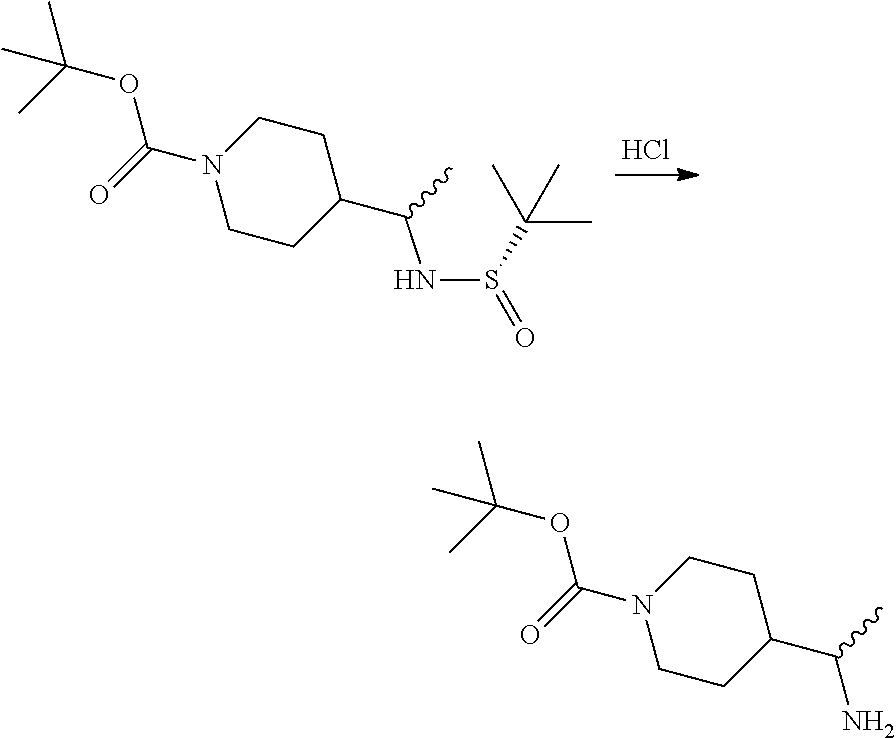
Step 1: (S,E)-tert-butyl 4-(((tert-butylsulfinyl)imino)methyl)piperidine-1-carboxylate
##STR00005## To a round bottomed flask charged with a magnetic stir bar was added (S)-2-methylpropane-2-sulfinamide (20.46 g, 169 mmol), tert-butyl 4-formylpiperidine-1-carboxylate (30 g, 141 mmol), DCM (300 mL), and Ti(OEt).sub.4 (59.0 ml, 281 mmol). The solution was stirred at room temperature for 3 h before it was quenched with brine (80 mL). The solution was stirred for 30 minutes before filtering. The filter cake was washed with DCM and the filtrate was placed in a separatory funnel and washed with water. The organics layer was dried over Na.sub.2SO.sub.4, filtered, and concentrated in vacuo. The crude residue solidified to the title compound (29 g, 92 mmol, 65.1% yield) m/z 217.
The intermediate shown in the following table was prepared according to the general procedure outlined in Step 1 using the appropriate starting materials and modifications.
TABLE-US-00001 Name Structure m/z (S,E)-2-methyl-N- ((tetrahydro-2H- pyran-4-yl)methylene) propane-2-sulfinamide ##STR00006##
Step 2: Tert-butyl 4-((S)-1-((R or S)-1,1-dimethylethylsulfinamido)ethyl) piperidine-1-carboxylate
##STR00007## To a round bottomed flask charged with a magnetic stir bar was added (S,E)-tert-butyl 4-((tert-butylsulfinylimino)methyl)piperidine-1-carboxylate (36.4 g, 115 mmol), DCM (400 mL), and the solution was cooled to 0.degree. C. in an ice bath with stirring. To this solution was added MeMgBr (77 ml, 230 mmol) (3M in diethyl ether) and the reaction stirred for 4 h while warming to room temperature. The reaction was carefully quenched via the addition of saturated aqueous NH.sub.4Cl. The solid were broken up by the addition of 1N HCl. The layers were separated and the aqueous phase was extracted with DCM. The combined organics phase was dried over Na.sub.2SO.sub.4, filtered, and concentrated in vacuo to afford the title compound (29 g, >9:1 dr) which is used without further purification in the next step.
The intermediate shown in the following table was prepared according to the general procedure outlined in Step 2 using the appropriate starting materials and modifications.
TABLE-US-00002 Name Structure m/z (S)-2-methyl-N-((R or S)-1- (tetrahydro-2H-pyran-4- yl)ethyl)propane- 2-sulfinamide ##STR00008## 234
Step 3: (R or S)-tert-butyl 4-(1-aminoethyl)piperidine-1-carboxylate
##STR00009## To a 1 L round bottomed flask charged with a magnetic stir bar was added crude tert-butyl 4-((S)-1-((S)-1,1-dimethylethylsulfinamido)ethyl)piperidine-1-carboxylate (29 g) was taken up in MeOH (200 mL) before addition of a 4 N solution of HCl in 1,4-dioxane (24.06 ml, 96 mmol). The resulting solution was then stirred at room temperature for 1 h at rt. The methanol was then removed in vacuo to afford viscous oil which was treated with sat'd aqueous NaHCO.sub.3 (.about.500 mL) and extracted with ethyl acetate (2.times.500 mL). This organic phase was combined, dried with MgSO.sub.4, filtered, and solvent was then removed in vacuo affording the title compound (22 g) which was used without further purification.
The intermediate shown in the following table was prepared according to the general procedure outlined in Step 3 using the appropriate starting materials.
TABLE-US-00003 Name Structure m/z (R or S)-1-(tetrahydro- 2H-pyran-4-yl)ethanamine ##STR00010## 130
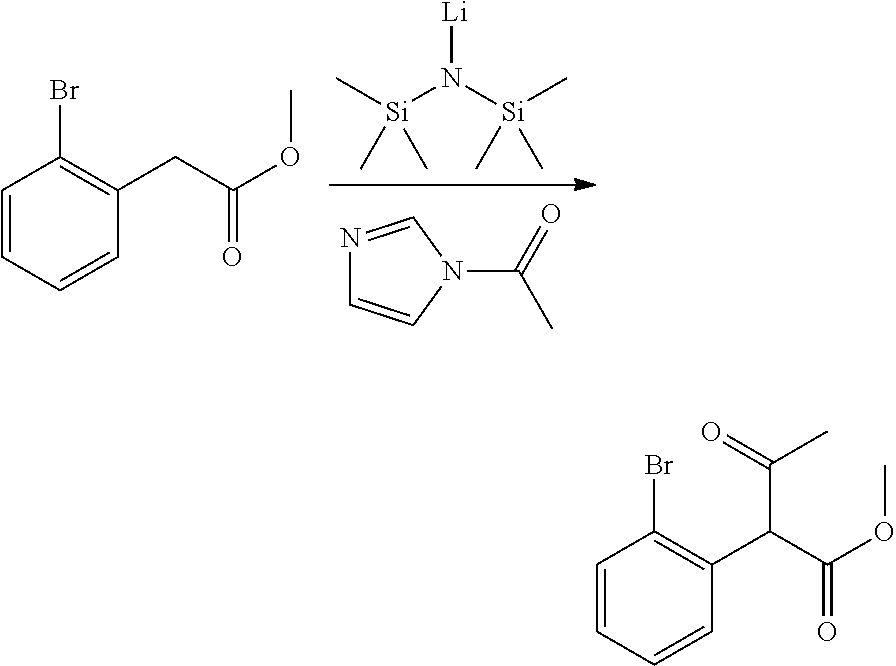
Step 4: Methyl 2-(2-bromophenyl)-3-oxobutanoate
##STR00011## A round bottomed flask was charged with a magnetic stir bar and methyl 2-(2-bromophenyl)acetate (25 g, 109 mmol) and THF (50 mL). This solution was cooled to -78.degree. C. before drop wise addition of a 1M solution of LiHMDS in THF (218 ml, 218 mmol). The reaction was stirred for 30 min at -78.degree. C. before addition of 1-(1H-imidazol-1-yl)ethanone (14.42 g, 131 mmol) dissolved in a mixture of THF:DMF (112 mL THF, 24 mL DMF). The solution was stirred for 1 h before quenching with sat'd aqueous NH.sub.4Cl (.about.250 mL) and diluting with EtOAc. The layers were separated and the aqueous phase was extracted with EtOAc (.about.2.times.250 mL). The combined organic extract was washed with brine, dried over Na.sub.2SO.sub.4, filtered, and concentrated in vacuo. The crude residue was purified via silica gel chromatography using an eluent of ethyl acetate/hexanes (10:1) to afford methyl 2-(2-bromophenyl)-3-oxobutanoate (32.5 g, 102 mmol, 93% yield).
The intermediates shown in the following table were prepared according to the general procedure outlined in Step 4 using the appropriate starting materials.
TABLE-US-00004 Name Structure m/z methyl 2-(2-bromo-4- chlorophenyl)-3-oxobutanoate ##STR00012## 304 methyl 2-(2-bromo-4- methoxyphenyl)-3- oxobutanoate ##STR00013## 302 methyl 2-(2-bromo-4- fluorophenyl)-3-oxobutanoate ##STR00014## 289
Step 5: (R or S, E and Z)-tert-butyl 4-(1-(3-(2-bromophenyl)-4-methoxy-4-oxobut-2-en-2-ylamino)ethyl)piperidin- e-1-carboxylate
##STR00015## To a round bottomed flask was added (R or S)-tert-butyl 4-(1-aminoethyl)piperidine-1-carboxylate (9.35 g, 40.9 mmol), EtOH (75 mL), and methyl 2-(2-bromophenyl)-3-oxobutanoate (7.40 g, 27.3 mmol) (from Step 4). To this solution was added AcOH (1.563 ml, 27.3 mmol) and the reaction was heated overnight at 85.degree. C. before cooling to room temperature and concentrating. The crude residue was purified via silica gel chromatography (330 g, 100% hexanes to 25% EA in hexanes) to afford the title compound (6.45 g, 13.40 mmol, 49.1% yield).
The intermediates shown in the following table were prepared according to the general procedure outlined in Step 5 using the appropriate starting materials.
TABLE-US-00005 Name Structure m/z (R or S,Z)-methyl 2- (2-bromophenyl)-3- ((1-(tetrahydro-2H- pyran-4-yl)ethyl) amino)but-2-enoate ##STR00016## 383 (R or S,Z)-methyl 2- (2-bromo-4- chlorophenyl)-3- ((1-(tetrahydro- 2H-pyran- 4-yl)ethyl)amino) but-2-enoate ##STR00017## 417 (R or S,Z)-methyl 2- (2-bromo-4- chlorophenyl)- 3-((1-(tetrahydro-2H- pyran-4-yl)ethyl) amino)but-2-enoate ##STR00018## 417 (R or S,Z)-methyl 2- (2-bromo-4- fluorophenyl)- 3-((1-(tetrahydro-2H- pyran-4-yl)ethyl) amino)but-2-enoate ##STR00019## 401
Step 6: (R or S)-methyl 1-(1-(1-(tert-butoxycarbonyl)piperidin-4-yl)ethyl)-2-methyl-1H-indole-3-c- arboxylate
##STR00020## A 250 mL round bottom flask was charged with a magnetic stir bar, (R or S,Z)-tert-butyl 4-(1-(3-(2-bromophenyl)-4-methoxy-4-oxobut-2-en-2-ylamino)ethyl)piperidin- e-1-carboxylate (3.33 g, 6.92 mmol), RuPhos Pre-catalyst II (Methanesulfonato(2-dicyclohexylphosphino-2',6'-di-i-propoxy-1,1'-bipheny- l)(2-amino-1,1'-biphenyl-2-yl) palladium(II)) (0.463 g, 0.553 mmol), dicyclohexyl(2',6'-diisopropoxybiphenyl-2-yl)phosphine (0.387 g, 0.830 mmol), anhydrous 1,4-dioxane (27.7 ml, 6.92 mmol), and sodium methoxide (0.561 g, 10.38 mmol). The reaction mixture was purged and back-filled with nitrogen and heated to 100.degree. C. with stirring overnight before being allowed to cool to rt. The reaction was diluted with ethyl acetate (.about.100 ml) and the mixture was filtered through a bed of diatomaceous earth. The filtrate was pre-absorbed onto silica gel (.about.30 g) and purified via silica gel chromatography (120 g) using ethyl acetate/hexanes (1:1) as eluent to afford the title compound (2.01 g, 4.77 mmol, 68.9% yield).
The intermediates shown in the following table were prepared according to the general procedure outlined in Step 6 using the appropriate starting materials.
TABLE-US-00006 Name Structure m/z (R or S)-methyl 2- methyl-1-(1-(tetrahydro- 2H-pyran- 4-yl)ethyl)-1H-indole-3- carboxylate ##STR00021## 302 (R or S)-methyl 6- chloro-2-methyl-1-(1- (tetrahydro-2H-pyran-4- yl)ethyl)-1H-indole-3- carboxylate ##STR00022## 337 (R or S)-methyl 6- methoxy-2-methyl-1-(1- (tetrahydro-2H-pyran-4- yl)ethyl)-1H-indole-3- carboxylate ##STR00023## 332 (R or S)-methyl 6-fluoro- 2-methyl-1-(1- (tetrahydro-2H-pyran-4- yl)ethyl)-1H-indole-3- carboxylate ##STR00024## 320
Step 7: (R or S)-2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole-3-carboxylic acid
##STR00025## A 1 L round bottom flask was charged with a magnetic stir bar, (R or S)-methyl 2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole-3-carboxylate (11.60 g, 38.5 mmol), ethanol (96 ml, 38.5 mmol), and 6 N aqueous NaOH (64.1 ml, 385 mmol). The flask was fitted with a reflux condenser and heated to reflux for 6 h before being allowed to cool to rt. The volatiles were removed in vacuo and the resulting mixture was poured into 10% HCl (.about.300 mL). A precipitate formed which was collected via vacuum filtration using a Buchner funnel. The filter cake was rinsed with an additional portion of water (.about.200 mL), collected, and dried under vacuum to afford the title compound (10.87 g, 35.9 mmol, 93% yield) as an off-white solid.
The intermediates shown in the following table were prepared according to the general procedure outlined in Step 7 using the appropriate starting materials.
TABLE-US-00007 Name Structure m/z (R or S)-2- methyl-1-(1- (tetrahydro- 2H-pyran-4- yl)ethyl)- 1H-indole-3- carboxylic acid ##STR00026## 287 (R or S)-6- chloro-2-methyl- 1-(1-(tetrahydro- 2H-pyran-4- yl)ethyl)-1H- indole-3- carboxylic acid ##STR00027## 321 (R or S)-6- methoxy-2- methyl-1-(1- (tetrahydro- 2H-pyran- 4-yl)ethyl)- 1H-indole-3- carboxylic acid ##STR00028## 317 (R or S)-6- fluoro-2- methyl-1- (1-(tetrahydro- 2H-pyran-4- yl)ethyl)-1H- indole-3- carboxylic acid ##STR00029## 306 (R or S)-2- methyl-6- (pyridin- 3-yl)-1-(1- (tetrahydro-2H- pyran-4-yl) ethyl)-1H-indole- 3-carboxylic acid ##STR00030## 365
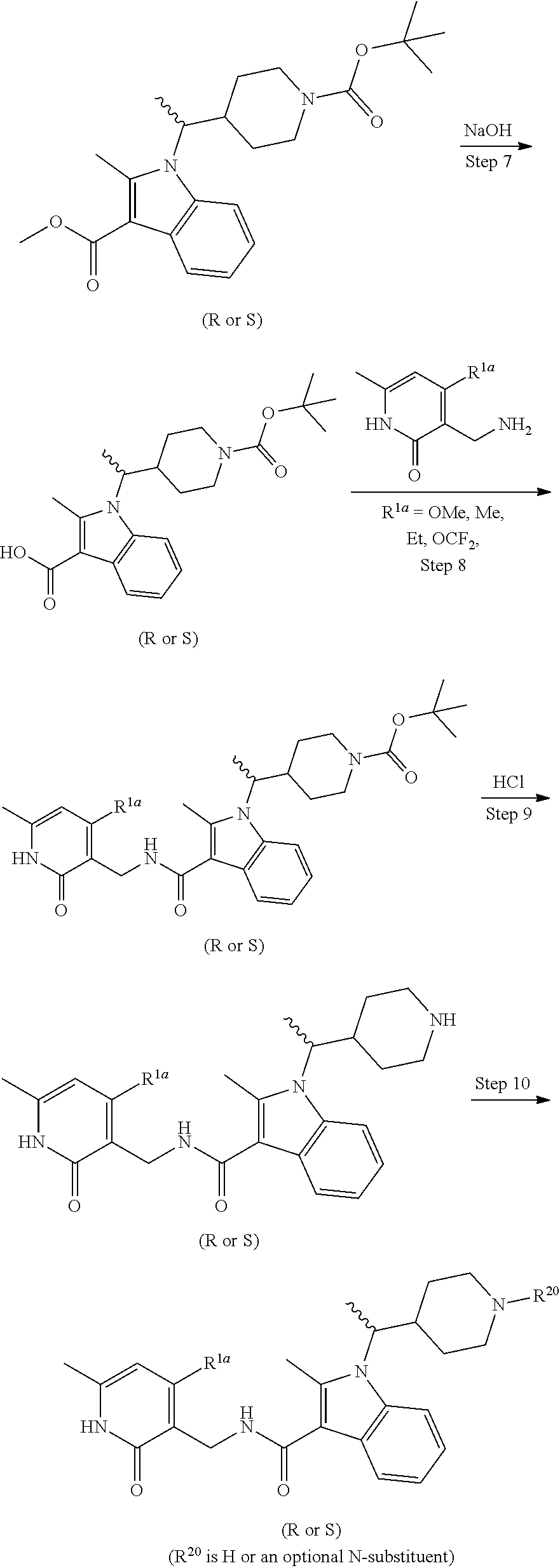
Step 8: (R or S)-tert-butyl 4-(1-(3-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methylcarbamoy- l)-2-methyl-1H-indol-1-yl)ethyl)piperidine-1-carboxylate (Compound 327)
##STR00031## A 250 mL round bottom flask was charged with a magnetic stir bar, (R or S)-1-(1-(1-(tert-butoxycarbonyl)piperidin-4-yl)ethyl)-2-methyl-1H-indole-- 3-carboxylic acid (1.950 g, 5.05 mmol), 3-(aminomethyl)-4-methoxy-6-methylpyridin-2(1H)-one hydrochloride (2.065 g, 10.09 mmol), DMF (25.2 ml, 5.05 mmol), Hunig's base (3.52 ml, 20.18 mmol). The reaction mixture was cooled to 0.degree. C. and COMU (2.16 g, 5.05 mmol) was added. The reaction was allowed to stir overnight to room temperature. The reaction mixture was diluted with water and extracted with EtOAc. The combined organic extract was washed with brine, dried with MgSO.sub.4, filtered and conc. in vacuo to afford the crude material which was purified via silica gel chromatography (120 g) using MeOH/ethyl acetate (1:5) as eluent to afford the title compound (1.86 g, 3.29 mmol, 65.3% yield). LCMS 537 (M+1).sup.+1H NMR (400 MHz, DMSO-d.sub.6) .delta.=11.83-11.71 (m, 1H), 7.80 (br. s., 1H), 7.73 (d, J=7.6 Hz, 1H), 7.62 (d, J=7.8 Hz, 1H), 7.06 (td, J=7.1, 14.4 Hz, 2H), 6.21 (s, 1H), 4.32 (br. s., 2H), 4.16 (br. s., 1H), 4.02 (br. s., 1H), 3.85 (s, 3H), 3.75 (br. s., 1H), 2.70 (br. s., 1H), 2.58 (s, 3H), 2.37 (br. s., 1H), 2.21 (s, 3H), 1.90 (d, J=12.9 Hz, 1H), 1.53 (d, J=6.9 Hz, 3H), 1.35 (s, 10H), 1.21 (br. s., 1H), 0.89 (d, J=8.7 Hz, 1H), 0.67 (d, J=11.8 Hz, 1H).
The compounds shown in the following table were prepared according to the general procedure outlined in Step 8 using the appropriate starting materials. The structures of the compounds are shown in FIG. 1.
TABLE-US-00008 Compound Number Name .sup.1H NMR m/z 435 (R or S)-tert-butyl 4-(1-(3-(((4,6- 521 dimethyl-2-oxo-1,2- dihydropyridin-3- yl)methyl)carbamoyl)-2-methyl- 1H-indol-1-yl)ethyl)piperidine-1- carboxylate 436 (R or S)-tert-butyl 4-(1-(3-(((4- 573 (difluoromethoxy)-6-methyl-2- oxo-1,2-dihydropyridin-3- yl)methyl)carbamoyl)-2-methyl- 1H-indol-1-yl)ethyl)piperidine-1- carboxylate 437 (R or S)-tert-butyl 4-(1-(3-(((4- 535 ethyl-6-methyl-2-oxo-1,2- dihydropyridin-3- yl)methyl)carbamoyl)-2-methyl- 1H-indol-1-yl)ethyl)piperidine-1- carboxylate 298 (R or S)-N-((4-methoxy-6- (400 MHz, DMSO-d6) .delta. 11.60 (s, 438 methyl-2-oxo-1,2-dihydropyridin- 1H), 7.73-7.62 (m, 3H), 7.60 (d, 3-yl)methyl)-2-methyl-1-(1- 2H) 7.07-7.05 (m, 2H), 6.15 (s, 1H) (tetrahydro-2H-pyran-4-yl)ethyl)- 4.33 (s, 1H), 4.21-4.11 (m, 1H), 1H-indole-3-carboxamide 3.92 (br. d., 1H), 3.65 (d, 1H), 3.34- 3.32 (m, 1H), 3.02 (t, 1H), 2.61 (s, 3H), 2.48-2.44 (m, 1H), 2.20 (s, 3H), 1.84-1.81 (m, 1H), 1.54 (d, 3H), 1.40-1.38 (m, 12H), 1.25-1.22 (m, 1H), 1.08-1.04 (m, 1H), 0.86 (br. s., 1H), 0.58 (br. d., 1H) 300 (R or S)-6-fluoro-N-((4-methoxy- (400 MHz, DMSO-d6) .delta. = 11.57 456 6-methyl-2-oxo-1,2- (br. s., 1 H), 7.75-7.67 (m, 2 H), dihydropyridin-3-yl)methyl)-2- 7.48 (d, J = 10.7 Hz, 1 H), 6.90 (t, methyl-1-(1-(tetrahydro-2H- J = 8.5 Hz, 1 H), 6.13 (s, 1 H), 4.29 pyran-4-yl)ethyl)-1H-indole-3- (d, J = 4.5 Hz, 2 H), 4.12 (br. s., 1 carboxamide H), 3.94-3.87 (m, 1 H), 3.83 (s, 3 H), 3.64 (dd, J = 3.6, 10.9 Hz, 1 H), 3.35 (br. s., 1 H), 3.05 (br. s., 1 H), 2.56 (s, 3 H), 2.45-2.37 (m, 1 H), 2.18 (s, 3 H), 1.81 (d, J = 12.7 Hz, 1 H), 1.50 (d, J = 6.9 Hz, 3 H), 1.40- 1.29 (m, 1 H), 1.11-0.99 (m, 1 H), 0.61 (br. s., 1 H) 314 (R or S)-6-chloro-N-((4-methoxy- (400 MHz, DMSO-d6) .delta. 11.57 (s, 1 472 6-methyl-2-oxo-1,2 H), 7.75 (s, 2 H), 7.66 (d, J = 8.9 dihydropyridin-3-yl)methyl)-2- Hz, 1 H), 7.08 (d, J = 8.5 Hz, 1 H), methyl-1-(1-(tetrahydro-2H- 6.14 (s, 1 H), 4.30 (d, J = 4.5 Hz, 2 pyran-4-yl)ethyl)-1H-indole-3- H), 4.21-4.05 (m, 2 H), 3.91 (d, carboxamide J = 11.4 Hz, 1 H), 3.85 (s, 3 H), 3.65 (d, J = 10.5 Hz, 1 H), 3.02 (t, J = 11.3 Hz, 1 H), 2.58 (s, 3 H), 2.46- 2.31 (m, 1 H), 2.19 (s, 3 H), 1.82 (d, J = 12.0 Hz, 1 H), 1.59-1.45 (m, 4 H), 1.44-1.29 (m, 1 H), 0.57 (d, J = 12.9 Hz, 1 H) 321 (R or S)-6-methoxy-N-((4- (400 MHz, DMSO-d6) .delta. = 11.59 (s, 468 methoxy-6-methyl-2-oxo-1,2- 1 H), 7.67-7.59 (m, 2 H), 7.03 (s, dihydropyridin-3-yl)methyl)-2- 1 H), 6.75-6.68 (m, 1 H), 6.14 (s, methyl-1-(1-(tetrahydro-2H- 1 H), 4.30 (d, J =5.1 Hz, 2 H), 4.10 pyran-4-yl)ethyl)-1H-indole-3- (dd, J = 7.5, 10.4 Hz, 1 H), 3.91 carboxamide (dd, J = 3.0, 11.3 Hz, 1 H), 3.83 (s, 3 H), 3.80-3.76 (m, 3 H), 3.68- 3.60 (m, 1 H), 3.38-3.32 (m, 1 H), 3.10-3.00 (m, 1 H), 2.56 (s, 3 H), 2.19 (s, 3 H), 1.83 (d, J = 12.7 Hz, 1 H), 1.55-1.43 (m, 4 H), 1.34 (br. s., 1 H), 1.10-0.96 (m, 1 H), 0.62 (d, J = 13.4 Hz, 1 H) 335 (R or S)-N-((4- 474 (difluoromethoxy)-6-methyl-2- oxo-1,2-dihydropyridin-3- yl)methyl)-2-methyl-1-(1- (tetrahydro-2H-pyran-4-yl)ethyl)- 1H-indole-3-carboxamide 394 (R or S)-N-((4,6-dimethyl-2-oxo- 422 1,2-dihydropyridin-3-yl)methyl)- 2-methyl-1-(1-(tetrahydro-2H- pyran-4-yl)ethyl)-1H-indole-3- carboxamide 442 (R or S)-6-chloro-N-((4,6- 456 dimethyl-2-oxo-1,2- dihydropyridin-3-yl)methyl)-2- methyl-1-(1-(tetrahydro-2H- pyran-4-yl)ethyl)-1H-indole-3- carboxamide
Step 9: (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide hydrochloride (Compound 326)
##STR00032## A 250 mL round bottom flask was charged with a magnetic stir bar, (R or S)-tert-butyl 4-(1-(3-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methylcarbamoy- l)-2-methyl-1H-indol-1-yl)ethyl)piperidine-1-carboxylate (Compound 327) (1.850 g, 3.45 mmol), MeOH (13.79 ml, 3.45 mmol), and HCl (2.59 ml, 10.34 mmol) (4 N in dioxane). The reaction was allowed to stir at rt for 6 h before being conc. in vacuo to afford the title compound (1.65 g, 3.14 mmol, 91% yield). LCMS 437 (M+1).sup.+.
The compound shown in the following table was prepared according to the general procedure outlined in Step 9 using the appropriate starting materials. The structure of this compound is shown in FIG. 1.
TABLE-US-00009 Compound Number Name .sup.1H NMR m/z 376 (R or S)-1-(1-(1- (400 MHz, DMSO-d.sub.6) .delta. 12.27-12.10 (m, 1 476 (azetidin-3-yl)piperidin- H), 11.96-11.72 (m, 1 H), 9.80 (br. s., 1 4-yl)ethyl)-N-((4,6- H), 9.19 (br. s., 2 H), 7.89-7.67 (m, 2 H), dimethyl-2-oxo-1,2- 7.62 (d, J = 7.6 Hz, 1 H), 7.09 (quin, J = 6.6 dihydropyridin-3- Hz, 2 H), 5.99 (s, 1 H), 4.59-4.36 (m, 3 H), yl)methyl)-2-methyl-1H- 4.24-3.95 (m, 2 H), 3.48 (d, J = 13.2 Hz, 1 indole-3-carboxamide H), 3.17 (d, J = 12.0 Hz, 1 H), 2.87 (br. s., 1 hydrochloride H), 2.70 (br. s., 2 H), 2.58 (s, 3 H), 2.34- 2.25 (m, 3 H), 2.19-2.10 (m, 3 H), 1.75 (d, J = 12.3 Hz, 1 H), 1.57 (d, J = 6.7 Hz, 3 H), 1.47 (d, J = 12.7 Hz, 2 H), 1.33-1.21 (m, 2 H), 0.85 (d, J = 13.6 Hz, 1 H)
Step 10: (R or S)-isopropyl 4-(1-(3-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methylcarbamoy- l)-2-methyl-1H-indol-1-yl)ethyl)piperidine-1-carboxylate (Compound 346)
##STR00033## A 250 mL round bottom flask was charged with a magnetic stir bar, (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide hydrochloride (0.467 g, 0.987 mmol), DMF (2.468 ml, 0.987 mmol), THF (2.468 ml, 0.987 mmol), and N-ethyl-N-isopropylpropan-2-amine (0.638 g, 4.94 mmol). The reaction was cooled to 0.degree. C. and isopropyl carbonochloridate (0.160 ml, 1.086 mmol) was added drop wise via syringe. The reaction was allowed to stir for 2 h to rt and was then treated with 5 N LiOH for 1 h to remove any acylated pyridone. This material was extracted with ethyl acetate, washed with brine, dried with MgSO.sub.4 and filtered and conc. in vacuo. The resulting material was purified via silica gel chromatography (50 g) using ethyl acetate/MeOH (5:1) as eluent to afford pure title compound as a pale yellow solid (0.300 g, 0.545 mmol, 55.2% yield). LCMS 523 (M+1).sup.+; .sup.1H NMR (DMSO-d6, 400 MHz) .delta. 11.59 (br. s., 1H), 7.74 (d, J=7.8 Hz, 1H), 7.69 (t, J=4.9 Hz, 1H), 7.62 (d, J=7.8 Hz, 1H), 7.13-7.01 (m, 2H), 6.15 (s, 1H), 4.78-4.67 (m, 1H), 4.32 (d, J=4.9 Hz, 2H), 4.23-4.12 (m, 1H), 4.12-4.02 (m, 1H), 3.84 (s, 3H), 3.82-3.74 (m, 1H), 2.79-2.66 (m, 1H), 2.58 (s, 3H), 2.46-2.34 (m, 2H), 2.20 (s, 3H), 1.96-1.88 (m, 1H), 1.58-1.46 (m, 4H), 1.15 (d, J=6.0 Hz, 6H), 0.95-0.89 (m, 1H), 0.74-0.65 (m, 1H).
The compounds shown in the following table were prepared according to the general procedure outlined in Step 10 using the appropriate starting materials. The structures of the compounds are shown in FIG. 1.
TABLE-US-00010 Compound Number Name .sup.1H NMR m/z 336 (R or S)-N-((4-methoxy-6-methyl-2- (400 MHz, DMSO-d.sub.6) .delta. = 11.59 515 oxo-1,2-dihydropyridin-3- (s, 1 H), 7.78-7.66 (m, 2 H), yl)methyl)-2-methyl-1-(1-(1- 7.64-7.57 (m, 1 H), 7.06 (s, 2 (methylsulfonyl)piperidin-4- H), 6.14 (s, 1 H), 4.31 (d, J = yl)ethyl)-1H-indole-3-carboxamide 4.9 Hz, 2 H), 4.25-4.15 (m, 1 H), 3.83 (s, 3 H), 3.63 (s, 1 H), 3.40-3.33 (m, 1 H), 2.79 (s, 3 H), 2.75-2.65 (m, 1 H), 2.60 (s, 3 H), 2.45-2.27 (m, 1 H), 2.19 (s, 3 H), 2.06-1.98 (m, 1 H), 1.55 (d, J = 6.9 Hz, 3 H), 1.45- 1.36 (m, 1 H), 1.28-1.18 (m, 1 H), 1.14-1.03 (m, 1 H), 0.83- 0.74 (m, 1 H) 337 (R or S)-1-(1-(1-(2-hydroxy-2- (400 MHz, DMSO-d.sub.6) .delta. 11.58 523 methylpropanoyl)piperidin-4- (br. s., 1 H), 7.77-7.67 (m, 2 yl)ethyl)-N-((4-methoxy-6-methyl-2- H), 7.66-7.60 (m, 1 H), 7.06 (s, oxo-1,2-dihydropyridin-3- 2 H), 6.14 (s, 1 H), 5.32-5.23 yl)methyl)-2-methyl-1H-indole-3- (m, 1 H), 4.31 (d, J = 4.5 Hz, 2 carboxamide H), 4.19-4.10 (m, 1 H), 3.83 (s, 3 H), 2.75-2.62 (m, 2 H), 2.58 (s, 3 H), 2.19 (s, 4 H), 2.00- 1.90 (m, 2 H), 1.54 (d, J = 6.7 Hz, 3 H), 1.32-1.18 (m, 8 H), 0.87-0.78 (m, 1 H), 0.77-0.67 (m, 1 H) 342 (R or S)-1-(1-(1-isobutyrylpiperidin- (400 MHz, DMSO-d.sub.6) .delta. = 11.59 507 4-yl)ethyl)-N-((4-methoxy-6-methyl- (s, 1 H), 7.75 (d, J = 7.4 Hz, 1 2-oxo-1,2-dihydropyridin-3- H), 7.72-7.67 (m, 1 H), 7.64 yl)methyl)-2-methyl-1H-indole-3- (d, J = 8.0 Hz, 1 H), 7.14-7.01 carboxamide (m, 2 H), 6.15 (s, 1 H), 4.58- 4.46 (m, 1 H), 4.32 (d, J = 4.9 Hz, 2 H), 4.09-3.99 (m, 1 H), 3.84 (s, 3 H), 3.81-3.72 (m, 1 H), 3.08-2.97 (m, 1 H), 2.92- 2.81 (m, 1 H), 2.78-2.65 (m, 3 H), 2.59 (br. s., 3 H), 2.20 (s, 3 H), 2.03-1.90 (m, 1 H), 1.59- 1.47 (m, 4 H), 1.02-0.86 (m, 6 H), 0.78-0.69 (m, 1 H) 344 (R or S)-N-((4-(difluoromethoxy)-6- (400 MHz, DMSO-d.sub.6) .delta. 12.02- 551 methyl-2-oxo-1,2-dihydropyridin-3- 11.95 (m, 1 H), 7.74 (d, J = 8.0 yl)methyl)-2-methyl-1-(1-(1- Hz, 1 H), 7.66-7.57 (m, 2 H), (methylsulfonyl)piperidin-4- 7.11-7.00 (m, 2 H), 6.08 (s, 1 yl)ethyl)-1H-indole-3-carboxamide H), 4.32 (d, J = 4.5 Hz, 2 H), 4.18 (d, J = 7.1 Hz, 1 H), 3.64 (d, J = 12.3 Hz, 1 H), 3.36 (d, J = 12.0 Hz, 1 H), 2.79 (s, 3 H), 2.75-2.65 (m, 2 H), 2.58 (s, 3 H), 2.45-2.27 (m, 2 H), 2.20 (s, 3 H), 2.07-1.98 (m, 1 H), 1.55 (d, J = 6.9 Hz, 3 H), 1.40 (d, J = 8.2 Hz, 1 H), 1.10 (d, J = 8.9 Hz, 1 H), 0.79 (d, J = 12.5 Hz, 1 H) 345 (R or S)-1-(1-(1- (400 MHz, DMSO-d.sub.6) .delta. 11.57 529 (ethylsulfonyl)piperidin-4-yl)ethyl)- (s, 1 H), 7.75 (d, J = 8.0 Hz, 1 N-((4-methoxy-6-methyl-2-oxo-1,2- H), 7.69 (t, J = 5.0 Hz, 1 H), dihydropyridin-3-yl)methyl)-2- 7.62 (d, J = 7.4 Hz, 1 H), 7.06 methyl-1H-indole-3-carboxamide (d, J = 7.1 Hz, 2 H), 6.15 (s, 1 H), 4.32 (d, J = 5.1 Hz, 2 H), 4.25-4.15 (m, 1 H), 3.84 (s, 3 H), 3.73-3.65 (m, 1 H), 3.45- 3.36 (m, 1 H), 3.02-2.93 (m, J = 7.8 Hz, 2 H), 2.87-2.77 (m, 1 H), 2.75-2.66 (m, 1 H), 2.60 (s, 3 H), 2.42-2.30 (m, 1 H), 2.20 (s, 3 H), 2.06-1.97 (m, 1 H), 1.58-1.48 (m, 4 H), 1.42-1.31 (m, 1 H), 1.17 (t, J = 7.5 Hz, 3 H), 1.13-1.00 (m, 1 H), 0.83- 0.73 (m, 1 H) 355 (R or S)-1-(1-(4- (400 MHz, DMSO-d.sub.6) .delta. 11.59 543 (isopropylsulfonyl)cyclohexyl)ethyl)- (s, 1H), 7.76-7.69 (m, 2H), 7.62 N-((4-methoxy-6-methyl-2-oxo-1,2- (d, 1H), 7.10-7.03 (m, 2H), 6.15 dihydropyridin-3-yl)methyl)-2- (s, 1H), 4.32 (d, 2H), 4.29-4.26 methyl-1H-indole-3-carboxamide (m, 2H), 3.84 (s, 3H), 3.72 (br. d., 1H), 3.45 (br. d., 1H), 3.26 (tt, 1H), 2.91 (dt, 1H), 2.60 (s, 3H), 2.20 (s, 3H), 1.97 (br. d., 1H), 1.54 (d, 3H), 1.35-1.24 (m, 2H), 1.18 (d, 3H), 1.16 (d, 3H), 1.05-0.78 (m, 2H) 357 (R or S)-isobutyl 4-(1-(3-(((4- (400 MHz, DMSO-d.sub.6) .delta. 537 methoxy-6-methyl-2-oxo-1,2- 11.60 (br. s., 1H), 7.75-7.60 (m, dihydropyridin-3- 3H), 7.10-7.03 (m, 2H), 6.15 (s, yl)methyl)carbamoyl)-2-methyl-1H- 1H) 4.33 (d, 1H), 4.13-4.06 (m, indol-1-yl)ethyl)piperidine-1- 1H), 3.84 (s, 3H), 3.74 (d, 1H), carboxylate 2.80-2.60 (m, 3H), 2.58 (s, 1H), 2.50-2.42 (m, 2H), 1.96-1.90 (m, 1H), 1.54 (d, 3H), 1.25-1.22 (m, 1H), 0.98-0.72 (m, 6H) 368 (R or S)-N-((4,6-dimethyl-2-oxo-1,2- (400 MHz, DMSO-d.sub.6) .delta. 11.59 513 dihydropyridin-3-yl)methyl)-1-(1-(1- (s, 1 H), 7.78-7.71 (m, 1 H), (ethylsulfonyl)piperidin-4-yl)ethyl)- 7.66-7.57 (m, 2 H), 7.07 (s, 2 2-methyl-1H-indole-3-carboxamide H), 5.89 (s, 1 H), 4.32 (s, 2 H), 4.25-4.15 (m, 1 H), 3.65-3.59 (m, 1 H), 3.19-3.10 (m, 1 H), 2.98 (d, J = 7.4 Hz, 2 H), 2.87- 2.77 (m, 1 H), 2.72-2.65 (m, 1 H), 2.58 (s, 3 H), 2.27 (s, 3 H), 2.12 (s, 3 H), 1.55 (d, J = 6.9 Hz, 4 H), 1.42-1.33 (m, 2 H), 1.17 (t, J = 7.4 Hz, 3 H), 1.12- 1.00 (m, 1 H), 0.84-0.74 (m, 1 H) 382 (R or S)-N-((4,6-dimethyl-2-oxo-1,2- (400 MHz, DMSO-d.sub.6) .delta. 11.60 499 dihydropyridin-3-yl)methyl)-2- (s, 1 H), 7.75 (d, J = 7.1 Hz, 1 methyl-1-(1-(1- H), 7.65-7.58 (m, 2 H), 7.12- (methylsulfonyl)piperidin-4- 7.02 (m, 2 H), 5.89 (s, 1 H), yl)ethyl)-1H-indole-3-carboxamide 4.38-4.25 (m, 2 H), 4.20 (dd, J = 7.0, 10.6 Hz, 1 H), 2.80 (s, 3 H), 2.76-2.67 (m, 2 H), 2.59 (s, 3 H), 2.46-2.31 (m, 2 H), 2.27 (s, 3 H), 2.12 (s, 3 H), 1.55 (d, J = 6.9 Hz, 3 H), 1.51 (br. s., 1 H), 1.47-1.34 (m, 1 H), 1.29- 1.21 (m, 1 H), 1.17-1.04 (m, 1 H), 0.80 (d, J = 12.9 Hz, 1 H)
Example 2
Synthesis of (R or S)-1-(1-(1-isopropylpiperidin-4-yl)ethyl)-N-((4-methoxy-6-methyl-2-oxo-1,- 2-dihydropyridin-3-yl)methyl)-2-methyl-1H-indole-3-carboxamide (Compound 358)
##STR00034## A 25 mL vial was charged with a magnetic stir bar, (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide hydrochloride, THF (2.114 ml, 0.211 mmol), propan-2-one (0.061 g, 1.057 mmol), and sodium triacetoxyborohydride (0.224 g, 1.057 mmol). The reaction was allowed to stir at rt for 12 h. The reaction was inverse quenched onto sat'd aqueous NaHCO.sub.3, extracted with ethyl acetate and conc. in vacuo. The resulting material was treated with 10 mL 7 N ammonia in MeOH and was conc in vacuo to yield material which was purified via silica gel chromatography (10 g) using DCM/MeOH/NH.sub.4OH (90:1:0.1) as eluent to afford 33 mg, (0.065 mmol, 31.0% yield) of the title compound as a white solid.). LCMS 479 (M+1).sup.+; .sup.1H NMR (DMSO-d6, 400 MHz) .delta. 11.59 (s., 1H), 7.64-7.82 (m, 2H), 7.59 (d, 1H), 6.95-7.17 (m, 2H), 6.15 (s, 1H), 4.32 (d, 2H), 4.04-4.24 (m, 1H), 3.84 (.quadrature., 3H), 2.77-2.93 (.quadrature., 2H), 2.68 (.quadrature., 1H), 2.60 (.quadrature., 3H), 2.20 (.quadrature., 3H), 2.08-2.15 (.quadrature., 1H), 1.92 (.quadrature., 1H), 1.83 (.quadrature.p. .quadrature.., 1H), 1.54 (.quadrature., 3H), 1.27-1.43 (.quadrature., 2H), 0.91 (.quadrature., 6H), 0.71-0.67 (.quadrature., 2H).
The compounds shown in the following table were prepared according to the general procedure outlined in this Example using the appropriate starting materials. The structures of the compounds are shown in FIG. 1.
TABLE-US-00011 Compound Number Name .sup.1H NMR m/z 341 (R or S)-N-((4-methoxy- (400 MHz, DMSO-d.sub.6) .delta. 11.58 (s, 1 H), 493 6-methyl-2-oxo-1,2- 7.76-7.65 (m, 2 H), 7.59 (d, J = 7.8 Hz, 1 dihydropyridin-3- H), 7.10-6.99 (m, 2 H), 6.14 (s, 1 H), 4.49 yl)methyl)-2-methyl-1- (t, J = 6.4 Hz, 1 H), 4.43 (t, J = 6.5 Hz, 1 (1-(1-(oxetan-3- H), 4.37 (t, J = 6.1 Hz, 1 H), 4.34-4.28 (m, yl)piperidin-4-yl)ethyl)- 3 H), 4.21-4.10 (m, 1 H), 3.83 (s, 3 H), 1H-indole-3-carboxamide 3.30-3.23 (m, 1 H), 2.75 (br. s., 1 H), 2.71- 2.64 (m, 1 H), 2.60 (s, 3 H), 2.19 (s, 4 H), 1.90 (br. s., 1 H), 1.75 (br. s., 1 H), 1.53 (d, J = 6.9 Hz, 3 H), 1.42 (br. s., 2 H), 1.11- 0.98 (m, 1 H), 0.72-0.63 (m, 1 H) 343 (R or S)-N-((4-methoxy- (400 MHz, DMSO-d.sub.6) .delta. 11.58 (s, 1 H), 451 6-methyl-2-oxo-1,2- 7.76-7.65 (m, 2 H), 7.59 (d, J = 7.6 Hz, 1 dihydropyridin-3- H), 7.11-6.99 (m, 2 H), 6.14 (s, 1 H), 4.31 yOmethyl)-2-methyl-1- (d, J = 5.1 Hz, 2 H), 4.13 (br. s., 1 H), 3.83 (1-(1-methylpiperidin-4- (s, 3 H), 2.83 (d, J = 10.0 Hz, 1 H), 2.61- yl)ethyl)-1H-indole-3- 2.52 (m, 5 H), 2.19 (s, 3 H), 2.09 (s, 4 H), carboxamide 1.88 (d, J = 10.7 Hz, 2 H), 1.53 (d, J = 6.7 Hz, 3 H), 1.34 (br. s., 1 H), 1.02 (d, J = 8.2 Hz, 1 H), 0.66 (br. s., 1 H) 359 (R or S)-N-((4-methoxy- (400 MHz, DMSO-d.sub.6) .delta. 11.59 (s, 1 H), 495 6-methyl-2-oxo-1,2- 7.77-7.66 (m, 2 H), 7.60 (d, J = 7.8 Hz, 1 dihydropyridin-3- H), 7.12-7.01 (m, 2 H), 6.15 (s, 1 H), 4.32 yl)methyl)-1-(1-(1-(2- (d, J = 4.9 Hz, 2 H), 4.13 (d, J = 7.1 Hz, 1 methoxyethyl)piperidin- H), 3.85 (s, 3 H), 3.36 (t, J = 5.9 Hz, 2 H), 4-yl)ethyl)-2-methyl-1H- 3.19 (s, 3 H), 2.94 (d, J = 10.5 Hz, 1 H), indole-3-carboxamide 2.71-2.56 (m, 5 H), 2.43-2.32 (m, 2 H), 2.24-2.12 (m, 4 H), 1.54 (d, J = 6.9 Hz, 4 H), 1.39-1.27 (m, 2 H), 1.02 (d, J = 8.7 Hz, 1 H), 0.65 (d, J = 12.7 Hz, 1 H) 360 (R or S)-1-(1-(1- (400 MHz, DMSO-d.sub.6) .delta. 11.79-11.45 (m, 1 465 ethylpiperidin-4- H), 7.78-7.65 (m, 2 H), 7.59 (d, J = 7.8 yl)ethyl)-N-((4- Hz, 1 H), 7.14-6.99 (m, 2 H), 6.15 (s, 1 H), methoxy-6-methyl-2- 4.32 (d, J = 4.9 Hz, 2 H), 4.20-4.08 (m, 1 oxo-1,2-dihydropyridin- H), 3.84 (s, 3 H), 2.98-2.89 (m, 1 H), 2.71- 3-yl)methyl)-2-methyl- 2.61 (m, 2 H), 2.59 (s, 3 H), 2.27-2.21 1H-indole-3- (m, 2 H), 2.20 (s, 3 H), 1.94-1.80 (m, 2 H), carboxamide 1.54 (s, 4 H), 1.38-1.28 (m, 1 H), 1.06- 0.98 (m, 1 H), 0.93 (t, J = 7.1 Hz, 3 H), 0.71-0.63 (m, 1 H) 363 (R or S)-ethyl 2-(4-(1-(3- (400 MHz, DMSO-d.sub.6) .delta. 11.59 (br. s., 1 H), 523 (((4-methoxy-6-methyl- 7.81-7.65 (m, 2 H), 7.60 (d, J = 7.4 Hz, 1 2-oxo-1,2- H), 7.16-6.98 (m, 2 H), 6.15 (s, 1 H), 4.32 dihydropyridin-3- (d, J = 4.9 Hz, 2 H), 4.23-4.11 (m, 1 H), yl)methyl)carbamoyl)-2- 4.04 (q, J = 7.0 Hz, 2 H), 3.84 (s, 3 H), 2.95- methyl-1H-indol-1- 2.86 (m, 1 H), 2.60 (s, 5 H), 2.20 (s, 4 yl)ethyl)piperidin-1- H), 1.94-1.79 (m, 2 H), 1.54 (d, J = 6.9 Hz, 4 yl)acetate H), 1.41-1.32 (m, 1 H), 1.15 (t, J = 7.1 Hz, 3 H), 1.04 (d, J = 6.0 Hz, 2 H), 0.71-0.61 (m, 1 H) 366 (R or S)-N-((4-ethyl-6- (400 MHz, DMSO-d.sub.6) .delta. 11.63 (s, 1 H), 449 methyl-2-oxo-1,2- 7.74 (d, J = 7.6 Hz, 1 H), 7.65-7.56 (m, 2 dihydropyridin-3- H), 7.12-7.01 (m, 2 H), 5.94 (s, 1 H), 4.34 yl)methyl)-2-methyl-1- (t, J = 5.1 Hz, 2 H), 4.19-4.09 (m, 1 H), (1-(1-methylpiperidin-4- 2.88 (br. s., 1 H), 2.71-2.56 (m, 6 H), 2.14 yl)ethyl)-1H-indole-3- (s, 7 H), 1.91 (d, J = 12.5 Hz, 1 H), 1.54 (d, carboxamide J = 6.9 Hz, 4 H), 1.41-1.31 (m, 2 H), 1.14 (t, J = 7.6 Hz, 3 H), 1.05 (d, J = 9.1 Hz, 1 H), 0.68 (d, J = 12.7 Hz, 1 H) 367 (R or S)-N-((4,6- (400 MHz, DMSO-d.sub.6) .delta. 11.59 (s, 1 H), 435 dimethyl-2-oxo-1,2- 7.74 (d, J = 6.9 Hz, 1 H), 7.65-7.56 (m, 2 dihydropyridin-3- H), 7.12-7.01 (m, 2 H), 5.89 (s, 1 H), 4.38- yl)methyl)-2-methyl-1- 4.25 (m, 2 H), 4.20-4.09 (m, 1 H), 2.95 (1-(1-methylpiperidin-4- (br. s., 1 H), 2.68 (br. s., 2 H), 2.58 (s, 3 H), yl)ethyl)-1H-indole-3- 2.27 (s, 3 H), 2.21 (br. s., 3 H), 2.12 (s, 3 carboxamide H), 1.94 (d, J = 13.8 Hz, 1 H), 1.54 (d, J = 6.9 Hz, 4 H), 1.44-1.31 (m, 2 H), 1.07 (d, J = 12.5 Hz, 1 H), 0.71 (d, J = 13.2 Hz, 1 H) 375 (R or S)-N-((4,6- (400 MHz, DMSO-d.sub.6) .delta. 11.59 (br. s., 1 H), 477 dimethyl-2-oxo-1,2- 7.73 (d, J = 7.6 Hz, 1 H), 7.65-7.55 (m, 2 dihydropyridin-3- H), 7.12-7.00 (m, 2 H), 5.89 (s, 1 H), 4.53- yl)methyl)-2-methyl-1- 4.48 (m, 1 H), 4.47-4.42 (m, 1 H), 4.38 (1-(1-(oxetan-3- (s, 1 H), 4.31 (t, J = 5.2 Hz, 3 H), 4.21- yl)piperidin-4-yl)ethyl)- 4.10 (m, 1 H), 3.31-3.24 (m, 2 H), 2.81- 1H-indole-3- 2.64 (m, 2 H), 2.59 (s, 3 H), 2.26 (s, 3 H), carboxamide 2.23-2.16 (m, 1 H), 2.12 (s, 3 H), 1.98- 1.85 (m, 1 H), 1.81-1.70 (m, 1 H), 1.54 (d, J = 6.9 Hz, 3 H), 1.51-1.22 (m, 1 H), 1.12- 0.96 (m, 2 H), 0.73-0.64 (m, 1 H) 380 (R or S)-N-((4-ethyl-6- (400 MHz, DMSO-d.sub.6) .delta. 11.63 (br. s., 1 H), 491 methyl-2-oxo-1,2- 7.74 (d, J = 7.36 Hz, 1 H), 7.60 (d, J = 8.47 dihydropyridin-3- Hz, 2 H), 7.06 (quin, J = 7.13 Hz, 3 H), yl)methyl)-2-methyl-1- 5.94 (s, 1 H), 4.51 (t, J = 6.47 Hz, 1 H), (1-(1-(oxetan-3- 4.46 (t, J = 6.35 Hz, 1 H), 4.40 (t, J = 6.13 yl)piperidin-4-yl)ethyl)- Hz, 1 H), 4.37-4.30 (m, 2 H), 4.28-4.11 1H-indole-3- (m, 1 H), 3.57 (s, 1 H), 3.34 (br. s., 2 H), carboxamide 2.81 (d, J = 10.70 Hz, 1 H), 2.67 (d, J = 14.94 Hz, 1 H), 2.64-2.57 (m, 4 H), 2.21 (d, J =10.93 Hz, 1 H), 2.14 (s, 3 H), 1.93 (d, J =12.49 Hz, 1 H), 1.83 (t, J = 11.37 Hz, 1 H), 1.54 (d, J = 6.91 Hz, 3 H), 1.37 (d, J = 10.48 Hz, 1 H), 1.25 (q, J = 6.91 Hz, 1 H), 1.14 (t, J = 7.58 Hz, 3 H), 1.06 (d, J = 9.81 Hz, 1 H), 0.70 (d, J = 12.49 Hz, 1 H) 381 (R or S)-N-((4,6- .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta. 11.60 505 dimethyl-2-oxo-1,2- (br. s., 1 H), 7.74 (d, J = 7.13 Hz, 1 H), 7.66- dihydropyridin-3- 7.50 (m, 2 H), 7.15-6.99 (m, 2 H), 5.89 yl)methyl)-2-methyl-1- (s, 1 H), 4.40-4.24 (m, 2 H), 4.21-4.07 (1-(1-(tetrahydro-2H- (m, 1 H), 3.95-3.78 (m, 2 H), 3.57 (s, 1 H), pyran-4-yl)piperidin-4- 3.32-3.17 (m, 3 H), 2.68 (br. s., 1 H), 2.58 yl)ethyl)-1H-indole-3- (s, 3 H), 2.33 (br. s., 2 H), 2.27 (s, 3 H), carboxamide 2.12 (s, 3 H), 2.00-1.88 (m, 2 H), 1.75 (d, J = 12.04 Hz, 1 H), 1.62 (br. s., 2 H), 1.54 (d, J = 6.91 Hz, 3 H), 1.46-1.30 (m, 2 H), 1.01 (br. s., 1 H), 0.72 (br. s., 1 H) 440 (R or S)-tert-butyl 3-(4- 576 (1-(3-(((4,6-dimethyl-2- oxo-1,2-dihydropyridin- 3-yl)methyl)carbamoyl)- 2-methyl-1H-indol-1- yl)ethyl)piperidin-1- yl)azetidine-1- carboxylate 377 (R or S)-N-((4,6- (400 MHz, DMSO-d.sub.6) .delta.11.63-11.56 (m, 1 490 dimethyl-2-oxo-1,2- H), 7.76-7.70 (m, 1 H ), 7.64-7.55 (m, 2 dihydropyridin-3- H), 7.05 (s, 2 H), 5.89 (s, 1 H), 4.56 (s, 4 yl)methyl)-2-methyl-1- H), 4.31 (s, 2 H), 4.19-4.09 (m, 1 H), 3.36 (1-(1-(1-methylazetidin- (d, J = 4.9 Hz, 1 H), 2.77-2.56 (m, 5 H), 3-yl)piperidin-4- 2.26 (s, 3 H), 2.18 (s, 3 H), 2.12 (s, 3 H), yl)ethyl)-1H-indole-3- 1.94-1.85 (m, 1 H), 1.78-1.67 (m, 1 H), carboxamide 1.53 (d, J = 6.9 Hz, 3 H), 1.50-1.45 (m, 1 H), 1.44-1.22 (m, 2 H), 1.07-0.93 (m, 1 H), 0.71-0.61 (m, 1 H)
Example 3
Synthesis of (R or S)-1-(1-(1-(2-fluoroethyl)piperidin-4-yl)ethyl)-N-((4-methoxy-6-methyl-2-- oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl-1H-indole-3-carboxamide (Compound 356)
##STR00035## A 25 mL vial was charged with a magnetic stir bar, (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide hydrochloride (0.062 g, 0.131 mmol), K.sub.2CO.sub.3 (0.072 g, 0.524 mmol), MeCN (0.655 ml, 0.131 mmol), DMF (0.262 ml, 0.131 mmol) and 1-bromo-2-fluoroethane (0.020 ml, 0.262 mmol). The reaction was capped and heated to 82.degree. C. with stirring for 4 h. The reaction was allowed to cool to rt, filtered, and the filtrate was pre-absorbed onto silica gel (12 g). The material was purified via SiO.sub.2 chromatography (25 g) using DCM/MeOH/Et.sub.3N (85:15:0.5) as eluent to afford the title compound as an off white solid (30 mg, 0.059 mmol, 45.1% yield). LCMS 483 (M+1).sup.+; .sup.1H NMR (DMSO-d6, 400 MHz) .delta. 11.59 (s, 1H), 7.75-7.68 (m, 2H), 7.60 (d, 1H) 7.09-7.03 (m, 2H), 6.15 (s, 1H) 4.53-4.51 (m, 1H), 4.42-4.39 (m, 1H), 4.32 (d, 2H), 4.24-4.2 (m, 1H), 3.84 (s, 3H), 2.98 (br. d., 1H), 2.70-2.49 (m, 4H), 2.60 (s, 3H), 2.20 (s, 3H), 2.01 (dt, 1H), 1.92-1.90 (m, 1H), 1.75-1.71 (m, 1H), 1.54 (d, 3H), 1.38-1.36 (m, 1H), 1.02-0.98 (m, 1H), 0.7-0.66 (br. d., 1H).
The compounds shown in the following table were prepared according to the general procedure outlined in this Example using the appropriate starting materials. The structures of the compounds are shown in FIG. 1.
TABLE-US-00012 Compound Number Name .sup.1H NMR m/z 362 (R or S)-1-(1-(1-(2,2- (400 MHz, DMSO-d.sub.6) .delta. = 11.60 (br. s., 1 501 difluoroethyl)piperidin- H), 7.77-7.66 (m, 2 H), 7.60 (d, J = 7.6 Hz, 4-yl)ethyl)-N-((4- 1 H), 7.14-7.00 (m, 2H ), 6.15 (s, 1 H), methoxy-6-methyl-2- 6.06 (t, J = 55.7 Hz, 1 H), 4.32 (d, J = 4.9 oxo-1,2-dihydropyridin- Hz, 2 H), 4.15 (br. s., 1 H), 3.84 (s, 3 H), 3-yl)methyl)-2-methyl- 3.03-2.93 (m, 2 H), 2.73-2.62 (m, 3 H), 1H-indole-3- 2.60 (s, 3 H), 2.26-2.10 (m, 4 H), 1.93- carboxamide 1.79 (m, 1 H), 1.59-1.46 (m, 4 H), 1.41- 1.29 (m, 1 H), 1.11-0.97 (m, 1 H), 0.67 (br. s., 1 H) 378 (R or S)-N-((4-methoxy- (400 MHz, DMSO-d.sub.6) .delta. = 11.60 (br. s., 1 H), 533 6-methyl-2-oxo-1,2 7.78-7.66 (m, 2 H), 7.60 (d, J = 8.2 Hz, 1 dihydropyridin-3- H), 7.13-7.00 (m, 2 H), 6.15 (s, 1 H), 4.32 yl)methyl)-2-methyl-1- (d, J = 4.9 Hz, 2 H), 4.22-4.09 (m, 1 H), (1-(1-(3,3,3- 3.84 (s, 3 H), 3.03-2.91 (m, 1 H), 2.73- trifluoropropyl)piperidin- 2.64 (m, 1 H), 2.60 (s, 3 H), 2.48-2.31 (m, 4-yl)ethyl)-1H-indole-3- 5 H), 2.20 (s, 3 H), 2.01-1.85 (m, 2 H), carboxamide 1.58-1.46 (m, 4 H), 1.36-1.29 (m, 1 H), 1.08-0.98 (m, 1 H), 0.73-0.62 (m, 1 H) 365 (R or S)-N-((4-methoxy- (500 MHz, DMSO-d.sub.6) .delta. = 11.59 (s, 1 H), 519 6-methyl-2-oxo-1,2- 7.74 (d, J = 7.6 Hz, 1 H), 7.71-7.66 (m, 1 dihydropyridin-3- H), 7.61 (d, J = 7.8 Hz, 1 H), 7.13-7.01 (m, yl)methyl)-2-methyl-1- 2 H), 6.15 (s, 1 H), 4.32 (d, J = 4.9 Hz, 2 H), (1-(1-(2,2,2- 4.22-4.12 (m, 1 H), 3.84 (s, 3 H), 3.15- trifluoroethyl)piperidin- 2.95 (m, 3 H), 2.75-2.66 (m, 1 H), 2.60 (s, 4-yl)ethyl)-1H-indole-3- 3 H), 2.39-2.31 (m, 1 H), 2.20 (s, 3 H), carboxamide 2.05-1.98 (m, 1 H), 1.92-1.84 (m, 1 H), 1.56-1.46 (m, 4 H), 1.42-1.32 (m, 1 H), 1.11-1.01 (m, 1 H), 0.69-0.62 (m, 1 H) 441 (R or S)-1-(1-(1-(2,2- (400 MHz, DMSO-d.sub.6) .delta. = 11.59 (s, 1 H), 485 difluoroethyl)piperidin- 7.73 (d, J = 7.8 Hz, 1 H), 7.65-7.55 (m, 2 4-yl)ethyl)-N-((4,6- H), 7.12-7.00 (m, 2 H), 6.22-5.90 (m, 1 dimethyl-2-oxo-1,2- H), 5.89 (s, 1 H), 4.36-4.25 (m, 2 H), 4.20- dihydropyridin-3- 4.09 (m, 1 H), 3.01-2.93 (m, 1 H), 2.72- yl)methyl)-2-methyl-1H- 2.59 (m, 3 H), 2.58 (s, 3 H), 2.26 (s, 3 H), indole-3-carboxamide 2.21-2.13 (m, 2 H), 2.12 (s, 3 H), 1.92- 1.79 (m, 2 H), 1.53 (s, 4 H), 1.41-1.29 (m, 1 H), 1.10-0.97 (m, 1 H), 0.70-0.59 (m, 1 H)
Example 4
Synthesis of (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(1-(pyrimidin-2-yl)piperidin-4-yl)ethyl)-1H-indole-3-carboxamide (Compound 361)
##STR00036## To a re-sealable vial was added 2-chloropyrimidine (185 mg, 1.611 mmol), (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide hydrochloride (508 mg, 1.074 mmol), and EtOH (8 mL). To this solution was added Et.sub.3N (449 .mu.l, 3.22 mmol). The vial was sealed and heated to 100.degree. C. overnight. The solution was allowed to cool to room temperature and concentrated in vacuo. The crude residue was purified via silica gel chromatography (hexanes: (3:2 DCM:IPA)) to afford the title compound as a solid (357 mg, 0.694 mmol, 64.6% yield). LCMS 515 (M+1).sup.+; .sup.1H NMR (DMSO-d6, 400 MHz) .delta. 11.60 (s, 1H), 8.30 (d, J=4.7 Hz, 2H), 7.76 (d, J=7.6 Hz, 1H), 7.73-7.64 (m, 2H), 7.14-7.01 (m, 2H), 6.55 (t, J=4.7 Hz, 1H), 6.15 (s, 1H), 4.84-4.75 (m, 1H), 4.57-4.47 (m, 1H), 4.33 (d, J=4.2 Hz, 2H), 4.22-4.11 (m, 1H), 3.84 (s, 3H), 2.92-2.81 (m, 1H), 2.63-2.52 (m, 4H), 2.20 (s, 3H), 2.05-1.94 (m, 1H), 1.61-1.49 (m, 4H), 1.34-1.21 (m, 1H), 1.04-0.91 (m, 1H), 0.83-0.75 (m, 1H).
The compound shown in the following table was prepared according to the general procedure outlined in this Example using the appropriate starting materials. The structure of the compound is shown in FIG. 1.
TABLE-US-00013 Compound Number Name .sup.1H NMR m/z 373 (R or S)-N-((4- (400 MHz, DMSO-d.sub.6) .delta. 11.60 (br. s., 1 H), 8.06 514 methoxy-6-methyl- (d, J =3.6 Hz, 1 H), 7.82-7.62 (m, 3 H), 7.51- 2-oxo-1,2- 7.39 (m, 1 H), 7.17-6.98 (m, 2 H), 6.75 (d, J = dihydropyridin-3- 8.5 Hz, 1 H), 6.61-6.49 (m, 1 H), 6.15 (s, 1 H), yl)methyl)-2- 4.49-4.38 (m, 1 H), 4.33 (d, J = 3.8 Hz, 2 H), methyl-1-(1-(1- 4.24-4.03 (m, 2 H), 3.85 (s, 3 H), 2.90-2.70 (pyridin-2- (m, 2 H), 2.58 (s, 3 H), 2.20 (s, 3 H), 2.06-1.91 yl)piperidin-4- (m, 1 H), 1.63-1.47 (m, 4 H), 1.40-1.27 (m, 1 yl)ethyl)-1H- H), 1.07-0.94 (m, 1 H), 0.82-0.72 (m, 1 H) indole-3- carboxamide
Example 5
Synthesis of (R or S)-1-(1-(1-(2-hydroxyethyl)piperidin-4-yl)ethyl)-N-((4-methoxy-6-methyl-2- -oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl-1H-indole-3-carboxamide (Compound 347)
##STR00037## To a sealed tube charged with a magnetic stir bar was added (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -1-(1-(piperidin-4-yl)ethyl)-1H-indole-3-carboxamide (0.1 g, 0.229 mmol) was added DCM (3 mL) and the reaction cooled to 0.degree. C. To the cooled reaction mixture was added oxirane which was condensed into the reaction vial (.about.1 mL). The reaction was allowed to stir to rt over 4 h and was then conc. in vacuo to afford the crude material which was purified via silica gel chromatography (12 g) using ethyl acetate/MeOH (4:1) as eluent to afford the title compound as a white solid (50 mg). LCMS 481 (M+1).sup.+; .sup.1H NMR (DMSO-d6, 400 MHz) .delta.) .delta. 11.58 (s, 1H), 7.77-7.65 (m, 2H), 7.59 (d, J=7.8 Hz, 1H), 7.11-6.99 (m, 2H), 6.14 (s, 1H), 4.54-4.44 (m, 1H), 4.31 (d, J=5.1 Hz, 3H), 4.13 (dd, J=7.1, 10.3 Hz, 1H), 3.83 (s, 3H), 3.42 (q, J=6.0 Hz, 2H), 2.93 (br. s., 1H), 2.71-2.56 (m, 4H), 2.31 (br. s., 2H), 2.19 (s, 3H), 2.03-1.83 (m, 2H), 1.64 (br. s., 1H), 1.53 (d, J=6.9 Hz, 3H), 1.32 (d, J=11.1 Hz, 1H), 1.02 (d, J=10.3 Hz, 1H), 0.65 (d, J=11.8 Hz, 1H).
The compounds shown in the following table were prepared according to the general procedure outlined in this Example using the appropriate starting materials. The structures of the compounds are shown in FIG. 1.
TABLE-US-00014 Compound Number Name .sup.1H NMR m/z 352 (R or S)-1-(1-(1-(2- NMR (400 MHz, DMSO-d6) .delta. 11.58 (br. s., 1 509 hydroxy-2- H), 7.76-7.65 (m, 2 H), 7.58 (d, J = 7.8 Hz, 1 methylpropyl)piperidin- H), 7.10-6.99 (m, 2 H), 6.14 (s, 1 H), 4.31 (d, 4-yl)ethyl)-N-((4- J = 4.9 Hz, 2 H), 4.14 (br. s., 1 H), 3.94 (s, 1 methoxy-6-methyl-2- H), 3.83 (s, 3 H), 3.56 (s, 2 H), 3.01 (d, J = oxo-1,2- 11.4 Hz, 1 H), 2.73-2.64 (m, 1 H), 2.59 (s, 3 dihydropyridin-3- H), 2.19 (s, 3 H), 2.16-2.03 (m, 2 H), 1.52 (d, yl)methyl)-2-methyl- J = 6.9 Hz, 4 H), 1.34 (br. s., 2 H), 1.02 (d, J = 1H-indole-3- 4.5 Hz, 7 H), 0.66-0.58 (m, 1 H) carboxamide 369 (R or S)-N-((4,6- (400 MHz, DMSO-d6) .delta. 11.59 (br. s., 1 H), 493 dimethyl-2-oxo-1,2- 7.77-7.69 (m, 1 H), 7.60 (br. s., 2 H), 7.06 dihydropyridin-3- (br. s., 2 H), 5.89 (s, 1 H), 4.31 (t, J = 5.7 Hz, 2 yl)methyl)-1-(1-(1-(2- H), 4.20-4.09 (m, 1 H), 4.00-3.92 (m, 1 H), hydroxy-2- 3.58-3.55 (m, 2 H), 3.19-3.10 (m, 1 H), 3.07- methylpropyl)piperidin- 2.95 (m, 1 H), 2.74-2.63 (m, 1 H), 2.59 (br. 4-yl)ethyl)-2-methyl- s., 3 H), 2.27 (s, 3 H), 2.12 (s, 3 H), 1.89-1.72 1H-indole-3- (m, 1 H), 1.54 (br. s., 4 H), 1.30-1.14 (m, 2 carboxamide H), 1.03 (br. s., 6 H), 0.84-0.57 (m, 2 H)
Example 6
Synthesis of (R or S)--N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl- -6-phenyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole-3-carboxamide (Compound 374)
##STR00038## A 25 mL reaction tube was charged with a magnetic stir bar, phenyl boronic acid (72.6 mg, 0.596 mmol), K.sub.3PO.sub.4 (103 mg, 0.447 mmol), X-Phos pre-catalyst (Chloro(2-dicyclohexylphosphino-2',4',6'-triisopropyl-1,1'-biphenyl)[2-(2- -aminoethyl)phenyl)]palladium(II)) (4.92 mg, 5.96 .mu.mol), and the vial was sealed. The vial was evacuated/backfilled with nitrogen (3.times.) before the addition of methyl 6-chloro-2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole-3-carbo- xylate (Compound 314) (100 mg, 0.298 mmol) as a solution in 1,4-dioxane (1 mL). The vial was then heated to 100.degree. C. overnight with stirring. The vial was then allowed to cool to room temperature and the reaction concentrated in vacuo. The crude residue was purified via SiO.sub.2 chromatography (10 g) using an eluent of ethyl acetate/hexanes (4:1) the title compound as a white solid (106 mg, 0.281 mmol, 94% yield).). LCMS 514 (M+1).sup.+; .sup.1H NMR .sup.1H NMR (400 MHz, DMSO-d.sub.6) .delta.=11.59 (s, 1H), 7.98-7.84 (m, 2H), 7.75-7.67 (m, 3H), 7.47 (t, J=7.8 Hz, 2H), 7.39 (d, J=8.5 Hz, 1H), 7.35-7.27 (m, 1H), 6.15 (s, 1H), 4.35 (d, J=4.9 Hz, 2H), 4.25-4.12 (m, 1H), 3.93 (d, J=8.5 Hz, 1H), 3.86-3.77 (m, 3H), 3.67 (d, J=8.5 Hz, 1H), 3.39-3.32 (m, 1H), 3.10-3.00 (m, 1H), 2.62 (s, 3H), 2.21 (s, 3H), 1.85 (d, J=10.0 Hz, 1H), 1.63-1.49 (m, 4H), 1.45-1.33 (m, 1H), 1.20-0.99 (m, 1H), 0.66 (d, J=12.0 Hz, 1H).
Example 7
Synthesis of (R or S)-2-(4-(1-(3-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methylca- rbamoyl)-2-methyl-1H-indol-1-yl)ethyl)piperidin-1-yl) acetic acid (Compound 364)
##STR00039## To a round bottomed flask was charged with a magnetic stir bar was added (R or S)-ethyl-2-(4-(1-(3-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)me- thylcarbamoyl)-2-methyl-1H-indol-1-yl)ethyl)piperidin-1-yl)acetate (Compound 363) (69 mg, 0.132 mmol), THF (1.5 mL), MeOH (1.5 mL), and water (0.75 mL). To this solution was added lithium hydroxide monohydrate (5.54 mg, 0.132 mmol) and the reaction stirred at room temperature for 1 h. The organics were removed under reduced pressure and the resulting aqueous solution purified via reverse phase-HPLC (water/MeCN) 0.fwdarw.95% to afford the title compound (66 mg, 0.108 mmol, 82% yield). LCMS 514 (M+1).sup.+ 1H NMR (400 MHz, DMSO-d.sub.6) .delta.=11.67 (s, 1H), 9.65 (s, 1H), 7.84-7.68 (m, 2H), 7.63 (d, J=7.4 Hz, 1H), 7.14-7.03 (m, 2H), 6.18 (s, 1H), 4.33 (d, J=3.6 Hz, 2H), 4.27-4.15 (m, 1H), 4.04 (br. s., 2H), 3.85 (s, 3H), 3.57 (s, 1H), 3.35-3.23 (m, 1H), 3.14-2.99 (m, 1H), 2.86-2.74 (m, 1H), 2.62 (s, 3H), 2.21 (s, 3H), 2.18-2.08 (m, 1H), 1.75 (s, 1H), 1.60-1.49 (m, 4H), 1.46-1.33 (m, 1H), 0.92-0.81 (m, 1H).
Example 8
Synthesis of (R or S)-methyl 2-methyl-6-(pyridin-3-yl)-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole- -3-carboxylate
This intermediate was used as an alternate starting material in Step 7 set forth in Example 1 for the synthesis of other compounds of the invention.
(R or S)-methyl 2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-6-(4,4,5,5-tetramethyl-1,3- ,2-dioxaborolan-2-yl)-1H-indole-3-carboxylate
##STR00040## To a round bottomed flask was added Pd(OAc).sub.2 (10.03 mg, 0.045 mmol), potassium acetate (219 mg, 2.233 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (567 mg, 2.233 mmol), and 2-dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl (XPhos) (85 mg, 0.179 mmol), and the vial was sealed. To this vessel was added (R or S)-methyl 6-chloro-2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole-3-carbo- xylate (Step 6) (500 mg, 1.489 mmol) dissolved in dioxane (3.4 mL) and the reaction evacuated/back-filled with N.sub.2 (3.times.) before heating to 100.degree. C. overnight. The reaction was then allowed to cool to rt and was diluted with EtOAc. The reaction was filtered through diatomaceous earth and the filtrate concentrated to afford the title compound which was used in subsequent reactions without further purification. LCMS 428 (M+1).sup.+.
(R or S)-methyl 2-methyl-6-(pyridin-3-yl)-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-1H-indole- -3-carboxylate
##STR00041## To a re-sealable vial was added K.sub.2CO.sub.3 (206 mg, 1.488 mmol), PdCl.sub.2(dppf)-CH.sub.2Cl.sub.2 adduct (60.8 mg, 0.074 mmol), and the vial was sealed. This vial was evacuated/backfilled with N.sub.2 (3.times.) before addition of (R or S)-methyl 2-methyl-1-(1-(tetrahydro-2H-pyran-4-yl)ethyl)-6-(4,4,5,5-tetramethyl-1,3- ,2-dioxaborolan-2-yl)-1H-indole-3-carboxylate (318 mg, 0.744 mmol) dissolved in 1,4-dioxane (4 mL), 3-bromopyridine (71.7 .mu.l, 0.744 mmol), and water (400 .mu.L). The reaction was evacuated/backfilled with N.sub.2 (3.times.) before heating to 100.degree. C. The solution was cooled to room temperature and diluted with EtOAc. The solution was filtered and concentrated in vacuo. The crude residue was purified via silica gel chromatography (10 g, EtOAc/hex (1:1)) to afford the title compound (101 mg, 0.267 mmol, 35.9% yield). LCMS 379 (M+1).sup.+.
Example 9
Other Alkyl Carboxylate Intermediates
The following alkyl carboxylate intermediates were synthesized in an analogous manner to that set forth in Step 2 of Example 1, using an appropriate starting material and reactant.
TABLE-US-00015 Name Structure m/z (.+-.)-ethyl 5- fluoro-1-(1- methoxypropan- 2-yl)-2-methyl- 1H-indole-3- carboxylate ##STR00042## 294 (.+-.)-ethyl 6- fluoro-1-(1- methoxypropan- 2-yl)-2-methyl- 1H-indole-3- carboxylate ##STR00043## 294 (.+-.)-ethyl 1-(1- methoxypropan- 2-yl)-2-methyl- 1H-indole-3- carboxylate ##STR00044## 276 (.+-.)-tert-butyl 1-(1- methoxypropan- 2-yl)-2-methyl- 1H-pyrrolo[2,3- b]pyridine-3- carboxylate ##STR00045## 305 (.+-.)-tert-butyl 1- (1-ethoxypropan- 2-yl)-2-methyl- 1H-pyrrolo[2,3- b]pyridine-3- carboxylate ##STR00046## 319 tert-butyl 1-(3- methoxybutan-2- yl)-2-methyl- 1H-pyrrolo[2,3- b]pyridine-3- carboxylate ##STR00047## 319 ethyl 1-(3- methoxybutan- 2-yl)-2- methyl-6- (methylsulfonyl)- 1H-indole-3- carboxylate ##STR00048## 368 (.+-.)-ethyl 1-(3- methoxypentan- 2-yl)-2-methyl- 1H-indole-3- carboxylate ##STR00049## 304
Example 10
Other Compounds of the Invention Produced from Carboxylic Acid Intermediates
The following compounds were synthesized in an analogous manner to that set forth in Step 4 of Example 1, using an appropriate starting material. Structures of these compounds are set forth in FIG. 1.
TABLE-US-00016 Compound Name .sup.1H NMR m/z 304 (.+-.)-1-(1-(4,4- (CDCl.sub.3 400 MHz) .delta. 12.63-12.64 (d, J = 3.2 427 difluorocyclohexyl)ethyl)- Hz, 1H), 7.84 (s, 1H), 7.49 (s, 1H), 7.42-7.40 N-((4-methoxy-6-methyl- (d, J = 9.2 Hz, 1H), 7.06-7.00 (m, 2H), 5.90- 2-oxo-1,2-dihydropyridin- 5.89 (d, J = 3.6 Hz 1H), 4.66-4.62 (t, J = 14 3-yl)methyl)-2-methyl- Hz, 2H), 4.11-4.08 (m, 1H), 3.88-3.87 (d, 1H-indole-3-carboxamide J = 3.6 Hz, 3H), 2.99-2.76 (m, 3H), 2.36 (s, 1H), 2.25 (s, 3H), 2.17-2.16 (d, J = 3.2 Hz, 2H), 2.08-2.05 (m, 2H), 1.84-1.70 (m, 2H), 1.61 (s, 1H), 1.51-1.47 (m, 2H) 230 (.+-.)-5-fluoro-N-((4- (400 MHz, CD.sub.3OD) .delta. 7.59-7.55 (m, 1H), 416 methoxy-6-methyl-2-oxo- 7.42-7.39 (m, 1H), 6.95-6.90 (m, 2H), 4.57 1,2-dihydropyridin-3- (s, 2H), 4.12 (s, 3H), 3.99-3.94 (m, 1H), yl)methyl)-1-(1- 3.72-3.65 (m, 1H), 3.19 (s, 3H), 2.64 (s, 3H), methoxypropan-2-yl)-2- 2.54 (s, 3H), 1.59-1.57 (d, 3H) methyl-1H-indole-3- carboxamide 231 (.+-.)-6-fluoro-N-((4- (400 MHz, CD.sub.3OD) .delta. 7.70-7.66 (m, 1H), 416 methoxy-6-methyl-2-oxo- 7.36-7.33 (m, 1H), 6.94-6.89 (m, 2H), 4.56 1,2-dihydropyridin-3- (s, 2H), 4.11 (s, 3H), 3.97-3.92 (m, 1H), yl)methyl)-1-(1- 3.71-3.67 (m, 1H), 3.20 (s, 3H), 2.62 (s, 3H), methoxypropan-2-yl)-2- 2.53 (s, 3H), 1.58-1.56 (d, 3H) methyl-1H-indole-3- carboxamide 218 (.+-.)-N-((4-methoxy-6- (400 MHz, CD.sub.3OD) .delta. 7.69 (d, J = 7.2 398 methyl-2-oxo-1,2- Hz, 1H), 7.53 (d, J = 7.6 Hz, 1H), 7.12 (m, dihydropyridin-3- 2H), 6.26 (s, 1H), 4.80 (m, 1H), 4.52 (s, 2H), yl)methyl)-1-(1- 3.99 (m, 4H), 3.75 (m, 1H), 3.20 (s, 3H), 2.62 methoxypropan-2-yl)-2- (s, 3H), 2.31 (s, 3H), 1.59 (d, J = 7.2 Hz, 3H) methyl-1H-indole-3- carboxamide 183 (.+-.)-N-((4,6-dimethyl-2- (400 MHz, CD.sub.3OD) .delta. 7.74 (m, 1H), 7.57 382 oxo-1,2-dihydropyridin-3- (d, J = 7.6 Hz, 1H), 7.15 (m, 2H), 6.14 yl)methyl)-1-(1- (s, 1H), 4.86 (m, 1H), 4.55 (s, 2H), 4.02 (m, methoxypropan-2-yl)-2- 1H), 3.77 (m, 1H), 3.22 (s, 3H), 2.65 (s, 3H), methyl-1H-indole-3- 2.43 (s, 3H), 2.26 (s, 3H), 1.62 (d, J = 7.2 Hz, carboxamide 3H) 204 (.+-.)-N-((4-methoxy-6- (400 MHz, CDCl.sub.3) .delta. 13.23 (s, 1H), 8.16- 399 methyl-2-oxo-1,2- 8.17 (m, 1H), 8.11-8.13 (m, 1H), 7.57-7.60 dihydropyridin-3- (t, J = 5.2 Hz, 1H), 6.93-6.96 (m, 1H), 5.92 yl)methyl)-1-(1- (s, 1H), 4.82-4.83 (d, J = 2.4 Hz, 1H), 4.65- methoxypropan-2-yl)-2- 4.66 (d, J = 6.4 Hz, 2H), 3.89 (s, 3H), 3.81- methyl-1H-pyrrolo[2,3- 3.85 (m, 1H), 3.22 (s, 3H), 2.79 (s, 3H), b]pyridine-3-carboxamide 2.17 (s, 3H), 1.64-1.66 (d, J = 8.0 Hz, 3H) 211 (.+-.)-1-(1- 379 cyclopropylethyl)-N-((4,6- dimethyl-2-oxo-1,2- dihydropyridin-3- yl)methyl)-2-methyl-1H- pyrrolo[2,3-b]pyridine-3- carboxamide 212 (.+-.)-1-(1-ethoxypropan-2- (400 MHz, CDCl.sub.3) .delta. 8.173-8.189 (m, 1H), 413 yl)-N-((4-methoxy-6- 8.13-8.153 (m, 1H), 7.563 (s, 1H), 6.977- methyl-2-oxo-1,2- 7.008 (m, 1H), 5.938 (s, 1H), 4.652-4.667 dihydropyridin-3- (d, 2H), 4.177 (s, 1H), 3.309-3.454 (m, 2H), yl)methyl)-2-methyl-1H- 3.94-3.98 (m, 1H), 2.806 (s, 3H), 2.212 (s, pyrrolo[2,3-b]pyridine-3- 3H), 1.665-1.682 (d, 3H), 1.044 (t, 3H) carboxamide 235 (.+-.)-N-((4-ethoxy-6- (400 MHz, CDCl.sub.3) .delta. 413 methyl-2-oxo-1,2- 12.5 (s, 1H), 8.11-8.18 (m, 2H), 7.60 (s, dihydropyridin-3- 1H), 6.95-6.98 (m, 1H), 5.90 (s, 1H), 5.96 yl)methyl)-1-(1- (s, 1H), 4.83 (s, 1H), 4.10-4.21 (m, 3H), methoxypropan-2-yl)-2- 3.82-3.83 (m, 1H), 3.23 (s, 3H), 3.79 (s, methyl-1H-pyrrolo[2,3- 3H), 2.15 (s, 3H), 1.65-1.66 (d, J = 6.8 Hz, b]pyridine-3-carboxamide 6H), 1.44-1.47 (t, J = 7.2 Hz, 3H). 241 N-((4,6-dimethyl-2-oxo- (400 MHz, CD.sub.3OD): .delta. 8.67-8.65 (d, 1H), 397 1,2-dihydropyridin-3- 8.45-8.44 (d, 1H), 7.59-7.55 (m, 1H), 6.70 yl)methyl)-1-(3- (s, 1H), 4.79 (s, 1H), 4.61 (s, 2H), 4.07 (s, methoxybutan-2-yl)-2- 1H), 3.32 (s, 3H), 2.75 (s, 3H), 2.56 (s, 3H), methyl-1H-pyrrolo[2,3- 2.42 (s, 3H), 1.68-1.66 (d, 3H), 1.16-1.15 (d, b]pyridine-3-carboxamide 3H). 280 (.+-.)-N-((6-ethyl-4- 1H NMR (400 MHz, CD.sub.3OD) .delta. 8.25-8.29 413 methoxy-2-oxo-1,2- (m, 2H). .delta. 7.28-7.31 (m, 1H). 6.89 (s, 1H), dihydropyridin-3- 4.93-4.95 (br, 1H), 4.58 (s, 2H), 4.2-4.25 yl)methyl)-1-(1- (m, 1H), 4.13 (s, 3H), 3.77-3.81 (m, 1H), methoxypropan-2-yl)-2- 3.24 (s, 3H), 2.79-2.84 (q, 1H), 2.72 (s, 3H), methyl-1H-pyrrolo[2,3- 1.66-1.68 (d, J = 7.2 Hz, 3H) 1.32-1.36 (t, b]pyridine-3-carboxamide 3H) 288 (R or S)-N-((4-Methoxy- (400 MHz, d6-DMSO) .delta. 11.57-11.65 (m, 439 6-methyl-2-oxo-1,2- 1H), 8.18-8.23 (m, 1H), 8.07-8.12 (m, dihydropyridin-3- 1H), 7.83-7.91 (m, 1H), 7.07-7.15 (m, yl)methyl)-2-methyl-1-(1- 1H), 6.15 (s, 1H), 4.31 (d, J = 4.46 Hz, 1H), (tetrahydro-2H-pyran-4- 4.04-4.20 (m, 1H), 3.88-3.97 (m, 1H), yl)ethyl)-1H-pyrrolo[2,3- 3.84 (s, 3H), 3.59-3.70 (m, 1H), 2.97- b]pyridine-3-carboxamide 3.10 (m, 1H), 2.79-2.93 (m, 1H), 2.67 (br. S., 3H), 2.20 (s, 3H), 1.78-1.88 (m, 1H), 1.53-1.68 (m, 3H), 1.28-1.41 (m, 2H), 0.97-1.13 (m, 2H), 0.56-0.68 (m, 1H) 306 (R or S)-N-((4,6-dimethyl- (400 MHz, DMSO-d.sub.6) .delta. = 11.73-11.56 (m, 423 2-oxo-1,2-dihydropyridin- 1 H), 8.19 (d, J = 3.1 Hz, 1 H), 8.06 (dd, J = 3-yl)methyl)-2-methyl-1- 1.4, 7.9 Hz, 1 H), 7.82 (br. S., 1 H), 7.10 (1-(tetrahydro-2H-pyran- (dd, J = 4.7, 7.8 Hz, 1 H), 5.91 (s, 1 H), 4.30 4-yl)ethyl)-1H-indole-3- (br. S., 2 H), 4.19-4.02 (m, 1 H), 3.90 (d, J = carboxamide 8.5 Hz, 1 H), 3.63 (d, J = 7.8 Hz, 1 H), 3.29 (s, 1 H), 3.06 (s, 1 H), 2.92-2.74 (m, 1 H), 2.64 (br. S., 3 H), 2.25 (s, 3 H), 2.11 (s, 3 H), 1.80 (br. S., 1 H), 1.59 (br. S., 3 H), 1.41-1.24 (m, 1 H), 1.09 (s, 2 H), 0.67- 0.52 (m, 1 H) 277 (.+-.)-1-(3-methoxy-3- (400 MHz, DMSO-d.sub.6) .delta. = 12.01-11.82 (m, 426 methylbutan-2-yl)-N-((4- 1 H), 7.91-7.82 (m, 2 H), 7.71-7.64 (m, 1 methoxy-6-methyl-2-oxo- H), 7.06-6.96 (m, 2 H), 6.25 (s, 1 H), 4.43 1,2-dihydropyridin-3- (q, J = 7.1 Hz, 1 H), 4.33 (br. S., 2 H), 3.86 yl)methyl)-2-methyl-1H- (s, 3 H), 3.14-3.09 (m, 3 H), 2.61 (s, 3 H), indole-3-carboxamide 2.23 (s, 3 H), 1.58-1.52 (m, 3 H), 1.27 (s, 3 H), 0.88 (s, 3 H) 275 (.+-.)-N-((4,6-dimethyl-2- 410 oxo-1,2-dihydropyridin-3- yl)methyl)-1-(3- methoxypentan-2-yl)-2- methyl-1H-indole-3- carboxamide 294 (.+-.)-N-((4-methoxy-6- (CDCl.sub.3, 400 MHz) .delta. 7.85 (t, J = 6.4 Hz, 412 methyl-2-oxo-1,2- 1H), 7.45 (s, 2H), 7.08-7.03 (m, 2H), 5.93 dihydropyridin-3- (s, 1H), 4.71-4.61 (m, 2H), 4.36 (s, 1H), yl)methyl)-1-(3- 3.90 (s, 4H), 2.95 (s, 3H), 2.75 (s, 3H), 2.17 methoxybutan-2-yl)-2- (s, 3H), 1.57 (d, J = 7.2 Hz, 3H), 1.23 (d, methyl-1H-indole-3- J =6.0 Hz, 3H) carboxamide 290 (.+-.)-1-(3-ethoxybutan-2- (400 MHz, DMSO-d.sub.6) .delta. = 11.60 (br. s., 1 426 yl)-N-((4-methoxy-6- H), 7.72 (d, J = 7.6 Hz, 1 H), 7.67 (d, J = methyl-2-oxo-1,2- 5.1 Hz, 2 H), 7.09-6.98 (m, 2 H), 6.14 (s, 1 dihydropyridin-3- H), 4.41-4.35 (m, 1 H), 4.32 (d, J = 4.9 Hz, yl)methyl)-2-methyl-1H- 2 H), 4.03-3.93 (m, 1 H), 3.83 (s, 3 H), indole-3-carboxamide 3.25 (d, J = 9.4 Hz, 1 H), 2.82-2.72 (m, 1 H), 2.62 (br. s., 3 H), 2.19 (s, 3 H), 1.52 (d, J = 7.1 Hz, 3 H), 1.15 (d, J = 6.0 Hz, 3 H), 0.68 (t, J = 6.9 Hz, 3 H) 293 N-((4-methoxy-6-methyl- (400 MHz, CDCl.sub.3): .delta. 8.19-8.13 (m, 2H), 427 2-oxo-1,2-dihydropyridin- 7.57-7.55 (t, 1H), 6.99-6.96 (m, 1H), 5.94 3-yl) methyl)-1-(3- (s, 1H), 4.67-4.65 (m, 2H), 4.40 (m, 1H), methoxypentan-2-yl)-2- 4.16 (m, 1H), 3.16 (s, 3H), 2.80 (s, 3H), methyl-1H-pyrrolo [2,3- 2.77 (s, 3H), 2.20 (s, 3H), 1.87-1.81 (m, b] pyridine-3-carboxamide 1H), 1.67-1.65 (m, 3H), 1.53-1.45 (m, 3H), 1.02-0.99 (m, 3H) 299 N-((4-methoxy-6-methyl- (400 MHz, CDCl.sub.3) .delta. 7.87-7.86 (d, 1H), 425 2-oxo-1,2-dihydropyridin- 7.52-7.45 (m, 2H), 7.10-7.02 (m, 2H), 4.72- 3-yl) methyl)-1-(3- 4.64 (dd, 2H), 4.45-4.42 (s 1H), 3.9 (s, 3H), methoxypentan-2-yl)-2- 3.73 (s, 1H), 2.8-2.7 (d, 6H), 2.17 (s, 3H), methyl-1H-indole-3- 1.80-1.75 (m, 1H), 1.58 (s, 3H), 1.25 carboxamide (m, 1H), 1.03-0.99 (t, 3H)
Example 11
Synthesis of Methyl 1-(1-(1,4-dioxan-2-yl)ethyl)-2-methyl-1H-indole-3-carboxylate
The title compound was used as an alternate alkyl carboxylate starting material in Step 3 of Example 1.
Step 1: 1-(1,4-dioxan-2-yl)ethanone
##STR00050## To a solution of benzoic peroxide (20 g, 141 mmol) in 200 mL 1,4-dioxane at room temperature under nitrogen atmosphere was added biacetyl (24.3 g, 282 mmol). After the addition, the mixture was heated to reflux and stirred for 24 hours. The reaction mixture was cooled to 0.degree. C. The pH was adjusted to around 9 by progressively adding 2N sodium hydroxide below 0.degree. C., extracted with 2-methoxy-2-methylpropane (10 mL.times.3), and concentrated to give 1-(1,4-dioxan-2-yl)ethanone (13 g, 36%) as a yellow oil which was used directly in the next step without purification.
Step 2: 1-(1,4-dioxan-2-yl)ethanamine
##STR00051## To a solution of 1-(1,4-dioxan-2-yl)ethanone (12 g, 92.2 mmol) in 1,2-dichloroethane (100 mL) was added (4-methoxyphenyl)methanamine (25 g, 184.4 mmol) at room temperature. The mixture was allowed to stir for 3 hours, and then sodium triacetoxyborohydride (39 g, 184.4 mmol) was added. The resulting mixture was allowed to stir for 48 hours at room temperature. The reaction mixture was quenched by adding water, extracted with dichloromethane (100 mL.times.3). The combined organic phase was dried by anhydrous sodium sulphate, and then filtered. The filtrate was concentrated and purified by column chromatograph on silica gel (elute: dichloromethane/methanol 100:1.fwdarw.50:1.fwdarw.20:1) to give 1-(1,4-dioxan-2-yl)-N-(4-methoxybenzyl)ethanamine (16.4 g, 71%) as a yellow solid. LCMS (M+H.sup.+) m/z: calcd. 251.15. found 251.9. To a solution of 1-(1,4-dioxan-2-yl)-N-(4-methoxybenzyl)ethanamine (5 g, 19.9 mmol) in anhydrous methanol (100 mL) was added palladium 10% on carbon (240 mg, 2 mmol), then purged with hydrogen (30 psi), the mixture was allowed to stir overnight at room temperature. The reaction mixture was filtered, and the filtrate was concentrated to afford the title compound (2.5 g, 96%) as a brown solid.
The amine intermediates shown in the following table were prepared according to the general procedure outlined above using the appropriate starting materials and modifications.
TABLE-US-00017 Name Structure m/z tert-butyl 3-(1- aminoethyl)piperidine- 1-carboxylate ##STR00052## 228 (.+-.)-1-(4,4- difluorocyclohexyl) ethanamine ##STR00053## 164 (.+-.)-1-(1- (methylsulfonyl)azetidin- 3-yl)ethanamine ##STR00054## 179 (.+-.)-tert-butyl 4-(4-(1- aminoethyl)47yridine- 2-yl)piperazine-1- carboxylate ##STR00055## 307
Step 3: (E)-methyl 3-((1-(1,4-dioxan-2-yl)ethyl)imino)-2-(2-bromophenyl)butanoate
##STR00056## To a solution of 1-(1,4-dioxan-2-yl)ethanamine (2.5 g, 19 mmol) in methanol (100 mL) was added methyl 2-(2-bromophenyl)-3-oxobutanoate (5.4 g, 20 mmol) and acetic acid (1.8 g, 30 mmol). The resulting reaction system was warm to reflux and allowed to stir overnight. The reaction mixture was concentrated and purified by column chromatographed on silica gel (eluted: dichloromethane/methanol 50:1.fwdarw.20:1.fwdarw.5:1) the title compound (1 g, 14%) as a brown solid. LCMS (M+H.sup.+) m/z: calcd. 383.07. found 384.9.
The imino-bromo intermediates shown in the following table were prepared according to the general procedure outlined above using the appropriate starting materials (e.g., one of the amines set forth in the table in Step 2 of this example) and modifications.
TABLE-US-00018 Name Structure m/z (E)-tert-butyl 3-(1-((3-(2- bromophenyl)- 4-methoxy-4- oxobutan-2- ylidene)amino) ethyl)piperidine- 1-carboxylate ##STR00057## 482 (.+-.)-(E)-methyl 2-(2-bromophenyl)- 3-((1-(4,4- difluorocyclohexyl) ethyl)imino) butanoate ##STR00058## 417 (E)-tert-butyl 4-((3-(2- bromophenyl)- 4-methoxy-4- oxobutan-2- ylidene)amino) piperidine-1- carboxylate ##STR00059## 454 (Z)-methyl 2-(2- bromophenyl)-3- (quinolin-5- ylamino)but-2- enoate ##STR00060## 398 (E)-methyl 2-(2- bromophenyl)-3- (cyclopentylimino) butanoate ##STR00061## 339 (E)-methyl 2-(2- bromophenyl)-3- ((6- methylquinolin-5- yl)imino)butanoate ##STR00062## 412 (.+-.)-(E)-methyl 2- (2-bromophenyl)- 3-((1-(1- (methylsulfonyl) azetidin-3- yl)ethyl)imino) butanoate ##STR00063## 432 (.+-.)-(E)-tert-butyl 4-(4-((3-(2- bromophenyl)-4- methoxy-4- oxobutan- 2-ylidene) amino)pyridine- 2-yl)piperazine- 1-carboxylate ##STR00064## 559 (E)-methyl 2-(2- bromophenyl)-3- ((2,5- dimethylphenyl) amino)but-2- enoate ##STR00065## 375 (E)-methyl 2-(2- bromophenyl)-3- ((2,3- dimethylphenyl) amino)but-2- enoate ##STR00066## 375 (E)-methyl 2-(2- bromophenyl)-3- (quinolin-6- ylimino)butanoate ##STR00067## 398
Step 4: Methyl 1-(1-(1,4-dioxan-2-yl)ethyl)-2-methyl-1H-indole-3-carboxylate
##STR00068## To a solution of (E)-methyl 3-((1-(1,4-dioxan-2-yl)ethyl)imino)-2-(2-bromophenyl)butanoate (400 mg, 1.1 mmol) in dioxane (3 mL) was added Chloro[2-(dicyclohexylphosphino)-3,6-dimethoxy-2',4',6'-triisopropylbiphe- nyl][2-(2-aminoethyl)phenyl]Pd(II) (160 mg, 0.2 mmol), 2-Dicyclohexyphosphino-2',6'-diisopropoxybiphenyl (93 mg, 0.2 mmol) and sodium tert-butoxide (192 mg, 2 mmol). The resulting reaction mixture was heated to 120.degree. C. with stirring for 30 mins in a microwave. The reaction mixture was quenched by adding water and was extracted with ethyl acetate (25 mL.times.3). The combined organic phase was dried by anhydrous sodium sulphate, and then filtered. The filtrate was concentrated and purified by column chromatograph on silica gel (eluted: petrol ether/acetic ester 10:1.fwdarw.5:1.fwdarw.2:1) to afford the title compound (282 mg, 89%) as yellow solid. LCMS (M+H.sup.+) m/z: calcd. 303.15. found 303.9.
The compound shown in the following table was prepared according to the general procedure outlined above using the appropriate starting materials (e.g., one of the imino-bromo intermediates shown in the table in Step 3 of this example) and modifications.
TABLE-US-00019 Name Structure m/z (.+-.)-methyl 1-(1-(4,4- difluorocyclohexyl) ethyl)- 2-methyl- 1H-indole-3- carboxylate ##STR00069## 336
These alkyl carboxylates were also used as starting material in Step 3 of Example 1 in the synthesis of certain compounds of the invention.
Example 12
Chiral Separation of Compound 219 to Afford Compounds 223 and 224
N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-(1-methoxy- propan-2-yl)-2-methyl-1H-indole-3-carboxamide (200 mg) (Compound 219) was subjected to chiral chromatography via supercritical fluid chromatography (SFC) (A: C.sub.2H.sub.5OH, B: NH.sub.3--H.sub.2O. A:B=55:45 AD column) to afford the separate enantiomers 223 (peak 1) and 224 (Peak 2) (60 mg each) LCMS 398 (M+1).sup.+ 1H NMR (400 MHz, CD.sub.3OD) .delta. 7.69 (d, J=7.2 Hz, 1H), 7.53 (d, J=7.6 Hz, 1H), 7.12 (m, 2H), 6.26 (s, 1H), 4.80 (m, 1H), 4.52 (s, 2H), 3.99 (m, 4H), 3.75 (m, 1H), 3.20 (s, 3H), 2.62 (s, 3H), 2.31 (s, 3H), 1.59 (d, J=7.2 Hz, 3H). The optical rotation of each enantiomer was not determined.
The compounds shown in the following table were prepared according to the general chiral chromatography procedure outlined above. The optical rotation of the separated enantiomers was not determined, but the elution peak ("Peak 1" or "Peak 2") is indicated. Structures of each compound are shown in FIG. 1.
TABLE-US-00020 Com- pound Name .sup.1H NMR m/z 217 (R or S)-N-((4,6-dimethyl-2- (400 MHz, CD.sub.3OD) .delta. 7.74 (m, 1H), 382 oxo-1,2-dihydropyridin-3- 7.57 (d, J = 7.6 Hz, 1H), 7.15 (m, 2H), yl)methyl)-1-(1- 6.14 (s, 1H), 4.86 (m, 1H), 4.55 (s, 2H), methoxypropan-2-yl)-2- 4.02 (m, 1H), 3.77 (m, 1H), 3.22 (s, methyl-1H-indole-3- 3H), 2.65 (s, 3H), 2.43 (s, 3H), 2.26 (s, carboxamide-PEAK 1 3H), 1.62 (d, J = 7.2 Hz, 3H) 218 (R or S)-N-((4,6-dimethyl-2- (400 MHz, CD.sub.3OD) 6 7.74 (m, 1H), 382 oxo-1,2-dihydropyridin-3- 7.57 (d, J = 7.6 Hz, 1H), 7.15 (m, 2H), yl)methyl)-1-(1- 6.14 (s, 1H), 4.86 (m, 1H), 4.55 (s, 2H), methoxypropan-2-yl)-2- 4.02 (m, 1H), 3.77 (m, 1H), 3.22 (s, 3H), methyl-1H-indole-3- 2.65 (s, 3H), 2.43 (s, 3H), 2.26 (s, 3H), carboxamide-PEAK 2 1.62 (d, J = 7.2 Hz, 3H) 252 (R or S)-(.+-.)-1-(1- NMR (400 MHz, CDCl.sub.3): 379 cyclopropylethyl)-N-((4,6- .delta. 8.32-8.34 (d, 1H), 8.18-8.2 (d, 1H), dimethyl-2-oxo-1,2- 7.27-7.30 (m, 1H), 6.70 (s, 1H), 4.47 (s, dihydropyridin-3-yl)methyl)- 2H), 3.94-3.95 (d, 1H), 2.61 (s, 3H), 2-methyl-1H-pyrrolo[2,3- 2.43 (s, 3H), 2.29-2.30 (s, 3H), 1.57- b]pyridine-3-carboxamide 1.59 (d, 3H), 0.63-0.64 (t, 1H), 0.27- PEAK 1 0.64 (m, 2H), 0.02-0.04 (t, 1H) 253 (R or S)-(.+-.)-1-(1- NMR (400 MHz, CDCl.sub.3): 379 cyclopropylethyl)-N-((4,6- .delta. 8.32-8.34 (d, 1H), 8.18-8.2 (d, 1H), dimethyl-2-oxo-1,2- 7.27-7.30 (m, 1H), 6.70 (s, 1H), 4.47 (s, dihydropyridin-3-yl)methyl)- 2H), 3.94-3.95 (d, 1H), 2.61 (s, 3H), 2-methyl-1H-pyrrolo]2,3- 2.43 (s, 3H), 2.29-2.30 (s, 3H), 1.57- b]pyridine-3-carboxamide 1.59 (d, 3H), 0.63-0.64 (t, 1H), 0.27- PEAK 2 0.64 (m, 2H), 0.02-0.04 (t, 1H) 256 (R or S)-N-((4-methoxy-6- (400 MHz, CDCl.sub.3) .delta. 13.23 (s, 1H), 399 methyl-2-oxo-1,2- 8.16-8.17 (m, 1H), 8.11-8.13 (m, 1H), dihydropyridin-3-yl)methyl)- 7.57-7.60 (t, J = 5.2 Hz, 1H), 6.93-6.96 1-(1-methoxypropan-2-yl)-2- (m, 1H), 5.92 (s, 1H), 4.82-4.83 (d, J = methyl-1H-pyrrolo[2,3- 2.4 Hz, 1H), 4.65-4.66 (d, J = 6.4 Hz, b]pyridine-3-carboxamide 2H), 3.89 (s, 3H), 3.81-3.85 (m, 1H), PEAK 1 3.22 (s, 3H), 2.79 (s, 3H), 2.17 (s, 3H), 1.64-1.66 (d, J = 8.0 Hz, 3H) 257 (R or S)-N-((4-methoxy-6- (400 MHz, CDCl.sub.3) .delta. 13.23 (s, 1H), 399 methyl-2-oxo-1,2 8.16-8.17 (m, 1H), 8.11-8.13 (m, 1H), dihydropyridin-3-yl)methyl)- 7.57-7.60 (t, J = 5.2 Hz, 1H), 6.93-6.96 1-(1-methoxypropan-2-yl)-2- (m, 1H), 5.92 (s, 1H), 4.82-4.83 (d, J = methyl-1H-pyrrolo[2,3- 2.4 Hz, 1H), 4.65-4.66 (d, J = 6.4 Hz, b]pyridine-3-carboxamide 2H), 3.89 (s, 3H), 3.81-3.85 (m, 1H), PEAK 2 3.22 (s, 3H), 2.79 (s, 3H), 2.17 (s, 3H), 1.64-1.66 (d, J = 8.0 Hz, 3H) 307 Trans-(R or S, R or S)-N-((4- (CDCl.sub.3, 400 MHz) .delta. 7.85 (t, J = 6.4 412 methoxy-6-methyl-2-oxo-1,2- Hz, 1H), 7.45 (s, 2H), 7.08-7.03 (m, dihydropyridin-3-yl)methyl)- 2H), 5.93 (s, 1H), 4.71-4.61 (m, 2H), 1-(3-methoxybutan-2-yl)-2- 4.36 (s, 1H), 3.90 (s, 4H), 2.95 (s, 3H), methyl-1H-indole-3- 2.75 (s, 3H), 2.17 (s, 3H), 1.57 (d, J = carboxamide 7.2 Hz, 3H), 1.23 (d, J = 6.0 Hz, 3H) PEAK 1 308 Trans-(R or S, R or S)-N-((4- (CDCl.sub.3, 400 MHz) .delta. 7.85 (t, J = 6.4 412 methoxy-6-methyl-2-oxo-1,2- Hz, 1H), 7.45 (s, 2H), 7.08-7.03 (m, dihydropyridin-3-yl)methyl)- 2H), 5.93 (s, 1H), 4.71-4.61 (m, 2H), 1-(3-methoxybutan-2-yl)-2- 4.36 (s, 1H), 3.90 (s, 4H), 2.95 (s, 3H), methyl-1H-indole-3- 2.75 (s, 3H), 2.17 (s, 3H), 1.57 (d, J = carboxamide 7.2 Hz, 3H), 1.23 (d, J = 6.0 Hz, 3H) PEAK 2
Example 1
Synthesis of tert-butyl 1-(2,3-dihydro-1H-inden-1-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carbox- ylate
The title compound as starting material in Step 3 of Example 36 in the synthesis of certain compounds of the invention.
Tert-butyl 2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate
##STR00070## To a 500 mL round-bottom flask that contains N-acetyl-N-(3-bromopyridin-2-yl)acetamide (14.815 g, 57.6 mmol), was added copper(I) iodide (1.098 g, 5.76 mmol), L-proline (1.327 g, 11.53 mmol), cesium carbonate (28.2 g, 86 mmol), then t-butyl acetoacetate (11.47 ml, 69.2 mmol) and dioxane (100 mL). The reaction was vac/purged with N.sub.2 3.times. then fitted with a septum and a N.sub.2 inlet and heated overnight at 70.degree. C. The inorganic solids were removed by filtration over celite and the cake was washed with 100 mL EtOAc. This solution was concentrated and the residue was partitioned between 250 mL brine and 250 mL EtOAc. The aq. Layer was further extracted with EtOAc (2.times.250 mL) and the combined organic layer was dried over Na.sub.2SO.sub.4, filtered, concentrated and purified by CC using 1:1 EtOAc:Hex as eluent to provide (2.7 g, 20.2%) of tert-butyl 2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate. LRMS (M+H.sup.+) m/z: calc'd 233.28. found 233.1.
Tert-butyl 1-(2,3-dihydro-1H-inden-1-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridin- e-3-carboxylate
##STR00071## A solution of ethyl tert-butyl 2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate (100 mg, 0.74 mmol), 2,3-dihydro-1H-inden-1-ol (176 mg, 0.74 mmol), PPh.sub.3 (195 mg, 1.49 mmol) was stirred in dry THF (10 mL) at 0.degree. C. under a nitrogen atmosphere. To this mixture was added drop-wise DIAD (150 mg, 1.48 mmol) over a period of 5 min, and the reaction was stirred at room temperature for 16 hours. The mixture was washed with brine, dried and concentrated to afford the crude product. The crude product was purified by silca gel chromatography (petroleum ether/ethyl acetate=5:1) to afford the tert-butyl-1-(2,3-dihydro-1H-inden-1-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridi- ne-3-carboxylate (150 mg, 60%).
The compound shown in the following table was prepared according to the general procedure outlined above using the appropriate starting materials and modifications.
TABLE-US-00021 Name Structure m/z (.+-.)-tert-butyl 1- (1-cyclopropylethyl)- 2-methyl-1H-pyrrolo [2,3-b]pyridine-3- carboxylate ##STR00072## 301
Each of the above alkyl carboxylates was used as starting material in Step 3 of Example 1 in the synthesis of certain compounds of the invention.
Example 13
Synthesis of isolated N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-((2R or 2S, 3R or 3S)-3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-c- arboxamide diastereomers (Compounds 261, 266, 267 and 302)
Step 1: Tert-butyl 2-methyl-1-(3-oxobutan-2-yl)-1H-pyrrolo[2,3-b]pyridine-3-carboxylate
##STR00073## To a solution of tert-butyl 2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate (5.0 g, 21.53 mmol) in CH.sub.3CN (50 mL) was added Cs.sub.2CO.sub.3 (21.0 g, 64.58 mmol), potassium iodide (3.57 g, 21.53 mmol). The mixture was stirred at 27.degree. C. for 30 minutes. Then 3-chlorobutan-2-one (2.75 g, 25.83 mmol) was added and the mixture was stirred at 70.degree. C. for 12 hours. The mixture was filtered and the filtrate was concentrated. The residue was purified by column (Elute: Petroleum ether:Ethyl acetate=50:1) to give tert-butyl 2-methyl-1-(3-oxobutan-2-yl)-1H-pyrrolo[2,3-b]pyridine-3-carboxylate as a yellow-green oil. (3.23 g, yield 50%) LCMS (M+H.sup.+) m/z: calcd 303.37. found 302.9. 1H NMR (400 MHz, CDCl.sub.3): .delta. 8.32-8.30 (m, 1H), 8.25-8.23 (m, 1H), 7.17-7.14 (m, 1H), 5.50-5.44 (m, 1H), 2.71 (s, 3H), 1.96 (s, 3H), 1.65-1.67 (d, 3H), 1.64 (s, 9H).
Step 2: Tert-butyl 1-(3-hydroxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate
##STR00074## To the solution of tert-butyl 2-methyl-1-(3-oxobutan-2-yl)-1H-pyrrolo[2,3-b]pyridine-3-carboxylate (3.1 g, 10.25 mmol) in methanol (30 mL) was added sodium borohydride (0.30 g, 8.2 mmol) at 0.degree. C. After 30 minutes, another batch of sodium borohydride (0.30 g, 8.2 mmol) was added at 0.degree. C. After the reaction completed about 2 h later, water (30 ml) was added dropwise very carefully to quench the reaction. The mixture was extracted with CH.sub.2Cl.sub.2. The extraction was dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to give tert-butyl 1-(3-hydroxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate as a yellow solid. (3.0 g, yield 96%) LCMS (M+H.sup.+) m/z: calcd 305.38. found 304.9. 1H NMR (400 MHz, CDCl.sub.3): .delta. 8.31-8.29 (m, 1H), 8.13-8.12 (m, 1H), 7.11-7.07 (m, 1H), 4.46-4.43 (m, 1H), 4.12 (m, 1H), 2.73 (s, 3H), 1.58 (s, 9H), 1.51-1.49 (d, 3H), 0.92-0.91 (d, 3H).
Step 3: Tert-butyl-1-(3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyrid- ine-3-carboxylate
##STR00075## To dry THF (20 mL) was added NaH (60% in mineral oil, 2.37 g, 59.14 mmol). Then the mixture was stirred at 27.degree. C. for 20 minutes, then tert-butyl 1-(3-hydroxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate (3.0 g, 9.86 mmol) was added. The mixture was stirred at 27.degree. C. for 1 hour, then added by CH.sub.3I (13.99 g, 98.6 mmol). The mixture was stirred for 12 hours at 27.degree. C. and then cooled to 0.degree. C. Sat. NH.sub.4Cl was added and extracted with CH.sub.2Cl.sub.2. The extraction was dried over sodium sulfate, filtered and concentrated to give tert-butyl-1-(3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridin- e-3-carboxylate as a yellow oil. (3.2 g, yield 100%) LCMS (M+H.sup.+) m/z: calcd. 319.41. found 318.9.
Step 4: 1-(3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carbo- xylic acid
##STR00076## To the pre-cooled solution of tert-butyl 1-(3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylate (3.0 g, 9.42 mmol) in CH.sub.2Cl.sub.2 (20 mL) was added trifluoroacetic acid (20 mL) dropwise. The solution was stirred at 27.degree. C. for 1.5 hours. The solvent was removed under vacuum at 27.degree. C. The residue was used for next step without purified. LCMS (M+H+) m/z: calcd 263.30. found 262.9.
Step 5: N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-(3-- methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide
##STR00077## To a solution of 1-(3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxylic acid (2.4 g, 9.15 mmol) in DMF (30 mL) was added TEA (4.2 g, 41.50 mmol), 3-(aminomethyl)-4-methoxy-6-methylpyridin-2(1H)-one hydrochloride (2.1 g, 12.81 mmol) After stirred for 10 minutes at 27.degree. C., the mixture was cooled and added HATU (5.56 g, 14.64 mmol). The mixture was stirred at 27.degree. C. for 72 hours and 30% of S.M. remained. Then the mixture was heated at 80.degree. C. for 5 hours. The solution was diluted with brine (100 mL) and extracted with CH.sub.2Cl.sub.2 (100 mL*3). The extractions were combined and dried over Na.sub.2SO.sub.4. The solvent was evaporated under vacuum and the residue was purified by flash column (Eluent: dichloromethane:methanol=95:5) to give N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-(3-methoxy- butan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide. (3.6 g, yield 95%)
Step 6: Separation of N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-(3-methoxy- butan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide: Isomers (Compounds 261, 266, 267, and 302)
##STR00078## ##STR00079## The mixture or isomers from step 5, N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-(3-methoxy- butan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide was purified by prep-HPLC (Condition: Column: SHIMADZU LC-8A, 250*50 mm*10 um; Mobile phase A: water with 0.2% formic acid; Mobile phase B: MeCN; column temperature: 30.degree. C.; Gradient: B in A 10-50%) to give a major isomer pair (Compound 261 and Compound 266 combined) (1.0 g, purity 98.8%) and a minor isomer pair (Compound 267 and Compound 302 combined) (180 mg, purity 63%). The resulting isomer pairs were individually separated by SFC (Condition: Column: Chiral-pak AD 250*30 mm*5 um; Mobile phase A: Supercritical CO.sub.2; Mobile phase B: IPA+NH.sub.3.H.sub.2O; Gradient: B/A: 75:25) to give the following individual single compounds:
Compound 261, N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-((2R or 2S, 3R or 3S)-3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-c- arboxamide (Major Isomer Pair; Peak 1): .sup.1H NMR (400 MHz, CDCl.sub.3): .delta. 8.173-8.157 (m, 1H), 8.140-8.116 (m, 1H), 7.582-7.555 (m, 1H), 6.968-6.936 (m, 1H), 5.927 (s, 1H), 4.707-4.609 (m, 2H), 4.348 (s, 1H), 3.892 (s, 3H), 2.869 (s, 3H), 2.788 (s, 3H), 2.173 (s, 3H), 1.644-1.627 (d, 3H), 1.263-1.249 (d, 3H).
Compound 266, N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-((2R or 2S, 3R or 3S)-3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-c- arboxamide (Major Isomer Pair; Peak 2): .sup.1H NMR (400 MHz, CDCl.sub.3): .delta. 8.179-8.163 (m, 1H), 8.143-8.120 (m, 1H), 7.558-7.531 (m, 1H), 6.986-6.954 (m, 1H), 5.931 (s, 1H), 4.702-4.605 (m, 2H), 3.897 (s, 3H), 2.892 (s, 3H), 2.789 (s, 3H), 2.189 (s, 3H), 1.647-1.629 (d, 3H), 1.267-1.252 (d, 3H).
Compound 267, N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-((2R or 2S, 3R or 3S)-3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-c- arboxamide (Minor Isomer Pair; Peak 1): .sup.1H NMR (400 MHz, CDCl.sub.3): .delta. 8.174-8.162 (d, 1H), 8.111-8.094 (d, 1H), 7.551-7.526 (m, 1H), 6.993-6.961 (m, 1H), 5.935 (s, 1H), 4.683-4.579 (m, 2H), 3.887 (s, 3H), 3.442 (s, 3H), 2.753 (s, 3H), 2.194 (s, 3H), 1.695-1.678 (d, 3H), 0.781-0.768 (d, 3H).
Compound 302, N-((4-methoxy-6-methyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-1-((2R or 2S, 3R or 3S)-3-methoxybutan-2-yl)-2-methyl-1H-pyrrolo[2,3-b]pyridine-3-c- arboxamide (Minor Isomer Pair; Peak 2): .sup.1H NMR (400 MHz, CDCl.sub.3): .delta. 8.177-8.166 (d, 1H), 8.122-8.104 (d, 1H), 7.587-7.562 (m, 1H), 6.984-6.952 (m, 1H), 5.933 (s, 1H), 4.698-4.591 (m, 2H), 4.426 (s, 2H), 3.983 (s, 3H), 3.448 (s, 3H), 2.764 (s, 3H), 2.180 (s, 3H), 1.701-1.684 (d, 3H), 0.786-0.772 (d, 3H).
Example 14
Synthesis of (.+-.)-N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl-1-- (1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxamide
Step 1: 1-(3-methoxyphenyl)ethanol
##STR00080## To a stirred solution of 3-Amino-4-picoline (7 g, 64.8 mmol) in anhydrous THF (200 mL), sec-BuLi (150 mL, 1.3M in cyclohexane, 194 mmol) was added dropwise over 20 minutes at -78.degree. C. The solution was warmed to room temperature and stirred at 3 hours. Ethyl acetate (2.3 g, 25.9 mmol) was added dropwise into the reaction at -78.degree. C. and the mixture was stirred at the same temperature for 2 hours. Methanol (50 mL) was added dropwise into the reaction over 10 minutes. The mixture was warmed to room temperature and stirred for 1 hour. A half-saturated NH4Cl (250 mL) was added. The mixture was extracted with EA. The combined organic layers were washed with brine, dried and concentrated to afford the crude product. The crude product was purified by silica gel chromatography (petroleum ether/ethyl acetate=10:1) to afford 2-methyl-1H-pyrrolo[2,3-c]pyridine (2.5 g, 73.5%).
Step 2: 2,2,2-trichloro-1-(2-methyl-1H-pyrrolo[2,3-c]pyridin-3-yl)ethanone
##STR00081## To a stirred solution of 2-methyl-1H-pyrrolo[2,3-c]pyridine (2.5 g, 18.9 mmol) and aluminum chloride (5 g, 37.8 mmol) in DCM (100 mL), trichloroacetylchloride (4.1 g, 22.7 mmol) was added dropwise into the reaction over 0.5 hours at room temperature. After stirring 2 hours, the reaction was cooled to 0.degree. C. and was quenched with water (100 mL). The resulting precipitate was isolated by filtration to afford 2,2,2-trichloro-1-(2-methyl-1H-pyrrolo[2,3-c]pyridin-3-yl)ethanone which was used for next step without further purification. Assumed 100% yield. (5.24 g).
Step 3: Methyl 2-methyl-1H-pyrrolo[2,3-c]pyridine-3-carboxylate
##STR00082## A mixture of 2,2,2-trichloro-1-(2-methyl-1H-pyrrolo[2,3-c]pyridin-3-yl)ethanone (5.24 g, 18.9 mmol) and KOH (1.2 g, 20.9 mmol) in MeOH (100 mL) was stirred at room temperature for 16 hour. The reaction mixture was concentrated to remove MeOH, the residue was partitioned between EA and Water. The organic layer was washed with brine, dried and concentrated to afford methyl 2-methyl-1H-pyrrolo[2,3-c]pyridine-3-carboxylate (3 g, 83%).
Step 4: Methyl methyl 2-methyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxylate
##STR00083## A mixture of methyl 2-methyl-1H-pyrrolo[2,3-c]pyridine-3-carboxylate (550 mg, 2.89 mmol) and sodium hydride (200 mg, 4.34 mmol) in N,N-dimethylformamide (3.0 mL) was stirred at room temperature for 0.5 hour, and then (1-bromoethyl)benzene (589 mg, 3.18 mmol) was added. The mixture was stirred at room temperature for 3 hours. The reaction mixture was poured into saturated NH.sub.4Cl and extracted with ethyl acetate. Organic layers were combined and concentrated to give a residue. The residue was purified by chromatography (petroleum ether/ethyl acetate=5:1) to give methyl 2-methyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxylate (800 mg, 94%).
Step 5: 2-methyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxylic acid
##STR00084## To a mixture of methyl 2-methyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxylate (800 mg, 2.72 mmol) and KOH (1.5 g, 27.2 mmol) in (15 mL) and water (5 mL) was refluxed for 2 hours. The mixture was adjust PH to 2 by 10% HCl and extracted with EA. The combined organic layers were washed with brine, dried and concentrated to afford the crude product. The crude product was used into the next step without more purification. 100% yield. (760 mg).
Step 6: (.+-.)-N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-me- thyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxamide (Compound 203)
##STR00085## A mixture of 2-methyl-1-(1-phenylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxylic acid (280 mg, 1.0 mmol) was added HATU (456 mg, 1.2 mmol), TEA (1 g, 10 mmol) and 3-(aminomethyl)-4,6-dimethylpyridin-2(1H)-one (182 mg, 1.2 mmol) in anhydrous dichloromethane (30 mL) was stirred at room temperature for 16 hours. To the reaction mixture was added water (10 mL), extracted with dichloromethane (30 mL.times.2). The organic layers were combined and concentrated to give a residue. The residue was rereystallized from MeCN to afford compound N-((4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl)-2-methyl-1-(1-phen- ylethyl)-1H-pyrrolo[2,3-c]pyridine-3-carboxamide as an off-white solid (80 mg, 21.6%). LRMS (M+H.sup.+) m/z: calcd 414.21. found 414. .sup.1H NMR (400 MHz, Methanol-d4) .delta.: 8.84 (s, 1H), 8.16 (d, J=7.6 Hz, 1H), 8.03 (d, J=6.8 Hz, 1H), 7.44-7.37 (m, 5H), 6.09 (s, 1H), 6.01-5.99 (m, 1H), 4.49 (s, 2H), 2.73 (s, 3H), 2.38 (s, 3H), 2.22 (s, 3H), 2.06 (d, J=7.2 Hz, 3H).
The compounds shown in the following table were prepared according to the general procedure outlined in this example using the appropriate starting materials and modifications. Structures are shown in FIG. 1.
TABLE-US-00022 Compound Name NMR m/z 240 (.+-.)-N-((4,6-dimethyl-2- (400 MHz, CHLOROFORM-d) .delta. ppm 1.63 383 oxo-1,2-dihydropyridin- (br. s., 3 H) 2.21 (s, 3 H) 2.41 (s, 3 H) 2.73 (s, 3-yl)methyl)-1-(1- 3 H) 3.24 (s, 3 H) 3.72 (dd, J = 9.81, 5.40 Hz, methoxypropan-2-yl)-2- 1 H) 3.80-3.88 (m, 1 H) 4.60 (d, J = 5.95 Hz, methyl-1H-pyrrolo[2,3- 2 H) 4.71 (dd, J = 13.23, 7.06 Hz, 1 H) 5.92 (s, c]pyridine-3- 1 H) 7.31 (d, J = 5.73 Hz, 1 H) 7.38 (br. s., 1 carboxamide H) 8.26 (d, J = 5.29 Hz, 1 H) 9.09 (br. s., 1 H) 11.07 (br. s., 1 H) 243 (.+-.)-N-((4-methoxy-6- (400 MHz, CHLOROFORM-d) .delta. ppm 1.62 431 methyl-2-oxo-1,2- (br. s., 3 H) 2.26 (s, 3 H) 2.75 (s, 3 H) 3.25 (s, dihydropyridin-3- 3 H) 3.72 (dd, J = 9.81, 5.40 Hz, 1 H) 3.80- yl)methyl)-2-methyl-1- 3.87 (m, 1 H) 3.90 (s, 3 H) 4.65 (d, J = 5.29 (1-phenylethyl)-1H- Hz, 2 H) 4.71 (dd, J = 13.78, 6.95 Hz, 1 H) pyrrolo[2,3-c]pyridine- 5.93 (s, 1 H) 7.32 (br. s., 1 H) 7.50 (br. s., 1 3-carboxamide H) 8.25 (br. s., 1 H) 9.11 (br. s., 1 H)
Example 15
General Procedures for Synthesizing Other Compounds of the Invention
General Procedure A: Indole Alkyation
##STR00086## To a cooled (0.degree. C.) solution of NH indole ester (1 equivalent) in N,N-dimethylformamide (volume to make concentration 0.4M) was added sodium hydride (60% w/w, 1.1 equivalents relative to indole). The resultant mixture was stirred for 15 minutes. Then RX (2 equivalents) was added and the reaction was allowed to warm to room temperature. The reaction was maintained at ambient temperature for 12 hours. The reaction mixture was poured into saturated ammonium chloride solution (100 mL) with stirring. The mixture was extracted with ethyl acetate (200 mL.times.2) and the combined organic phase was washed with brine, dried over magnesium sulfate, filtered, and concentrated to give crude product which was purified by column chromatography (silica gel, petroleum ether/ethyl acetate=20:1) to afford the desired alkylated Indole ester product.
General Procedure B: Saponification of Alkylated Indole Ester
##STR00087## To a solution of alkylated Indole ester (1 equivalent) in tetrahydrofuran:methanol:water (2.5:5:1, volume to make concentration 0.05M) was added lithium hydroxide (4 equivalents). The resultant reaction mixture was stirred at 60.degree. C. for 48 hours. The mixture was concentrated in vacuo. Then the residue was diluted with water (40 mL) and slowly acidified with 1N hydrogen chloride to pH=4-5. The mixture was extracted with ethyl acetate (100 mL.times.3). The combined organic layers were washed with brine, dried over magnesium sulfate, filtered and concentrated to give crude indole acid, which was used in the subsequent step without additional purification.
General Procedure C: Amide Bond Formation
##STR00088## To a solution of Indole acid (1 equivalent) in dichloromethane (volume to make concentration 0.05M) were added 1-hydroxybenzotriazole (1.5 equivalents), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (1.5 equiv.) and triethylamine (3 equiv.). The resultant mixture was stirred at room temperature for 30 minutes. Then Pyridone amine (1.2 equiv.) was added and the resultant mixture was stirred at room temperature for 16 hours. Water (50 mL) was added to the mixture. The mixture was extracted with dichloromethane (100 mL.times.2). The organic layer was concentrated in vacuo to provide crude product which was purified by column chromatography (silica gel, dichloromethane/methanol=20:1) to afford the target compound.
General Procedure D: Chiral Chromatography
Separation of chiral compounds was accomplished via normal phase HPLC or SFC (supercritical carbon dioxide fluid chromatography). Separated compounds were typically>95% ee. The absolute configuration of chiral centers was not determined.
General Procedure L: Sulphonylation
##STR00089## To a solution of Chiral amine (1 equiv.) in dichloromethane (volume to make concentration 0.1M) was added triethylamine (4 equiv.) at 18.degree. C. under N.sub.2. The reaction was cooled to 0.degree. C. and methanesulfonyl chloride (1.5 equiv.) was added. The reaction was stirred at 0.degree. C. for 1 h. Then the mixture was concentrated in vacuo and methanol and potassium carbonate were added and the reaction was stirred for another 1 h. The mixture was filtered and the crude product was purified by preparative-HPLC.
The table below lists compounds of the invention and which of the above general methods was used in their synthesis. Structures of these compounds are set forth in FIG. 1.
TABLE-US-00023 General Com- Methods pound Used and Notes Name NMR data m/z 370 General .sup.1H NMR (CDCl3, 400 MHz) 517 procedure L .delta. 12.12 (s, 1H), 9.20 (s, 1H), on (.+-.)-N-((4- 8.78 (s, 1H), 7.68 (s, 1H), methoxy-6- 5.97 (s, 1H), 4.70-4.58 (m, methyl-2-oxo- 2H), 3.91 (s, 4H), 3.62 (d, J = 1,2-dihydro- 12.0 Hz, 1H), 2.76-2.67 (m, pyridin-3- 7H), 2.45 (dd, J1 = 2.0 Hz, J, = yl)methyl)-6- 11.6 Hz, 1H), 2.32 (s, 3H), methyl-7-(1- 2.10 (t, J = 12.0 Hz, 1H), (piperidin-4- 1.67 (d, J = 6.8 Hz, 4H), yl)ethyl)-7H- 1.48-1.40 (m, 1H), 1.37-1.29 pyrrolo[2,3- (m, 1H), 1.27-1.19 (m, 1H), d]pyrimidine- 0.9-0.78 (m, 1H). 5-carboxamide
Example 16
IC.sub.50 Measurements for Inhibitors Using EZH2
EZH2 Assay:
Assays were carried out by mixing rPRC2 together with biotinylated oligonucleosome substrates in the presence of the radio-labeled enzyme co-factor, S-adenosyl-L-methionine (.sup.3H SAM) (Perkin Elmer) and monitoring the enzymatically mediated transfer of tritiated methyl groups from .sup.3H SAM to histone lysine residues. The amount of resulting tritiated methyl histone product was measured by first capturing the biotinylated oligonucleosomes in streptavidin (SAV) coated FlashPlates (Perkin Elmer), followed by a wash step to remove un-reacted .sup.3H SAM, and then counting on a TopCount NXT 384 well plate scintillation counter (Perkin Elmer). The final assay conditions for EZH2 were as follows: 50 mM Tris Buffer pH 8.5, 1 mM DTT, 69 .mu.M Brij-35 detergent, 5.0 mM MgCl.sub.2, 0.1 mg/mL BSA, 0.2 .mu.M .sup.3H SAM, 0.2 .mu.M biotinylated oligonucleosomes, 3.6 .mu.M H3K27me3 peptide and 2 nM EZH2.
Compound IC.sub.50 measurements were obtained as follows: Compounds were first dissolved in 100% DMSO as 10 mM stock solutions. Ten point dose response curves were generated by dispensing varying amounts of the 10 mM compound solution in 10 wells of the 384 well plate (Echo; Labcyte), pure DMSO was then used to backfill the wells to insure all wells have the same amount of DMSO. A 12.5 .mu.L volume of the HMT enzyme, H3K27me3 peptide and oligonucleosome substrate in assay buffer was added to each well of the assay plate using a Multidrop Combi (Thermo-Fisher). Compounds were pre-incubated with the enzyme for 20 min, followed by initiation of the methyltransferase reaction by addition of 12.5 .mu.L of .sup.3H SAM in assay buffer (final volume=25 .mu.L). The final concentrations of compounds ranged from a top default concentration of 80 .mu.M down to 0.16 .mu.M in ten 2-fold dilution steps. Reactions were carried out for 60 minutes and quenched with 20 .mu.L per well of 1.96 mM SAH, 50 mM Tris pH 8.5, 200 mM EDTA. Stopped reactions were transferred to SAV coated Flash-plates (Perkin Elmer), incubated for 120 min, washed with a plate washer, and then read on the TopCount NXT (1.0 min/well) to measure the amount of methyl histone product formed during the reaction. The amount of methyl histone product was compared with the amount of product formed in the 0% and 100% inhibition control wells allowing the calculation of % Inhibition in the presence of the individual compounds at various concentrations. IC.sub.50's were computed using a 4 parameter fit non-linear curve fitting software package (XLFIT, part of the database package, ActivityBase (IDBS)) where the four parameters were IC.sub.50, Hill slope, pre-transitional baseline (0% INH), and post-transitional baseline (100% INH); with the latter two parameters being fixed to zero and 100%, respectively, by default.
Assay for Y641N EZH2 was performed as above using reconstituted H3K27Me2 oligonucleosomes as substrate.
Table 2 shows the activity of selected compounds of this invention in the EZH2 and Y641N EZH2 activity inhibition assay. IC.sub.50 values are reported as follows: "A" indicates an IC.sub.50 value of less than 100 nM; "B" indicates an IC.sub.50 value of 100 nM to 1 .mu.M; "C" indicates an IC.sub.50 value of greater than 1 .mu.M and less than 10 .mu.M for each enzyme; "D" indicates an IC.sub.50 value of greater than 10 .mu.M for each enzyme; and "*(X .mu.M)" indicates that no inhibition was observed at the highest concentration (i.e., X .mu.M) of compound tested.
TABLE-US-00024 TABLE 2 IC50 Values for Compounds of Formula I against EZH2 and Y641N EZH2 Mutant Enzymes. Compound Y641N No. EZH2 IC.sub.50 EZH2 IC.sub.50 183 A A 204 A B 211 A B 212 A B 217 B B 218 A A 219 A A 223 A B 224 A A 229 C D 230 A B 231 A B 234 C D 235 B C 236 *(0.5 .mu.M) *(10 .mu.M) 240 A B 241 A B 243 A B 252 A B 253 A B 256 A B 257 B C 261 A A 266 B B 267 A B 273 A A 275 A A 277 A A 280 B C 284 A B 288 A A 290 A B 293 A B 294 A A 298 A A 299 A A 300 A A 302 B C 304 A A 306 A A 307 A A 308 B B 310 A A 313 A A 314 A A 316 A A 317 A A 321 A A 327 A A 335 A A 336 A A 337 A A 341 A A 342 A A 343 A A 344 A A 345 A A 346 A A 347 A A 352 A A 355 A A 356 A A 357 A A 358 A A 359 A A 360 A A 362 A A 363 A A 364 A B 365 A A 366 A A 367 A A 368 A A 369 A A 370 A A 373 A A 375 A A 376 A A 377 A A
Example 17
EC50 Measurements for Inhibitors in HeLa Cell Assays
H3K27me3 MSD Hela Assay.
Trypsinized HeLa cells were counted and diluted in 10% DMEM (Life Technologies, Cat. #10569) to 5000 cells/75 .mu.L. Seventy-five .mu.L of cells were place in each well of a 96-well flat-bottomed plate and incubated at 37.degree. C. for 4 hours. Twenty-five .mu.L of test compound (at various concentrations) was added to the cells and incubation continued at 37.degree. C. for 96 hours. Media was then removed and the cells rinsed once with ice cold PBS. Forty .mu.L of ice-cold MSD Buffer AT (10 mM HEPES, pH 7.9, 5 mM MgCl.sub.2, 0.25M sucrose, Benzonase (1:10000), 1% Triton X-100 supplemented with fresh 1.times. Protease Inhibitor cocktail and 1 mM 4-(2-Aminoethyl)benzenesulfonyl fluoride hydrochloride (AEBSF)) was added to each well and the plates placed on ice for 30 minutes. Ten .mu.L of 5M NaCl was then added to each well and incubation on ice continued for another 15 minutes. The material in each well was suspended pipetting up and down and then transferred to a new 96 well plate. The emptied wells were rinsed with 150 uL ice-cold 20 mM Tris pH 7.5, 1 mM EDTA, 1 mM EGTA, supplemented with fresh 1.times. Protease Inhibitor cocktail and 1 mM AEBSF ("NO salt NO detergent buffer) and transferred to the respective wells in the new plate. Three hundred .mu.L of NO Salt NO detergent buffer was then added to each well of lysates and the plates frozen at -80.degree. C.
On the same day, an appropriate number of MSD standard bind 96-well plates were coated with 30 .mu.L/well of total H3 capture antibody (Millipore, Cat # MAB3422) at 1 .mu.g/mL concentration in PBS. The antibody solution was evenly distributed first by tapping gently on the sides of the plates and then by shaking the plates for a few minutes at 1000 rpm. Antibody coated plates were stored at 4.degree. C. overnight.
The next day the lysates are thawed to RT. The antibody coated MSD plates are washed 3.times. with TBS-T (Tris-buffered saline (Fisher Scientific, Cat #BP2471-1)+0.2% Tween-20). One-hundred fifty .mu.L of 5% Blocker A in TBS-T is added to each well. The wells are covered and shaken on a shaker at RT for one hour. The Blocker A step is repeated a second time. After removing the blocker, 25 .mu.L of cell lysate is transferred into each antibody coated well. The plates are shaken for 2 hours at RT, the lysate removed and the plates again washed with Blocker A in TBS-T. Twenty-five .mu.L of appropriate freshly prepared antibody mix (including both primary and secondary antibodies) is added to each well and the plates shaken for 1 hour at RT. The antibody mix used was one (or both) of those indicated in the table below:
TABLE-US-00025 Concen- Anti-rabbit 1% tration Primary detection blocker Ab (.mu.g/mL) Ab (.mu.L) Ab (.mu.L) A (.mu.L) H3K27me3 33 37.88 5.00 5000 H3 12 52.08 5.00 5000
Both H3 antibodies were obtained from Cell Signalling (Cat #s 4499 and 9733). The goat anti-rabbit antibody was obtained from Meso-Scale Discovery (Cat #R32AB-1).
The antibody mix was then removed and the wells washed with Blocker A. One hundred-fifty .mu.L of freshly prepared 1.times.MSD Read Buffer (Meso-Scale Discovery; Cat #R927C-2) was then added to each well and the plates read on a MSD Sector 2400 Plate Reader.
Data was analyzed using Assay Assistant (Constellation Pharmaceuticals In-house product) and Activity Base (IDBS Ltd, Surrey, UK) template. Data files were imported to Assay Assistant and assay conditions were specified. A unique Analysis ID was created and the data files exported to Activity Base. An analysis template was created on Activity Base to measure dose-dependent inhibition of H3K27me3 mark and cell viability respectively. Readout of DMSO wells were used to normalize the data. Resulting curves were fitted using Activity base software Model 205 (IDBS Ltd, Surrey, UK). The data was checked for quality, validated and integrated in excel format using SARview (IDBS Ltd, Surrey, UK).
H3K27me3 Alpha Hela Assay (AlphaLISA).
Ten different doses of each test compound (in a series of 3-fold dilutions) were plated in duplicate 384-well tissue culture treated plates (Catalog #781080; Greiner Bio One; Monroe, N.C.). Hela cells grown in culture were trypsinized and counted using a Countess.RTM. cell counter (Catalog #C10281; Life Technologies, Grand Island, N.Y.). Cell were diluted to 67,000 cells per mL in 10% DMEM (Catalog #10569-010 Life Technologies, Grand Island, N.Y.) and 15 .mu.L (1,000 cells) were plated into each well using the Biotek MicroFlo.TM. Select Dispenser (BioTek Instruments, Inc. Vermont, USA),) of the 384-well plate. Plates were incubated at 37.degree. C./5% CO.sub.2 for 72 hrs. One of the duplicate plates was processed for HeLa assay and the other for viability.
To the plate processed for AlphaLISA was added 5 .mu.L per well Cell-Histone Lysis buffer (1.times.) (Catalog #AL009F1 Perkin Elmer; Waltham, Mass.) and the plate was incubated at RT for 30 minutes on a plate shaker with low speed (Model #4625-Q Thermo Scientific; Waltham, Mass.). Then, 10 .mu.L per well Histone Extraction buffer (catalog #AL009F2; Perkin Elmer; Waltham, Mass.) was added and the plate further incubated at RT for 20 min on plate shaker with low speed. To each well was then added 10 .mu.L per well of a 5.times. mix of anti-K27me3 acceptor beads plus Biotinylated anti-Histone H3 (C-ter) Antibody (diluted to 3 nM final) (Catalog #AL118 Perkin Elmer; Waltham, Mass.). Dilution of the acceptor beads and then anti-Histone H3 was with 1.times. Histone Detection buffer (Catalog #AL009F3 Perkin Elmer; Waltham, Mass.) which was produced diluted from the 10.times. stock provided. The plate was sealed with an aluminum plate sealer and incubated at 23.degree. C. for 60 min. We then added 10 .mu.L 5.times. solution of Streptavidin Donor beads (Catalog #6760002 Perkin Elmer; Waltham, Mass.) (20 .mu.g/mL final in 1.times. Histone Detection Buffer), sealed the plate with Aluminum plate sealer and incubated at 23.degree. C. for 30 min. The plates were then read using an EnVision-Alpha Reader (model #2104 Perkin Elmer; Waltham, Mass.).
Cell viability was assayed by adding 15 .mu.L of Cell Titer Glo ((Catalog #G7571 Promega Madison, Wis.) to each well with cells with media. The plates were incubated foat RT for 15-20 minutes on a plate shaker at low speed. The plates were then read using an EnVision-Alpha Reader (model #2104 Perkin Elmer; Waltham, Mass.).
Data from both assays was analyzed using Assay Assistant (Constellation Pharmaceuticals In-house product) and Activity Base (IDBS Ltd, Surrey, UK) template. Data files were imported to Assay Assistant and assay conditions were specified. A unique Analysis ID was created and the data files exported to Activity Base. An analysis template was created on Activity Base to measure dose-dependent inhibition of H3K27me3 mark and cell viability respectively. Readout of DMSO wells were used to normalize the data. Resulting curves were fitted using Activity base software Model 205 (IDBS Ltd, Surrey, UK). The data was checked for quality, validated and integrated in excel format using SARview (IDBS Ltd, Surrey, UK).
Table 3 shows the activity of selected compounds of this invention in the two different HeLa cell assays described above. EC.sub.50 values are reported as follows: "A" indicates an EC.sub.50 value of less than 400 nM; "B" indicates an EC.sub.50 value of 400 nM to 2 .mu.M; "C" indicates an EC.sub.50 value of greater than 2 .mu.M and less than 10 .mu.M for each enzyme; "D" indicates an EC.sub.50 value of greater than 10 .mu.M for each enzyme; and "*(X .mu.M)" indicates that no inhibition was observed at the highest concentration (i.e., X .mu.M) of compound tested.
TABLE-US-00026 TABLE 3 Ec50 Values for Selected Compounds of the Invention In Hela Cells Expressing H3k27 Mutant EZH2. H3K27me3 H3K27me3 _Alpha_ _MSD_ Compound HeLa HeLa_ No. (EC50) (EC50) 204 B 211 B 212 B 218 A 219 B 224 A A 230 B 240 C 241 B 243 C 253 A 256 B 261 A A 273 A 284 B 288 A B 294 A A 298 A A 300 A A 304 A A 310 A A 313 A A 314 A 315 D 316 B 317 A 321 A 327 A 335 A 336 A 337 A 341 A 342 A 343 B 344 A 345 A 346 A 347 B 352 B 355 A 356 A 357 A 358 B 359 B 360 C 362 A 363 A 364 *(3.33 .mu.M) 365 A 366 B 367 A 368 A 369 A 370 *(3.33 .mu.M) 373 A 374 A 375 A 376 NaN 377 B
Example 18
Tumor Growth Inhibition Analysis
The anti-tumor efficacy of Compound 362 and 365 in the subcutaneous Karpas422 human lymphoma xenograft model in female CB-17 SCID mice was as follows.
Animals
Species: Mus Musculus
Strain: CB-17 SCID mice
Age: 6-8 weeks
Sex: female
Body weight: 18-22 g
Number of animals: 50 mice plus spare
Animal supplier: Shanghai SLAC Laboratory Animal Co., LTD.
Cell Culture
The Karpas422 tumor cells were maintained in vitro as suspension culture in RPMI1640 medium supplemented with 10% heat inactivated fetal calf serum at 37.degree. C. in an atmosphere of 5% CO.sub.2 in air. The tumor cells were routinely subcultured twice weekly. The cells growing in an exponential growth phase were harvested and counted for tumor inoculation.
Tumor Inoculation
Each mouse was inoculated subcutaneously at the right flank with the Karpas422 tumor cells (5.times.10.sup.6) in 0.2 ml of PBS with Matrigel (1:1) for tumor development. Day 23 after tumor inoculation was as day 0 after the start of treatment when the average tumor size reached approximately 300 mm.sup.3. Each group consisted of 10 tumor-bearing mice.
Tumor Measurements
Tumor size was measured three times weekly in two dimensions using a caliper, and the volume was expressed in mm.sup.3 using the formula: V=0.536a.times.b.sup.2 where a and b are the long and short diameters of the tumor, respectively. The tumor size was then used for calculations of T/C values. The T/C value (in percent) is an indication of antitumor effectiveness; T and C are the mean volumes of the treated and control groups, respectively, on a given day. TGI was calculated for each group using the formula: TGI (%)[1-(Ti-T0)/(Vi-V0)].times.100; Ti is the average tumor volume of a treatment group on a given day, T0 is the average tumor volume of the treatment group on the day of treatment start, Vi is the average tumor volume of the vehicle control group on the same day with Ti, and V0 is the average tumor volume of the vehicle group on the day of treatment start.
Experimental Endpoint and Sample Collection
1). Plasma, tumor and muscle in EPZ-6438 group were collected on day 16 after the start of treatment at 6 h after dosing. Plasma, tumor and muscle in vehicle, CPI-524369, CPI-524416 and CPI-591780 groups were collected on day 25 after the start of treatment at 1 h after dosing. 2). All the blood was taken from each animal with EDTA-K2 as anticoagulant. Plasma was divided into two parts. The first part was for PK analysis; the second part was frozen for backup. 3). Tumor was divided into three parts. The first part was snap-frozen for PK; the second part was snap-frozen for PD analysis; the third part was frozen for backup. 4). Muscle was divided into two parts. The first part was snap-frozen for PK; the second part was frozen for backup.
Tumor Growth Inhibition Analysis
TABLE-US-00027 TABLE 4 Tumor growth inhibition calculation for Compounds 362 and 365 in the karpass422 xenograft model calculated based on tumor volume measurements on day 25 or day 16 after the start of treatment Tumor Size (mm3).sup.a T/C.sup.b TGI Treatment on day 25 (%) (%) Significance.sup.d Vehicle 1704 .+-. 123 -- -- -- Compound 362 385 .+-. 66 22.59 92.75 *** (160 mg/kg) Compound 365 319 .+-. 67 18.72 97.25 *** (160 mg/kg) Note: .sup.aMean .+-. SEM. .sup.bTumor Growth Inhibition is calculated by dividing the group average tumor volume for the treated group by the group average tumor volume for the control group (T/C). For a test article to be considered to have anti-tumor activity, T/C must be 0.5 or less. .sup.dStatistically significant difference (one-way ANOVA), vs vehicle: *** p < 0.001.
* * * * *
References
C00001

C00002

C00003

C00004

C00005

C00006

C00007

C00008

C00009

C00010

C00011

C00012

C00013
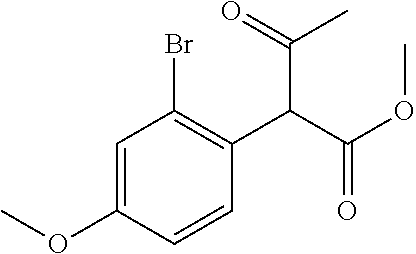
C00014

C00015
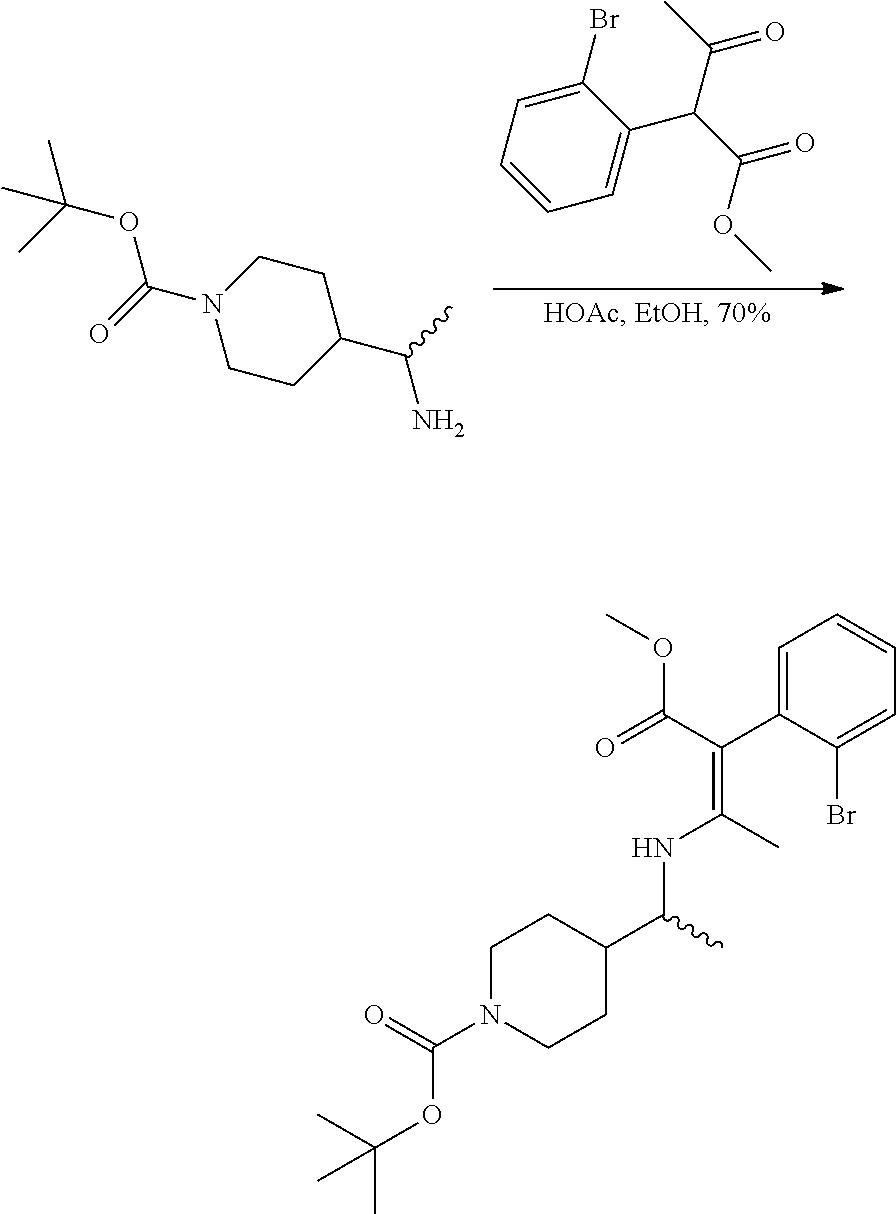
C00016
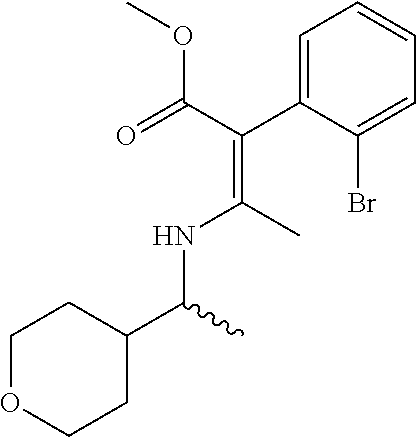
C00017

C00018

C00019

C00020

C00021
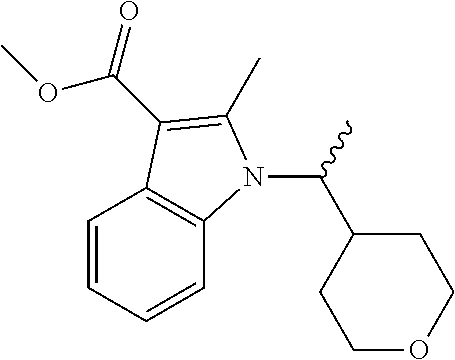
C00022

C00023

C00024

C00025
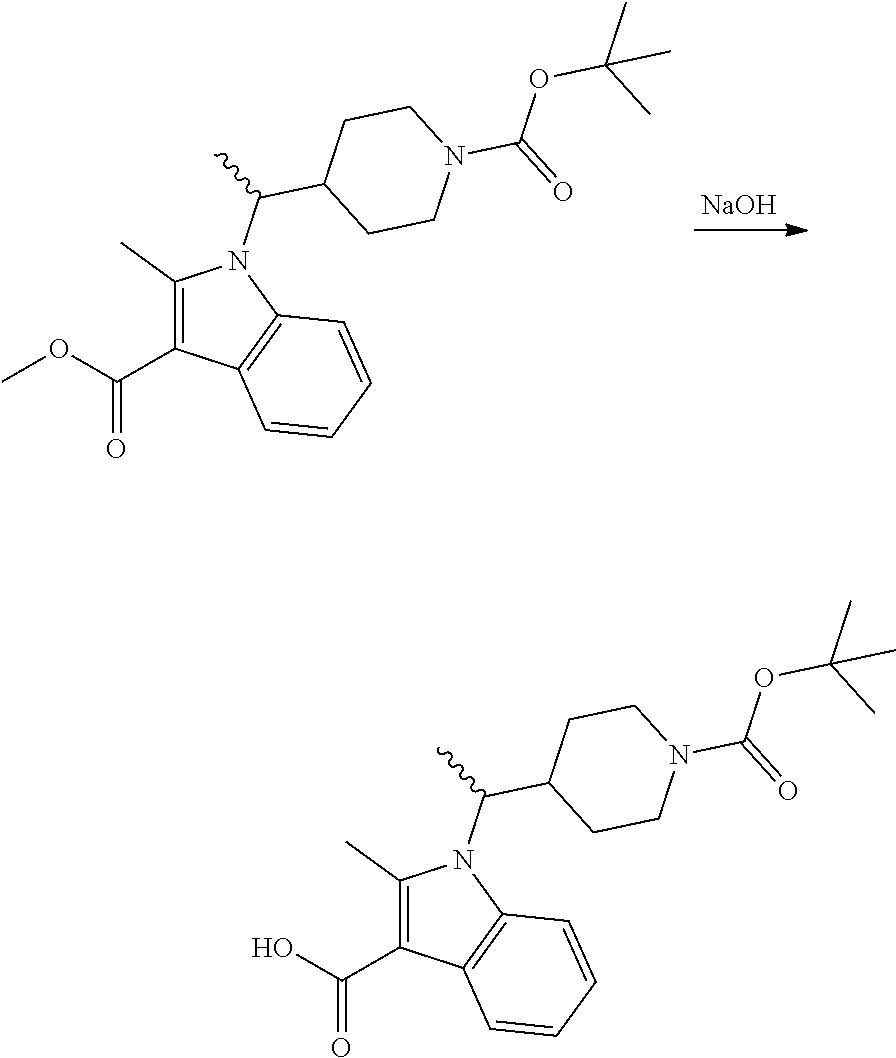
C00026

C00027

C00028

C00029

C00030

C00031
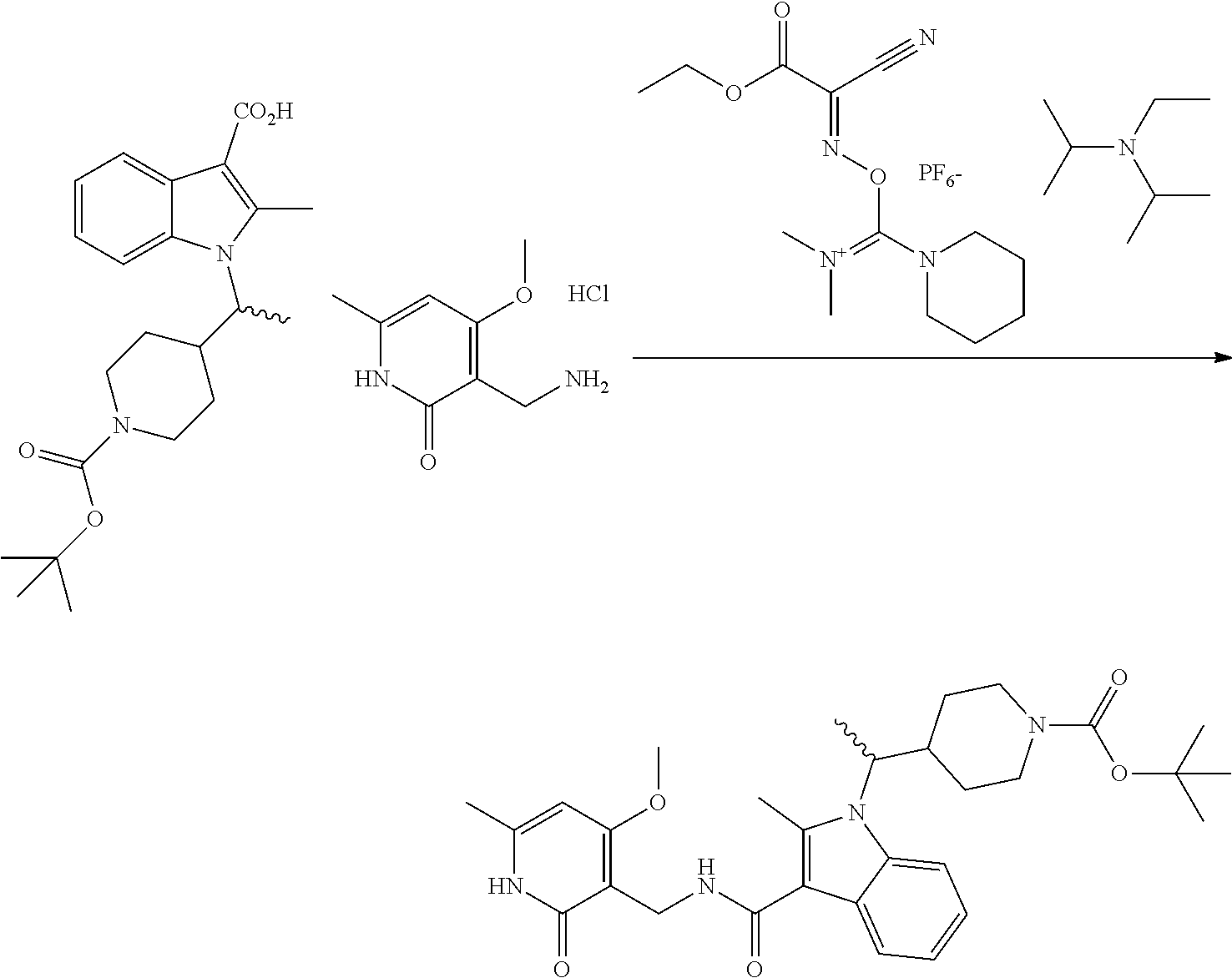
C00032
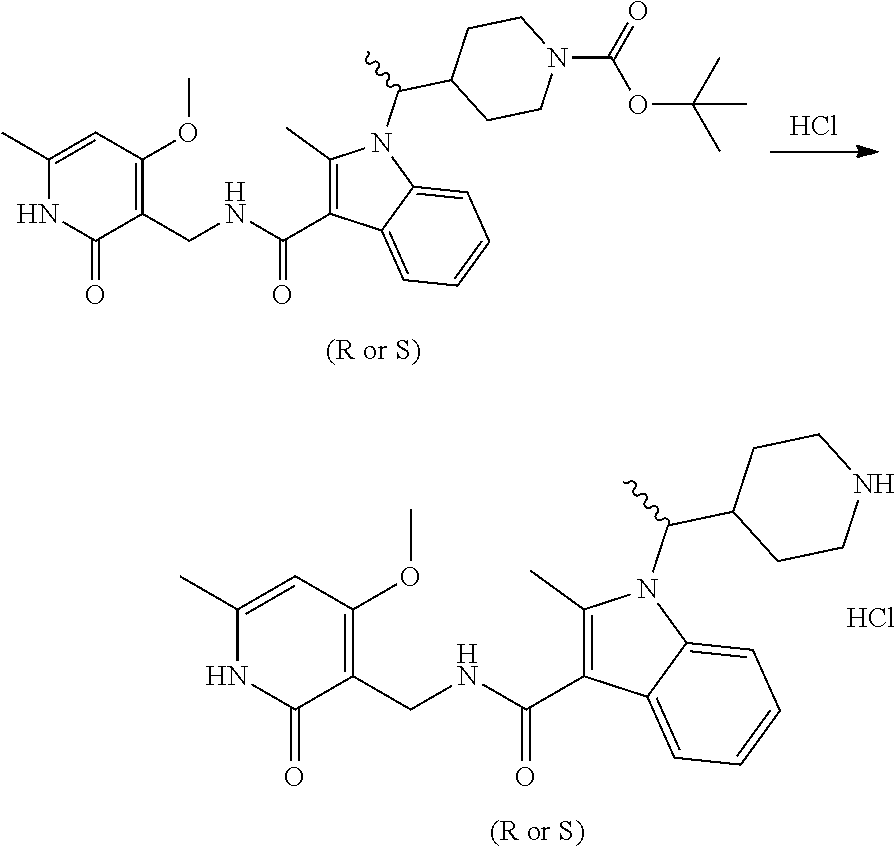
C00033

C00034

C00035
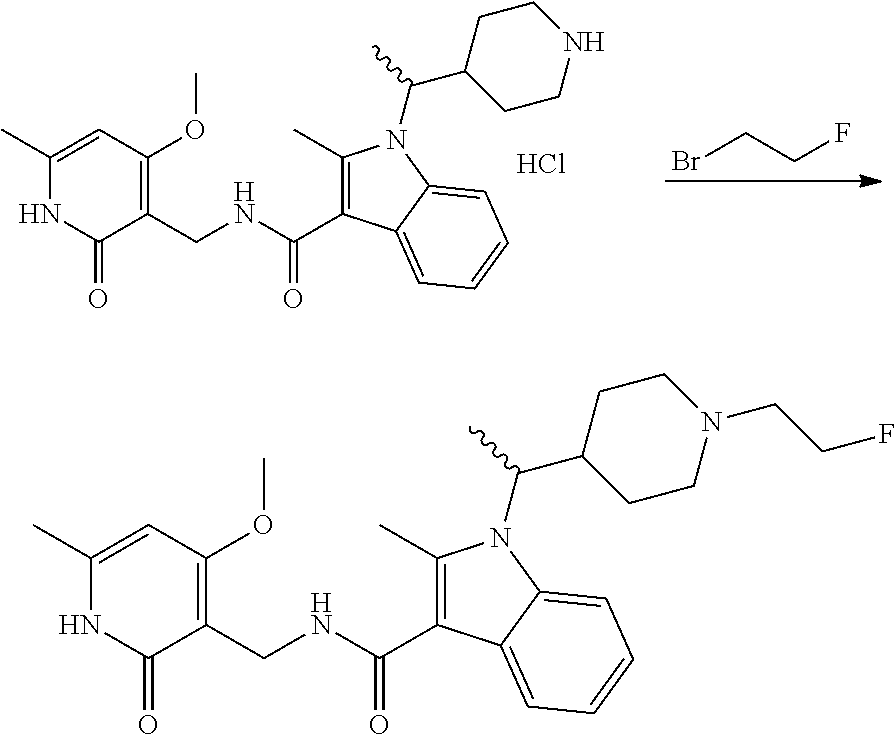
C00036

C00037
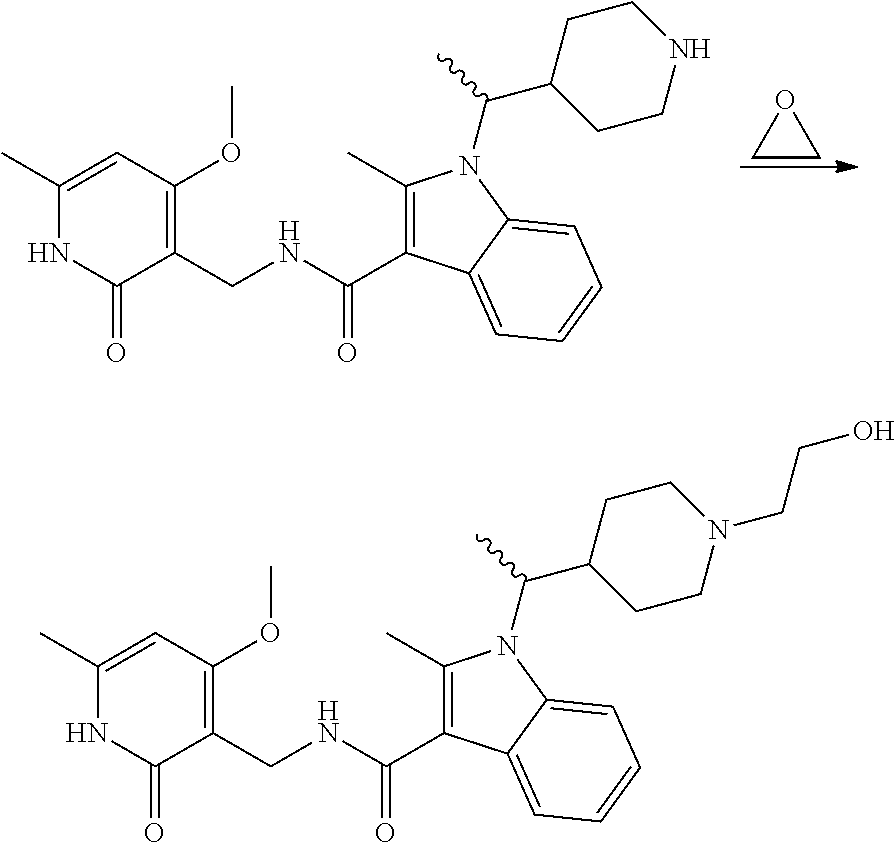
C00038

C00039
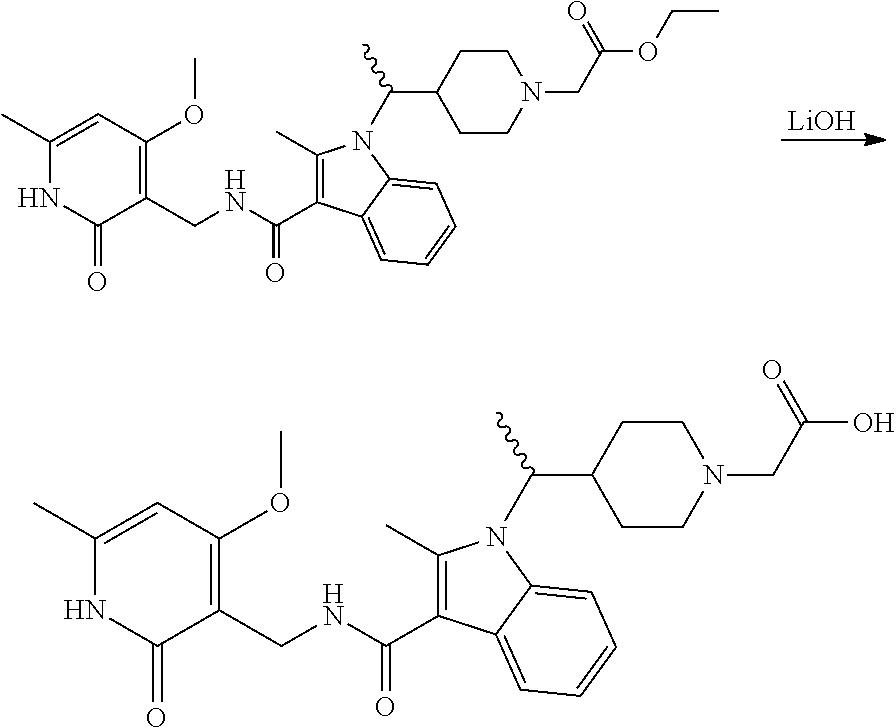
C00040

C00041
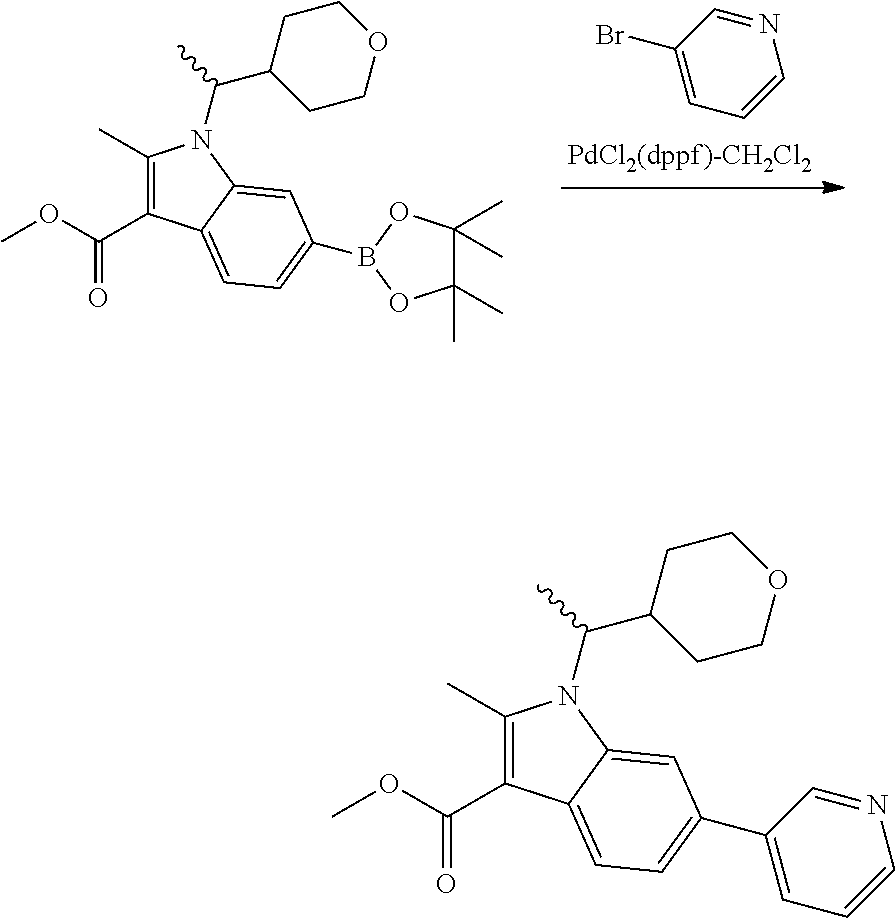
C00042

C00043

C00044

C00045
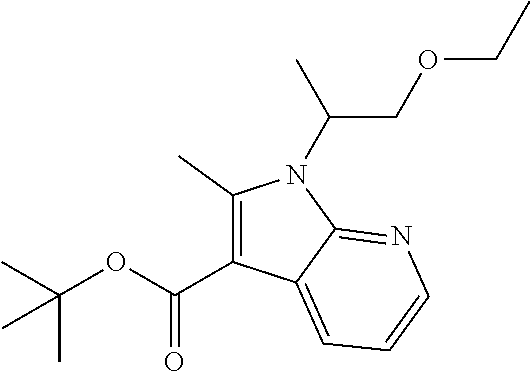
C00046

C00047

C00048

C00049

C00050

C00051
C00052

C00053

C00054
C00055

C00056

C00057
C00058

C00059
C00060
C00061

C00062
C00063
C00064
C00065

C00066

C00067
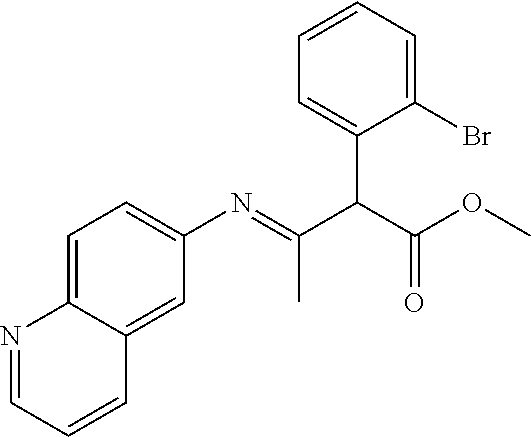
C00068

C00069
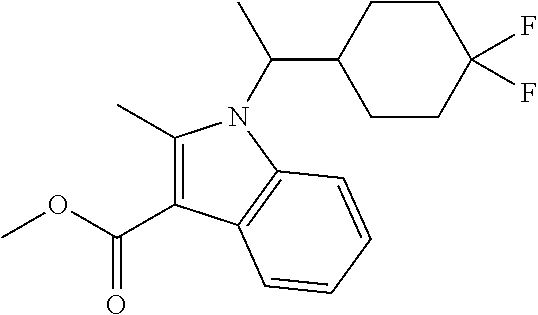
C00070

C00071

C00072

C00073

C00074
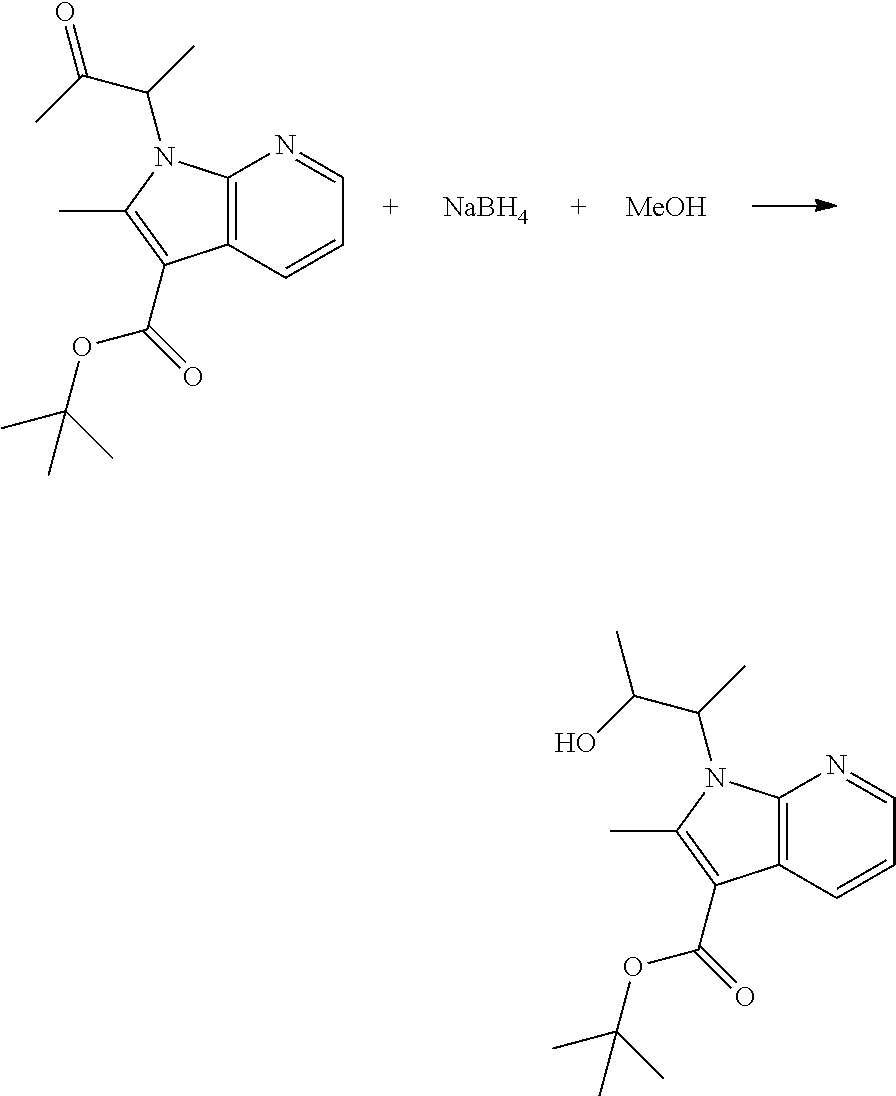
C00075

C00076

C00077

C00078

C00079

C00080

C00081
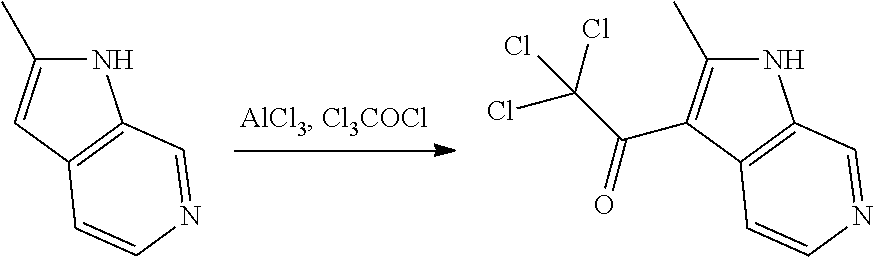
C00082

C00083
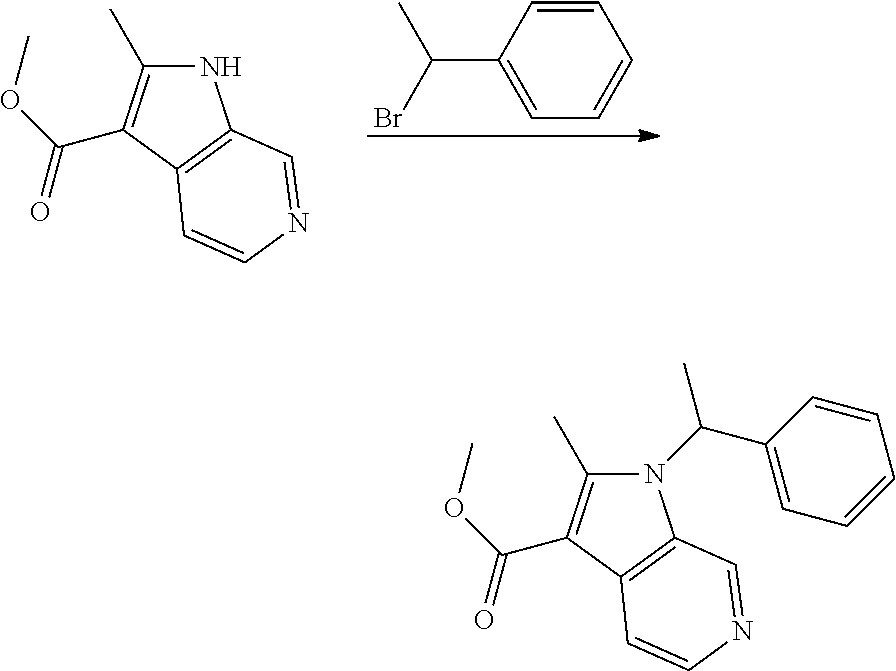
C00084
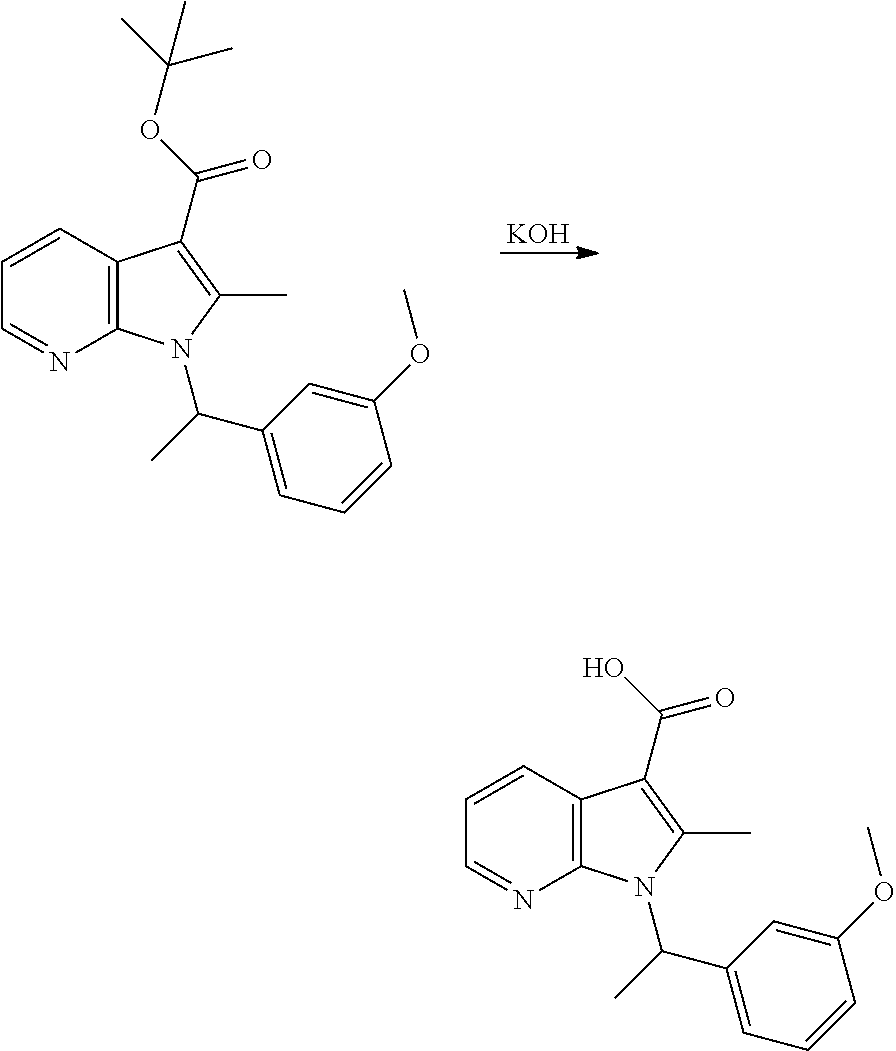
C00085

C00086
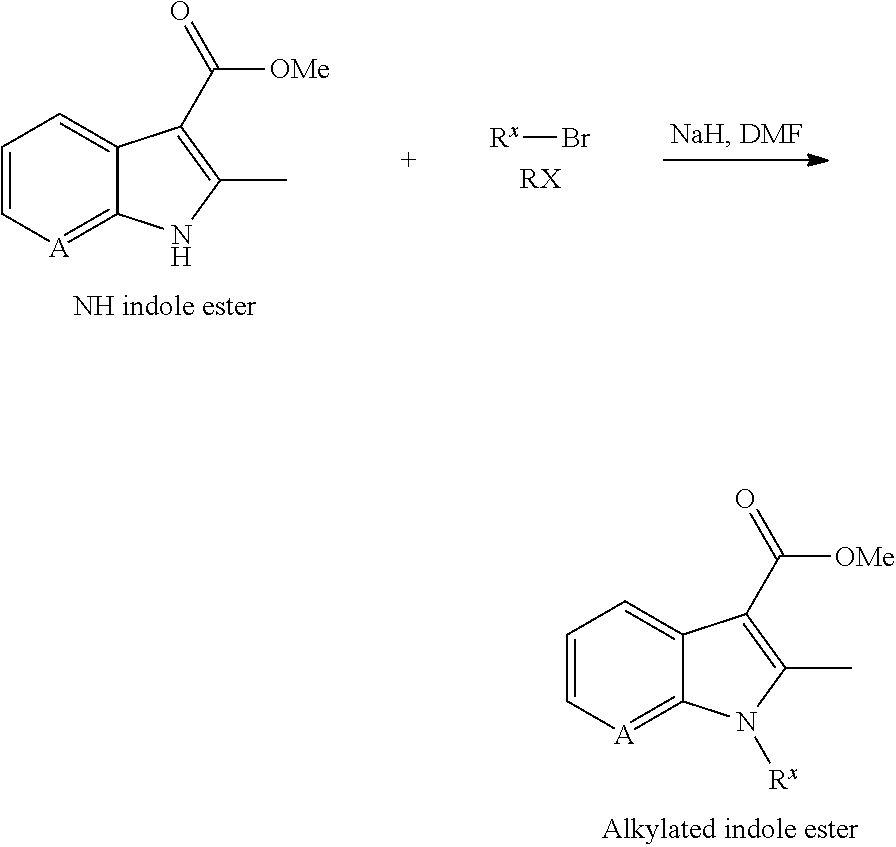
C00087

C00088
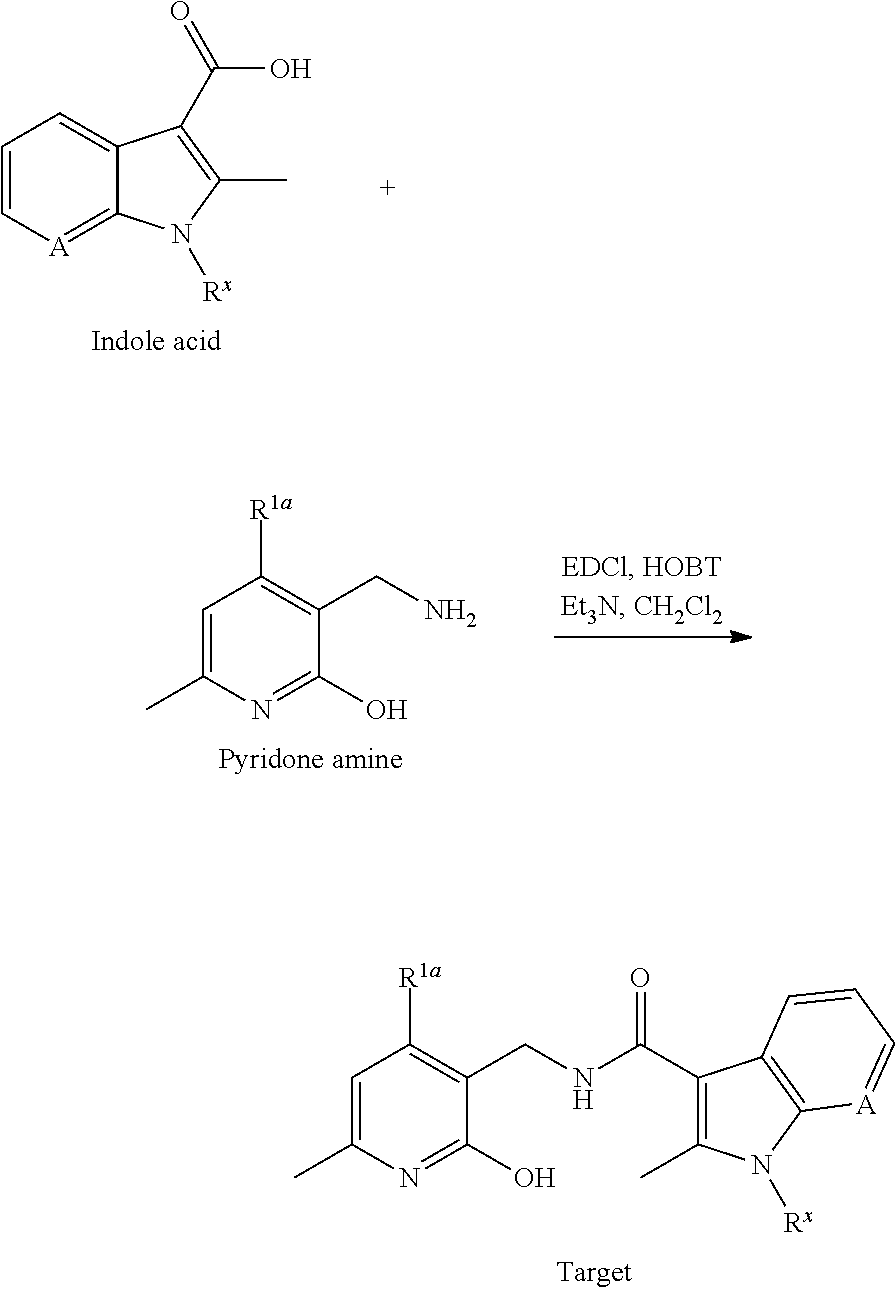
C00089

C00090
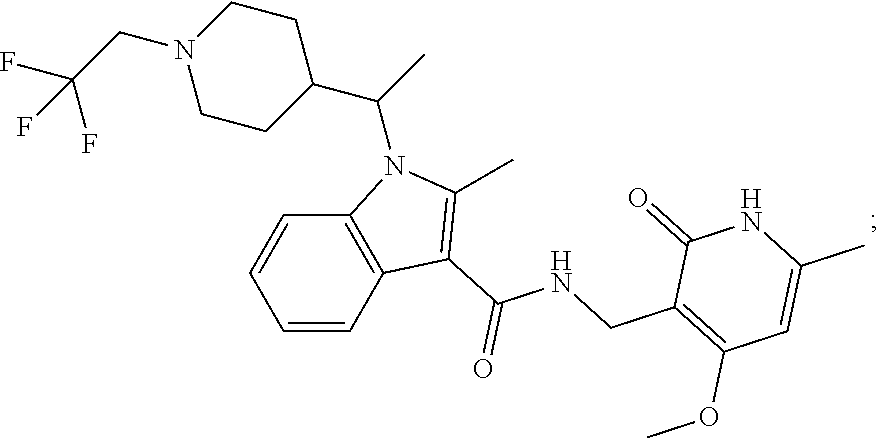
C00091

C00092
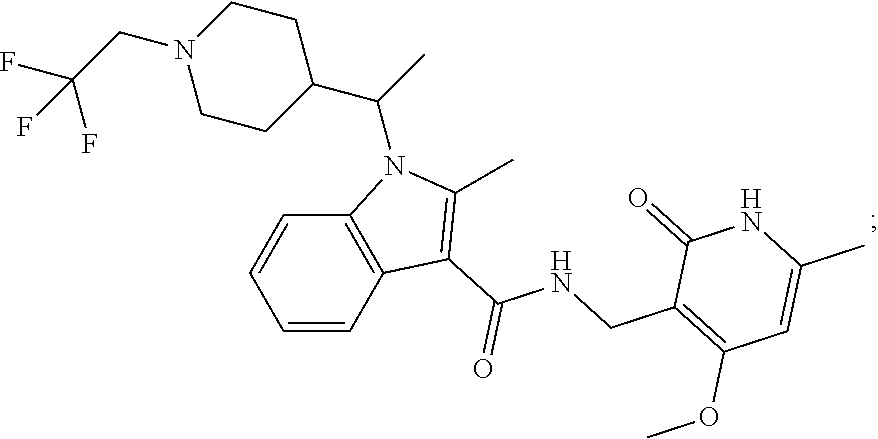
C00093

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008
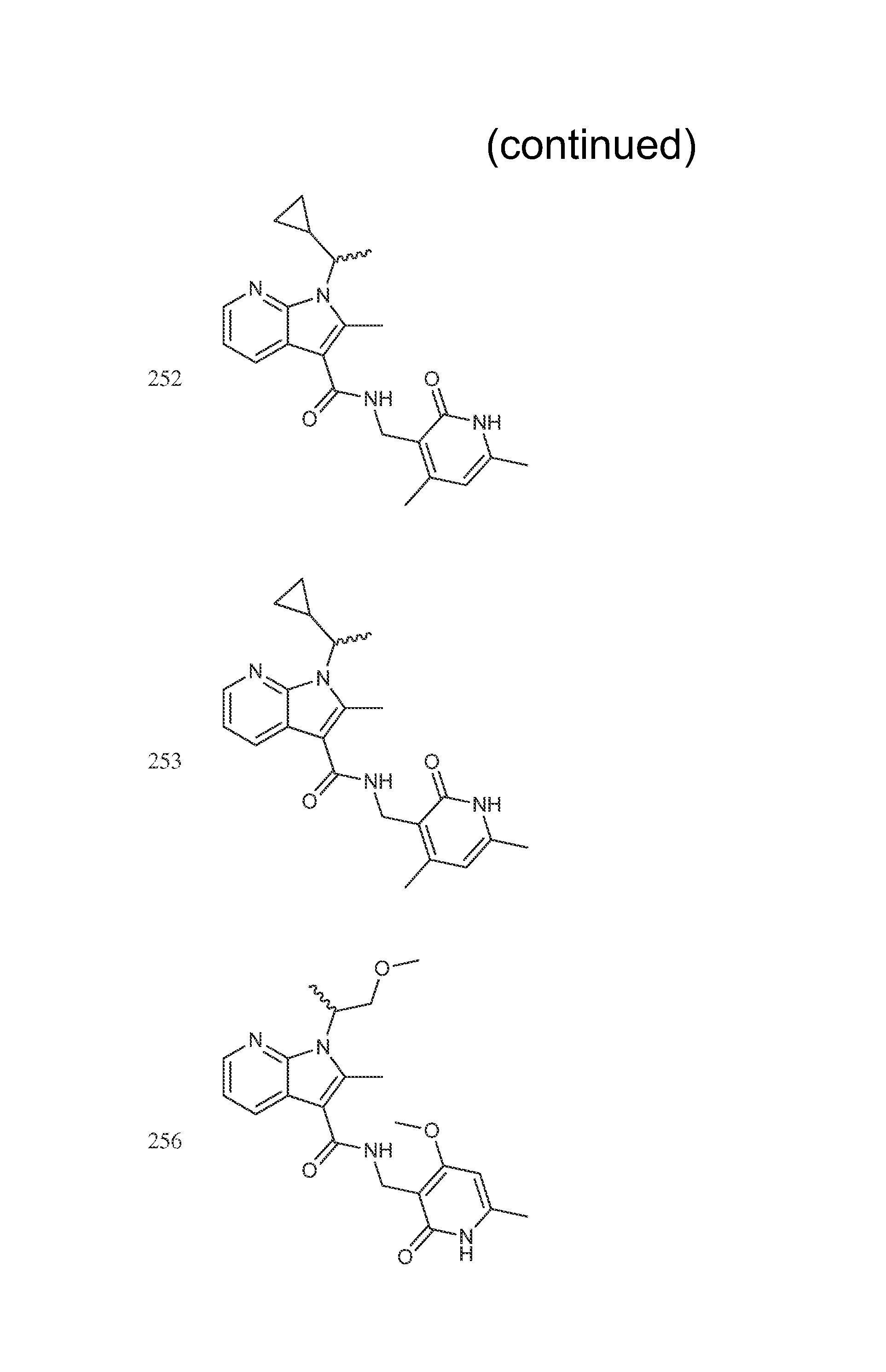
D00009

D00010

D00011

D00012
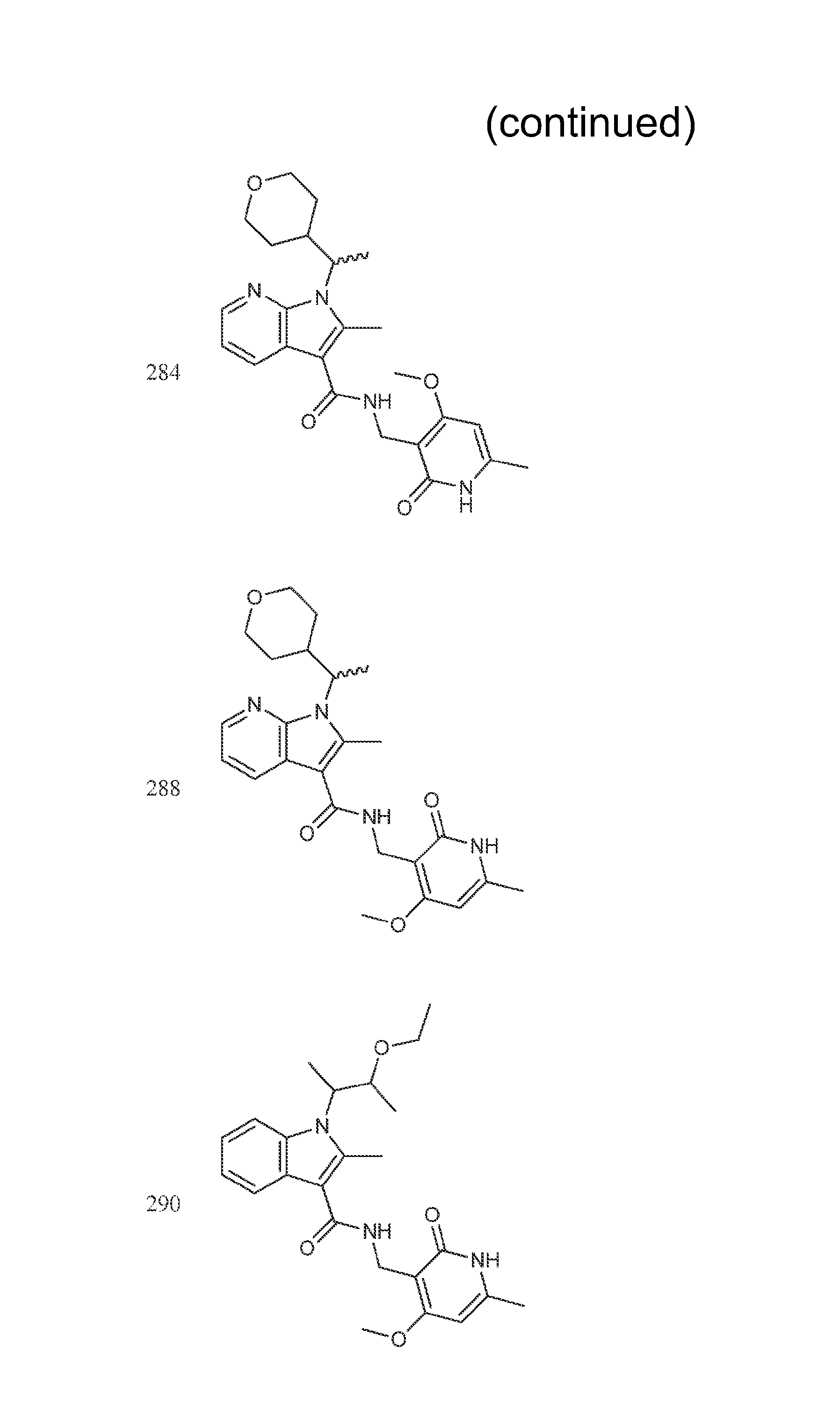
D00013
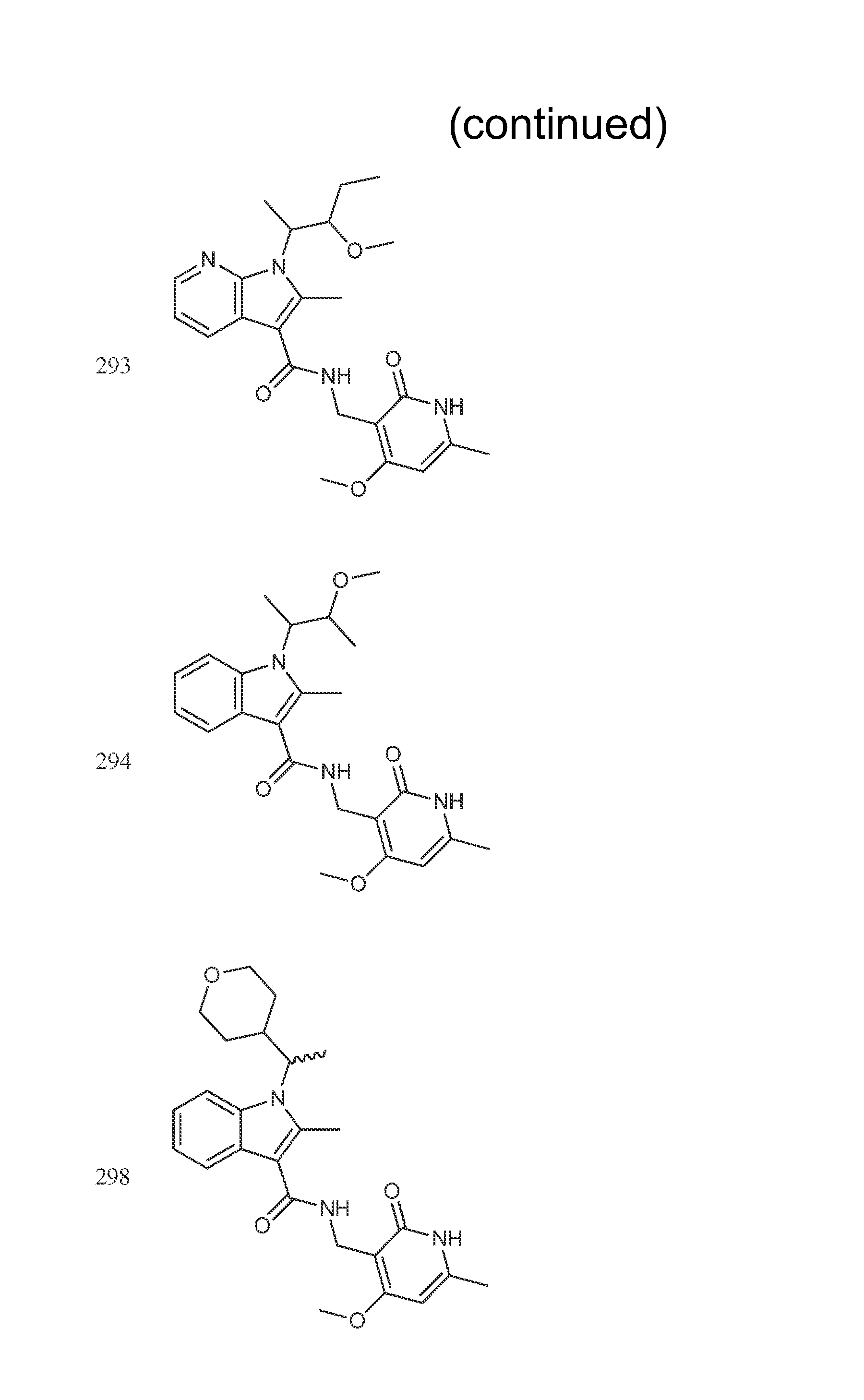
D00014
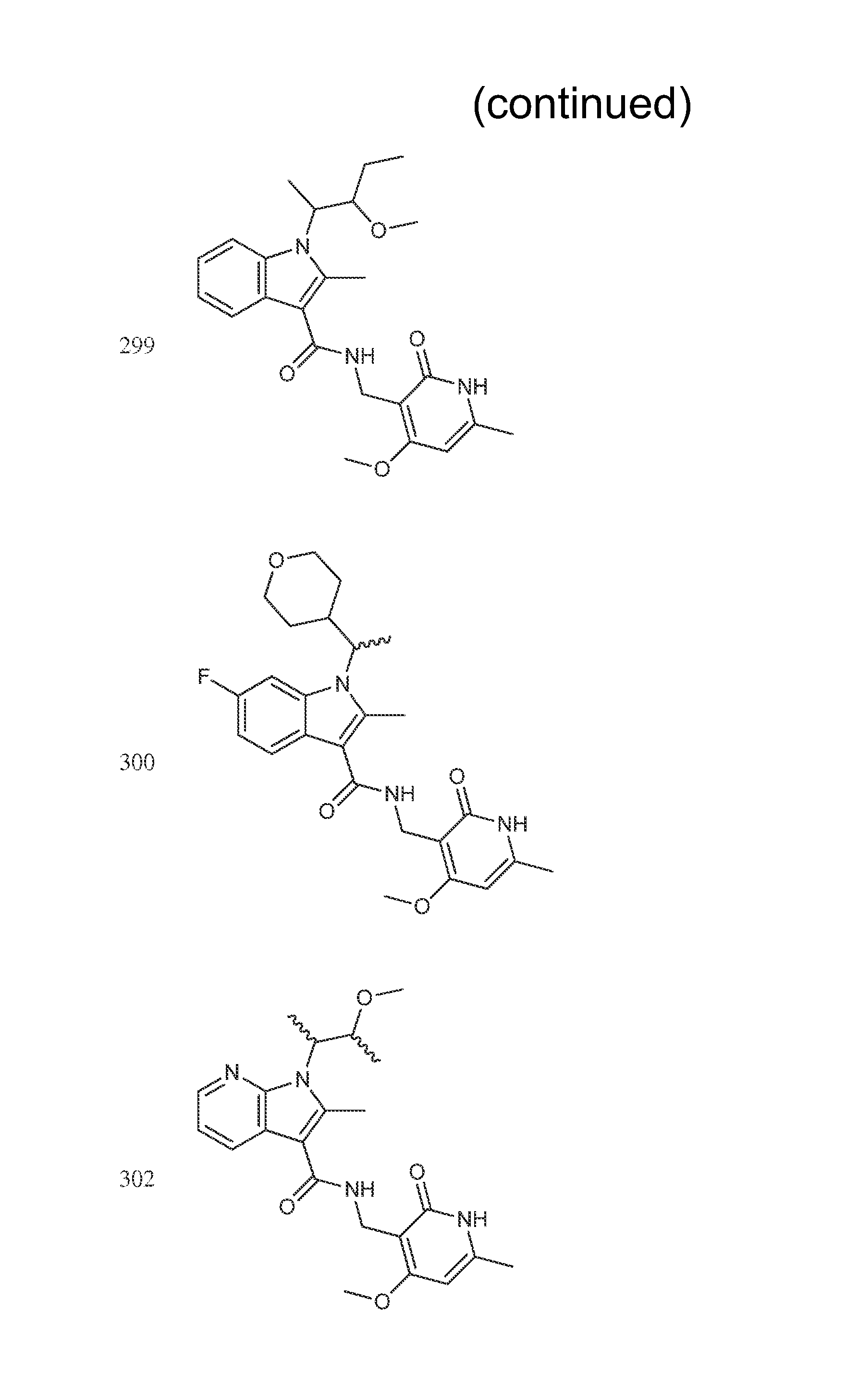
D00015
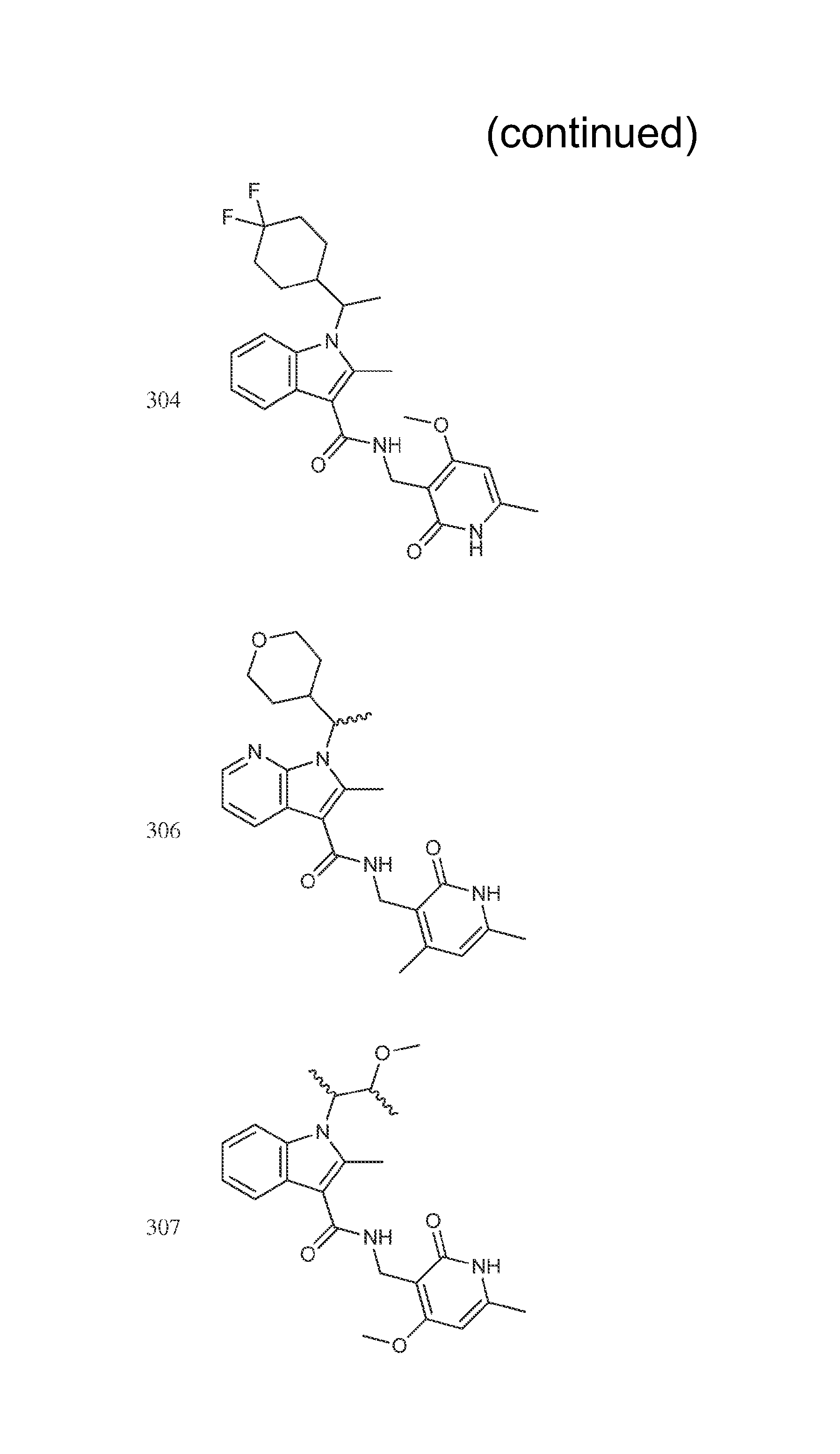
D00016

D00017

D00018
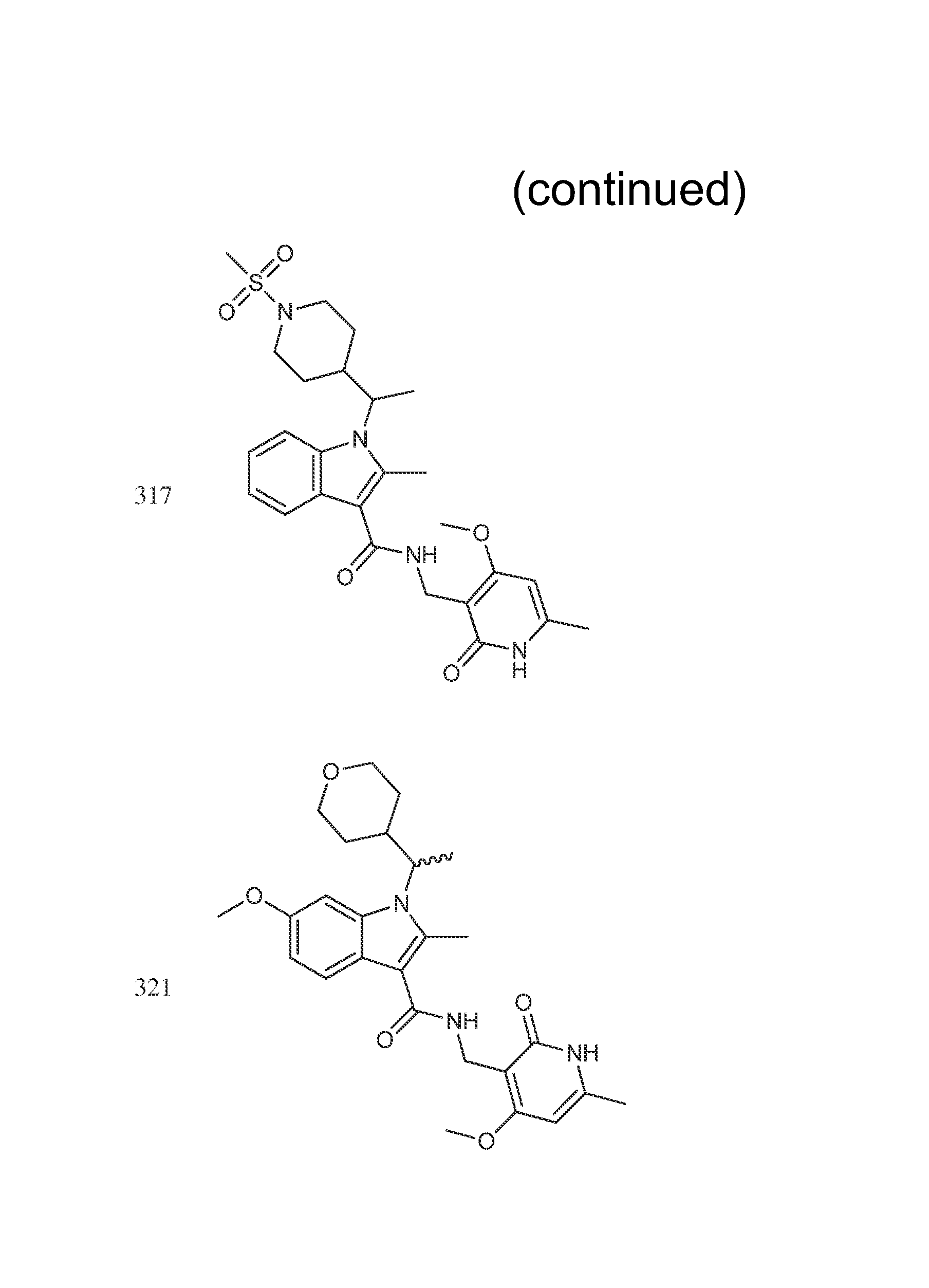
D00019
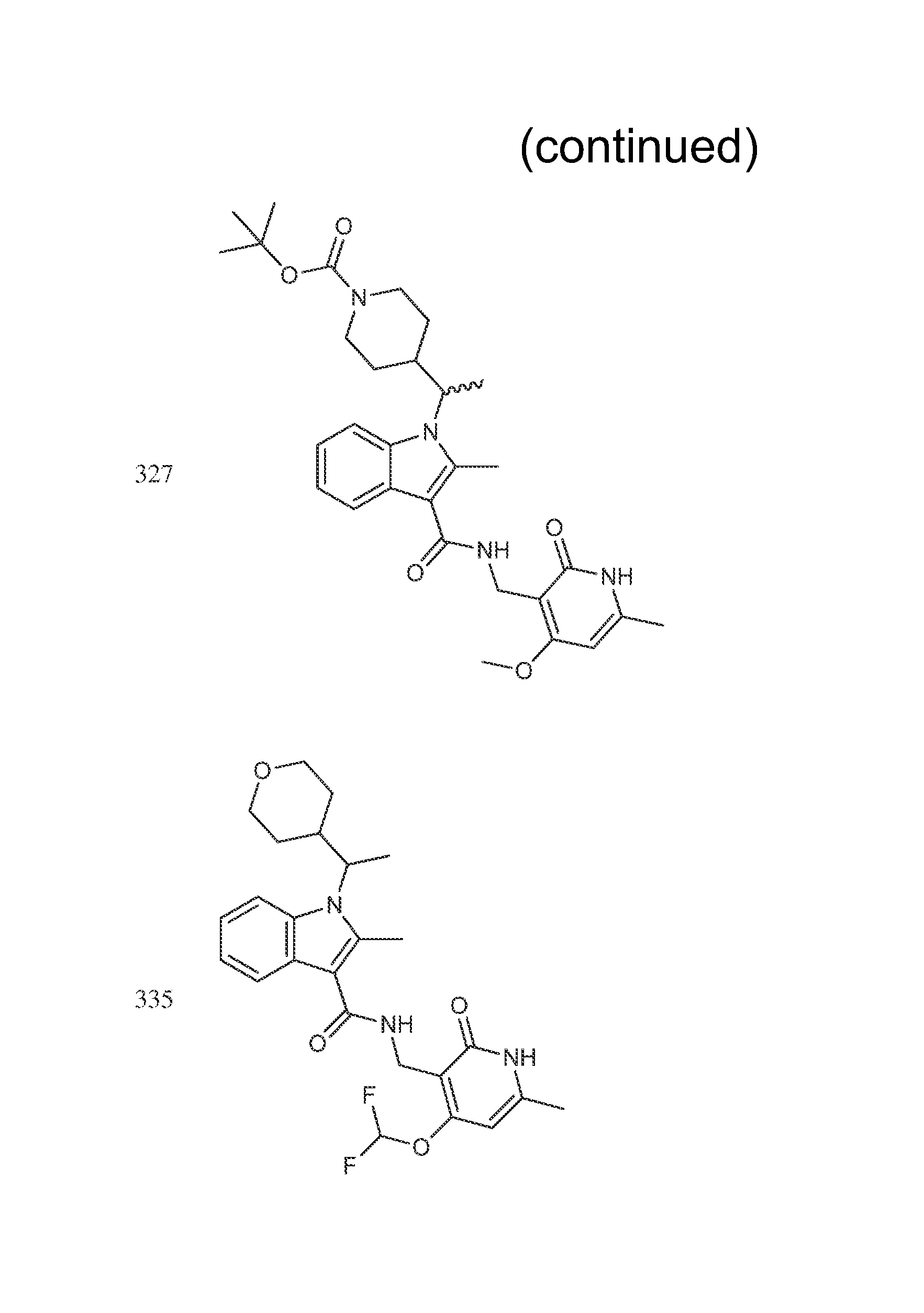
D00020

D00021
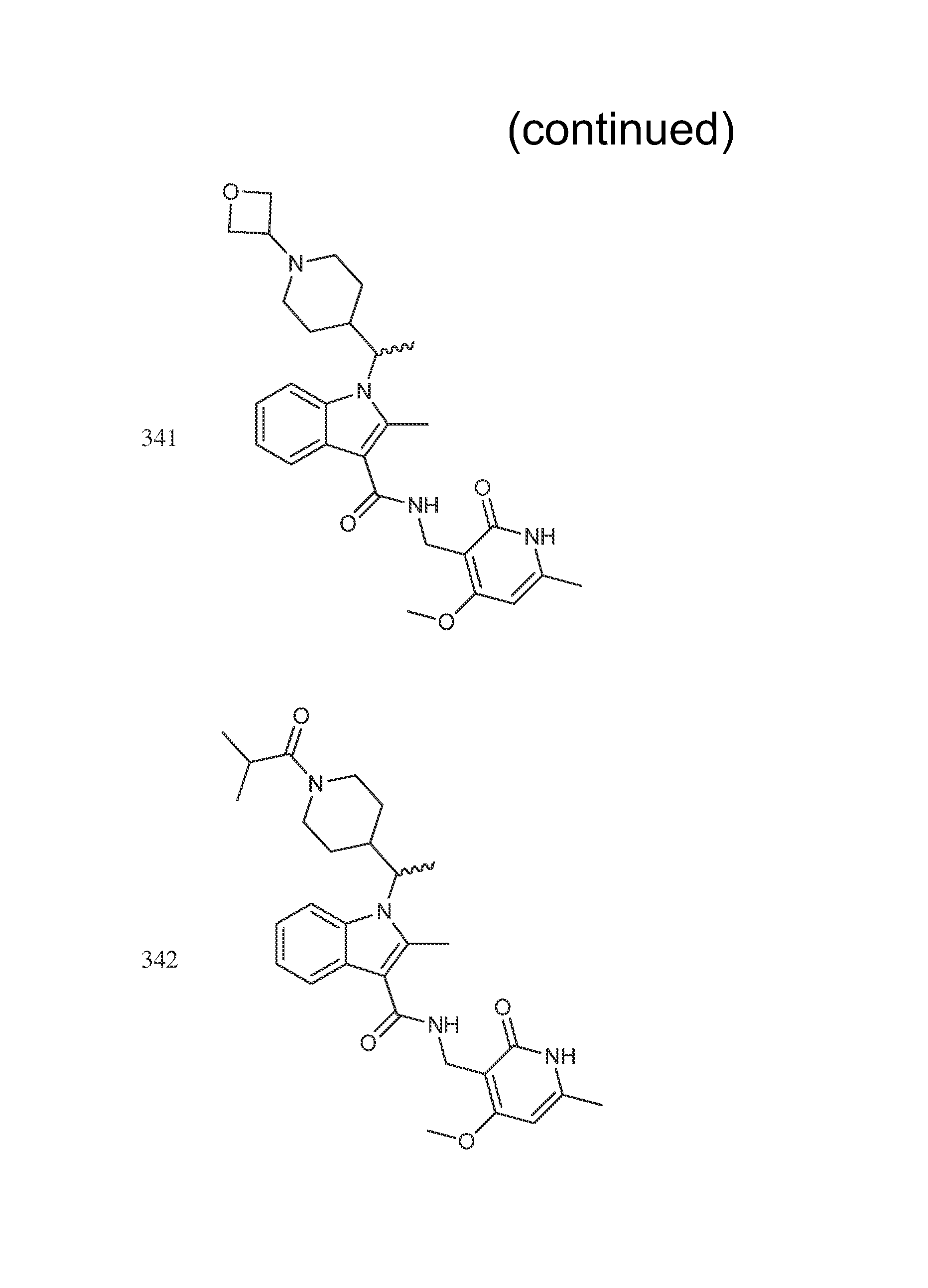
D00022

D00023

D00024
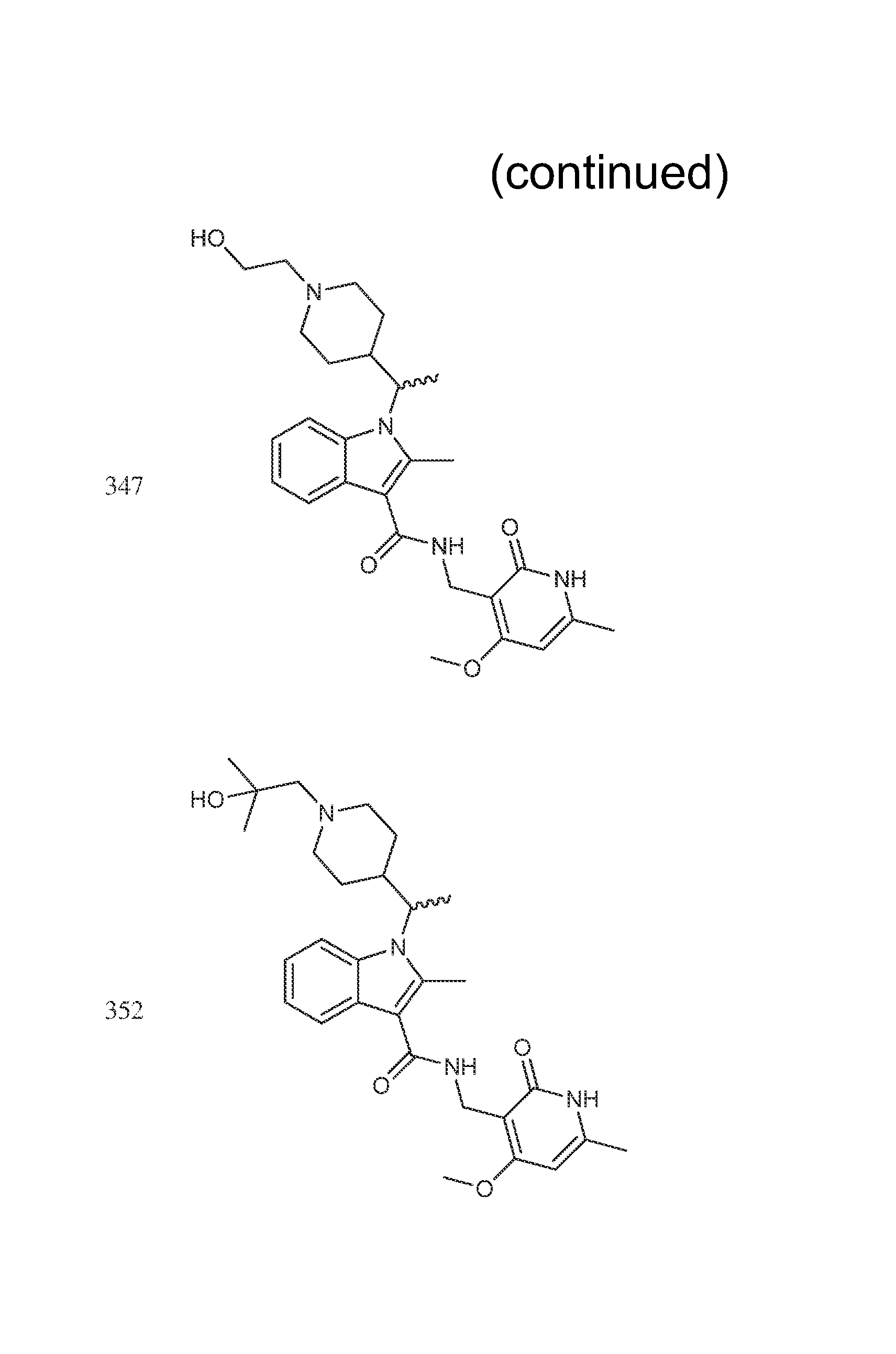
D00025

D00026

D00027
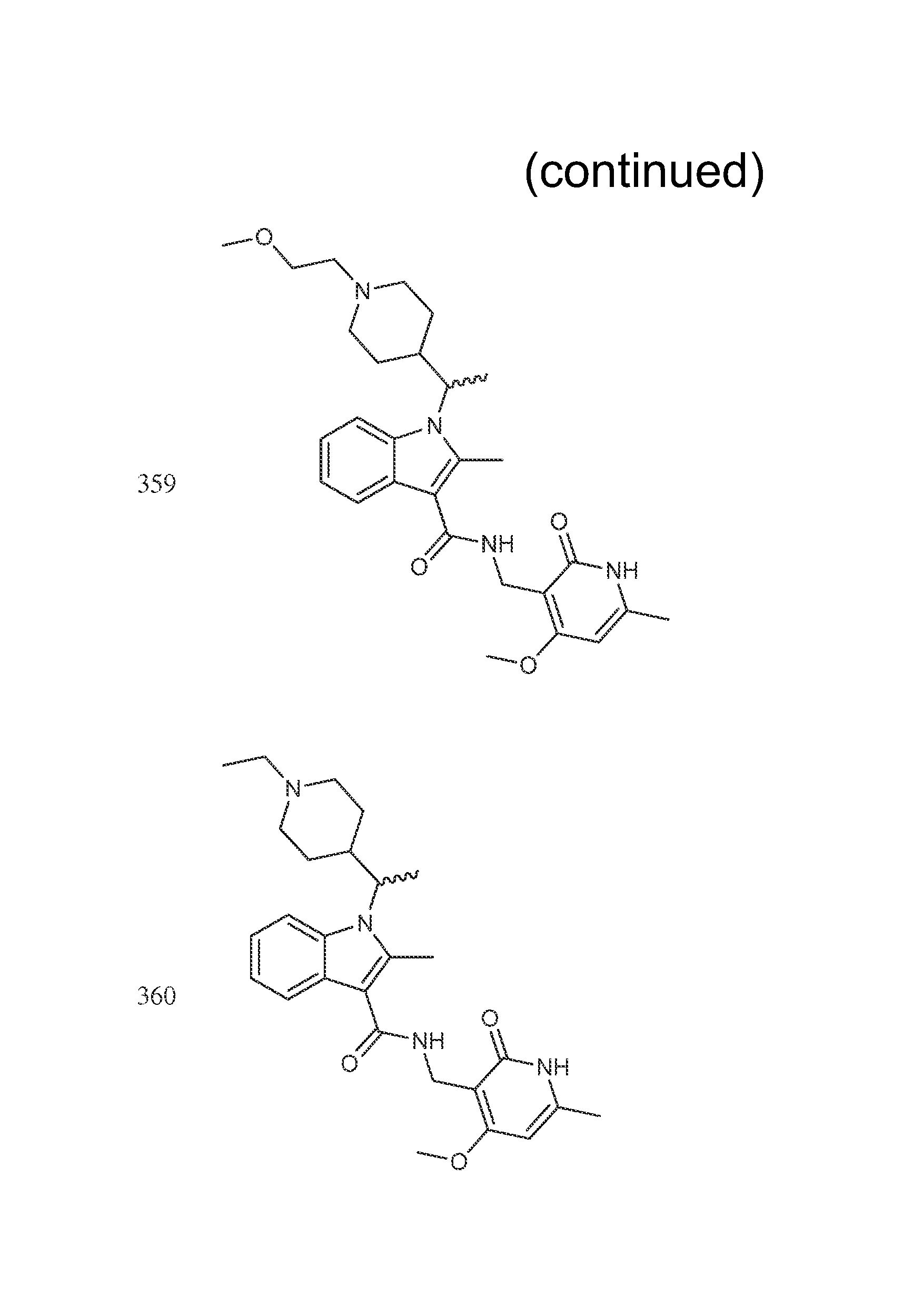
D00028

D00029

D00030

D00031
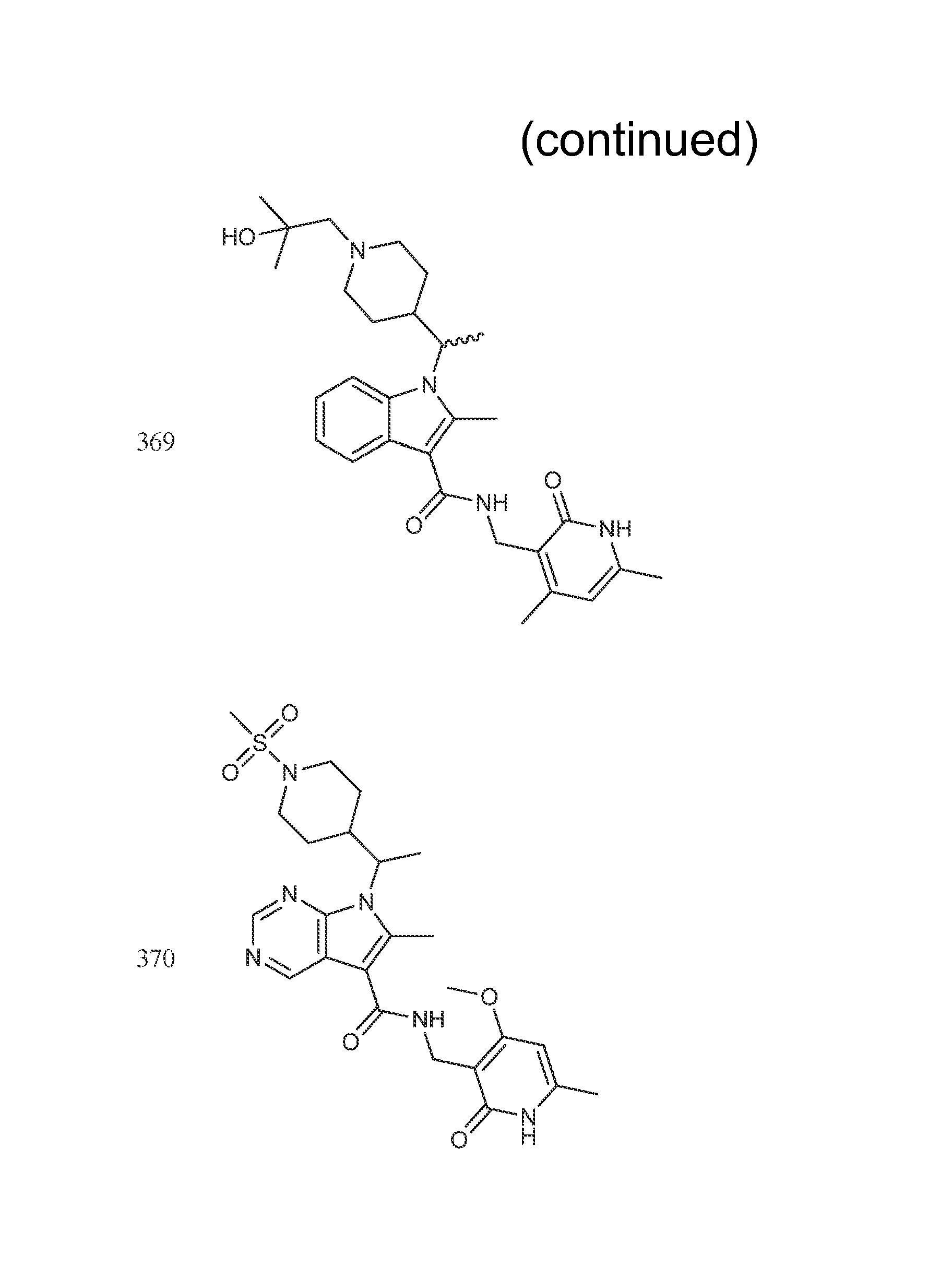
D00032

D00033

D00034
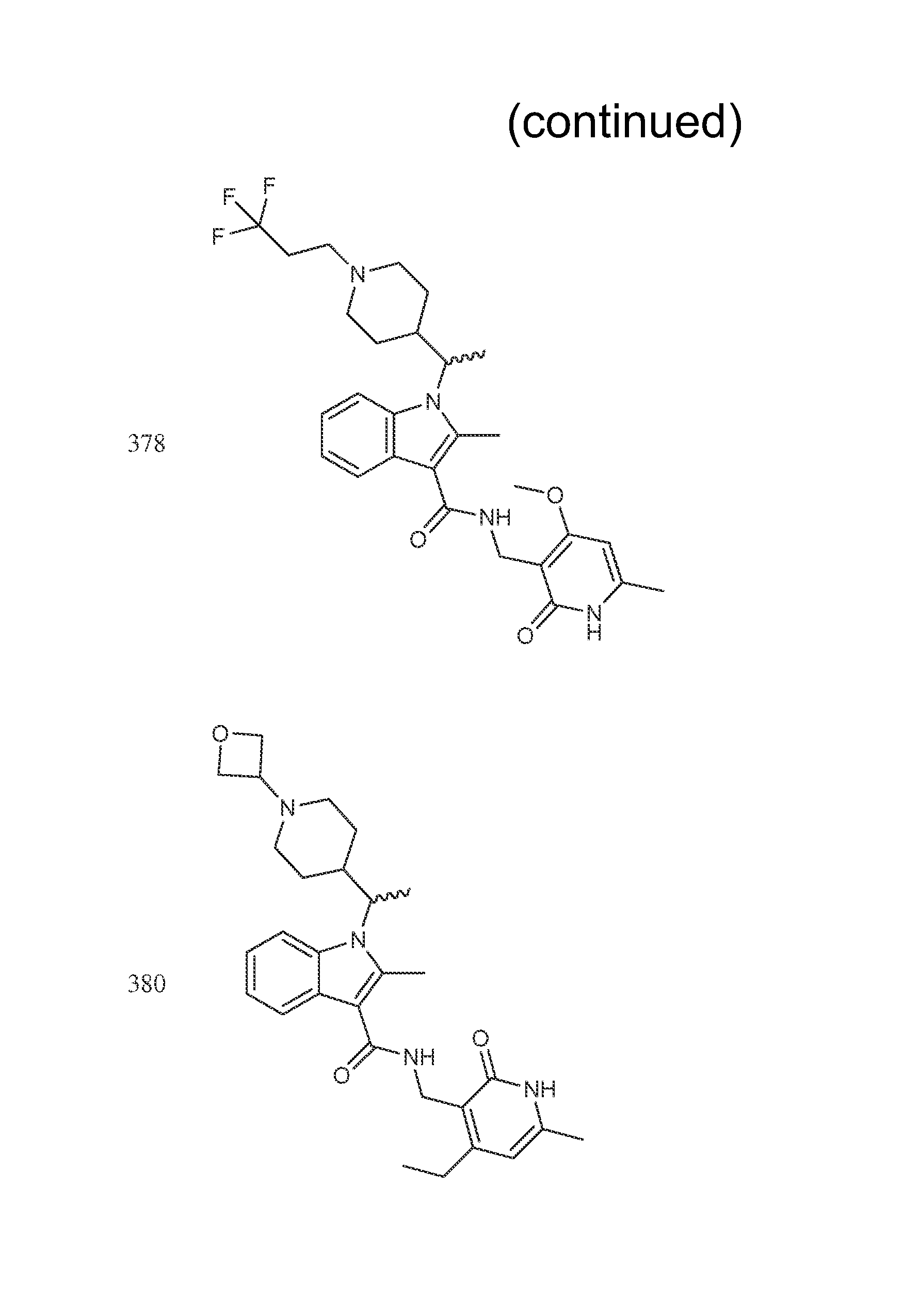
D00035
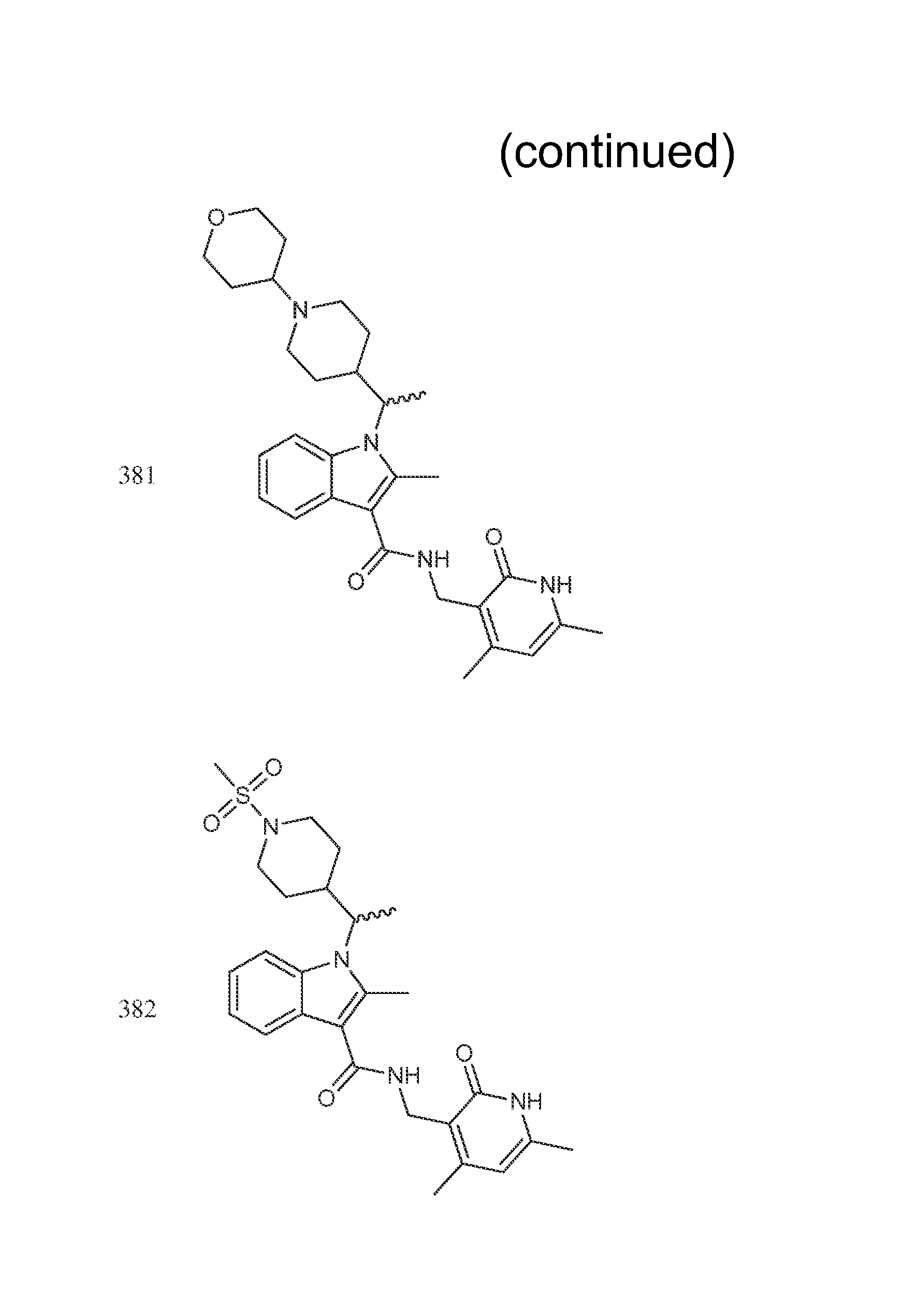
D00036

D00037

D00038
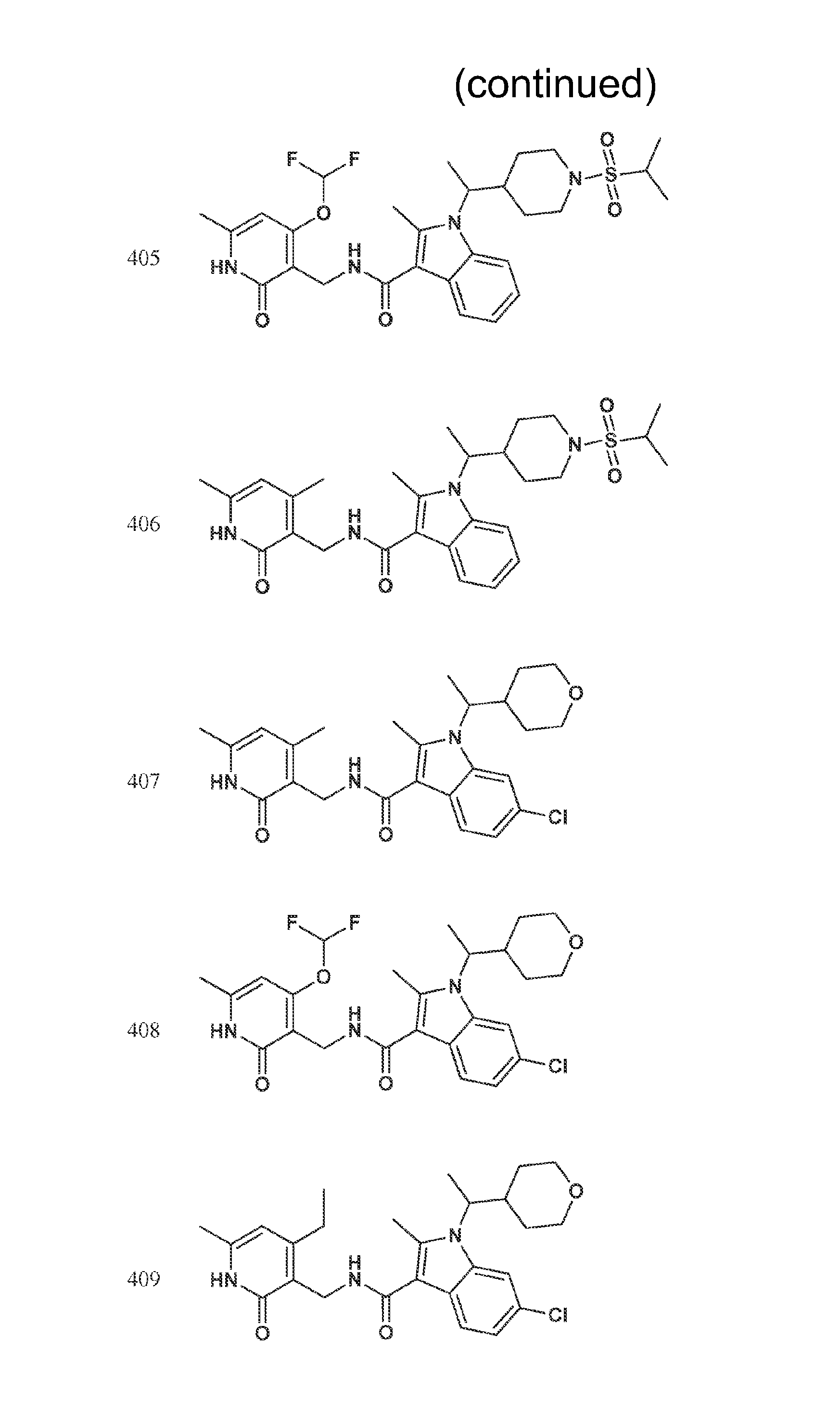
D00039
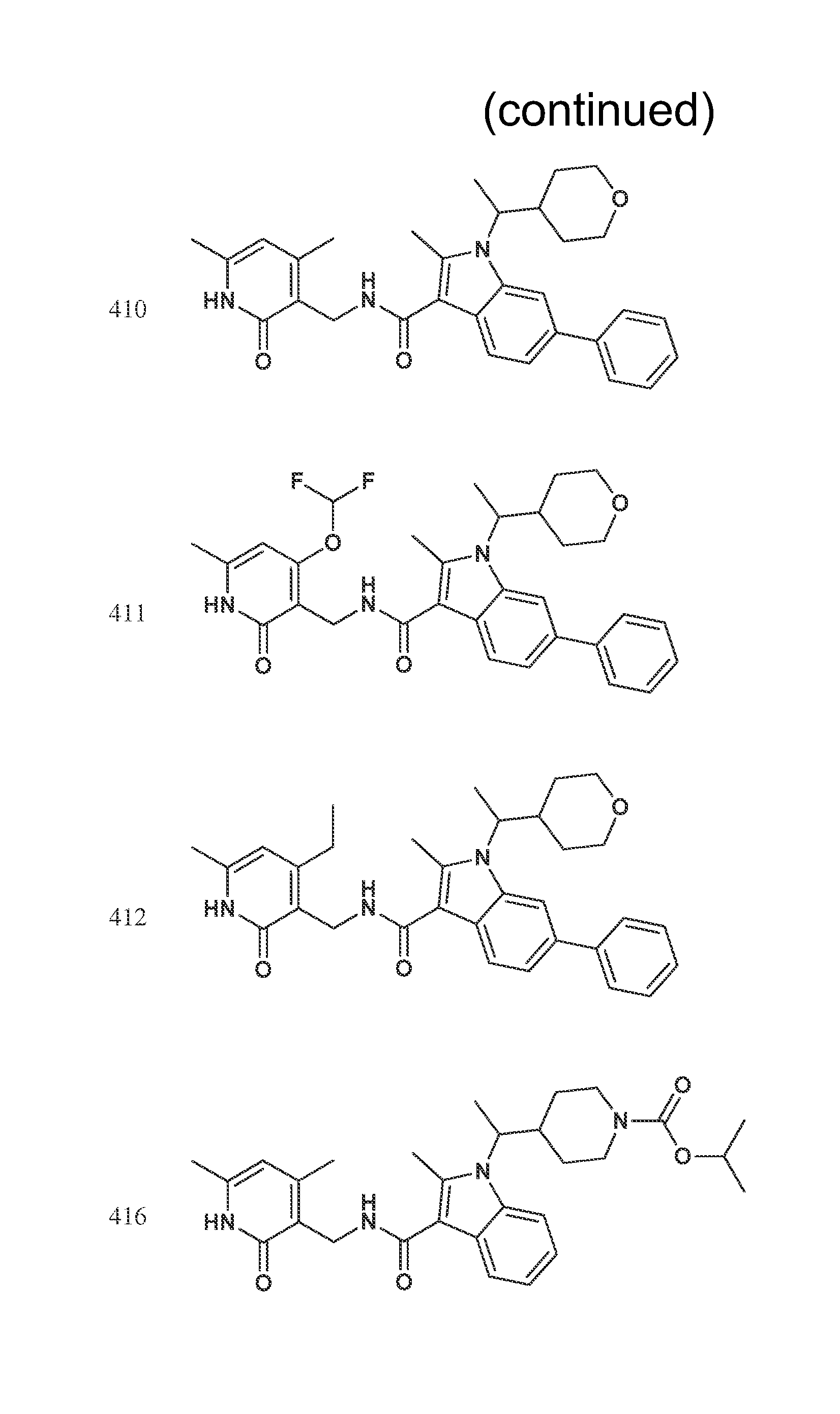
D00040
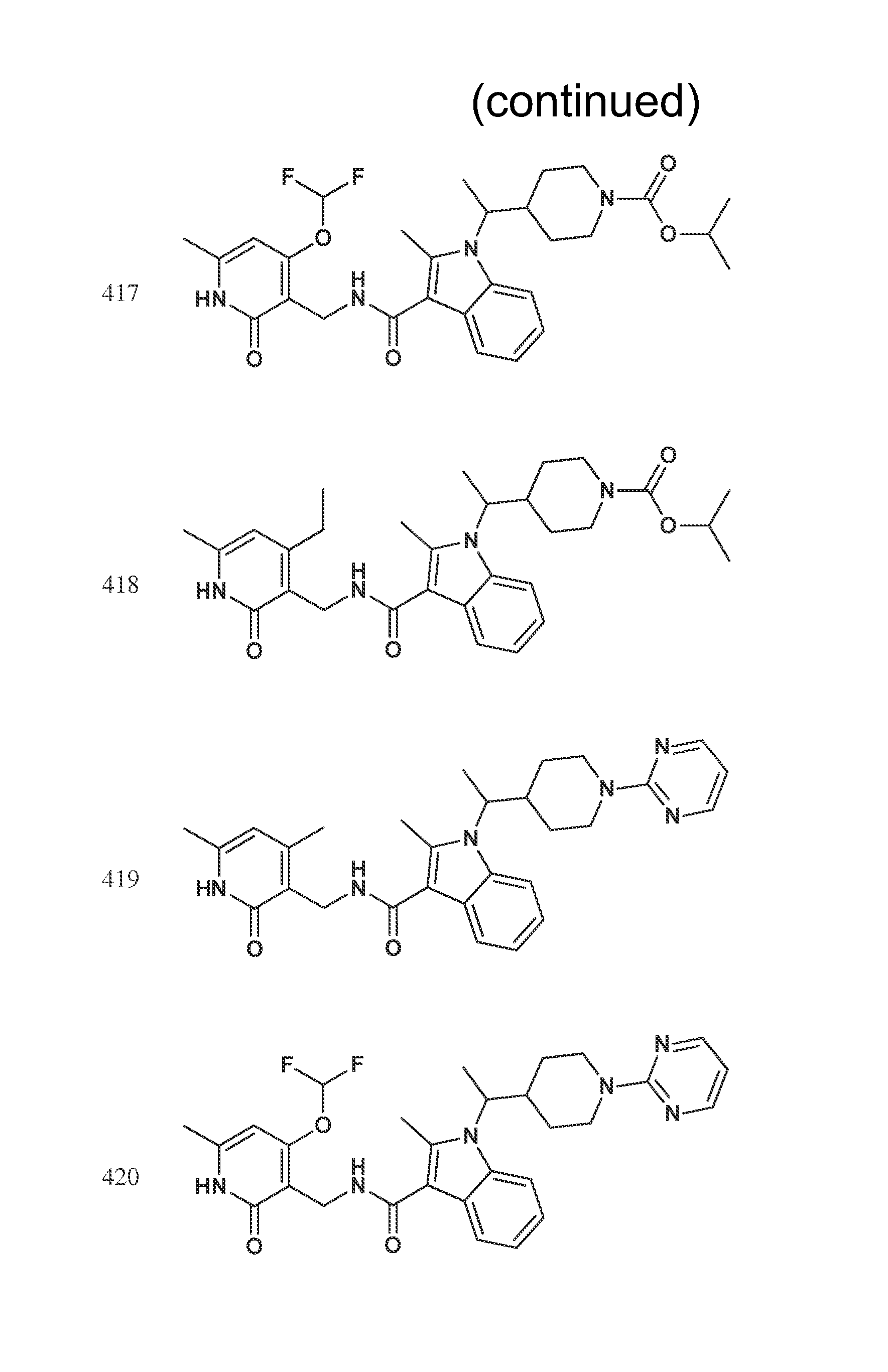
D00041
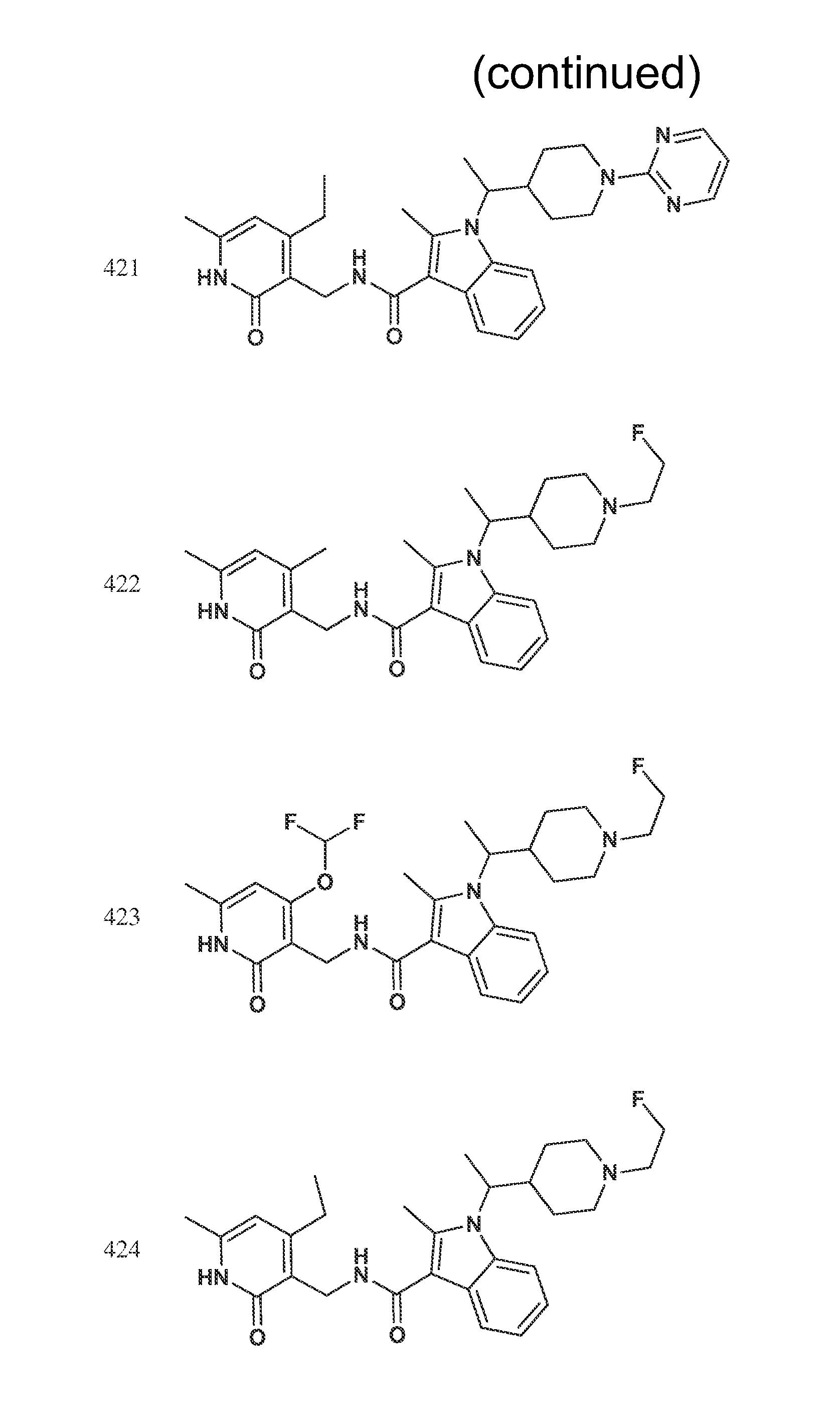
D00042
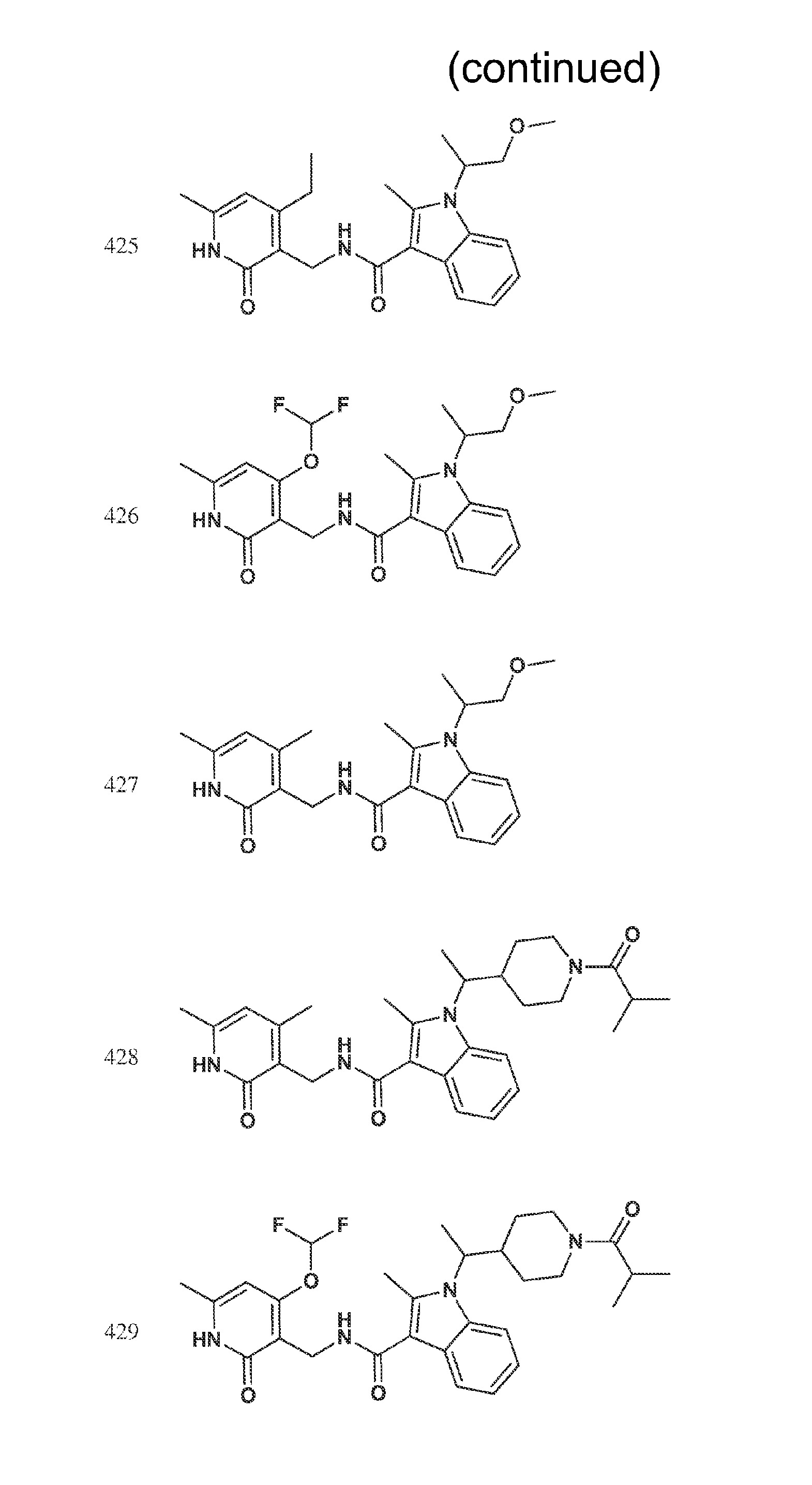
D00043
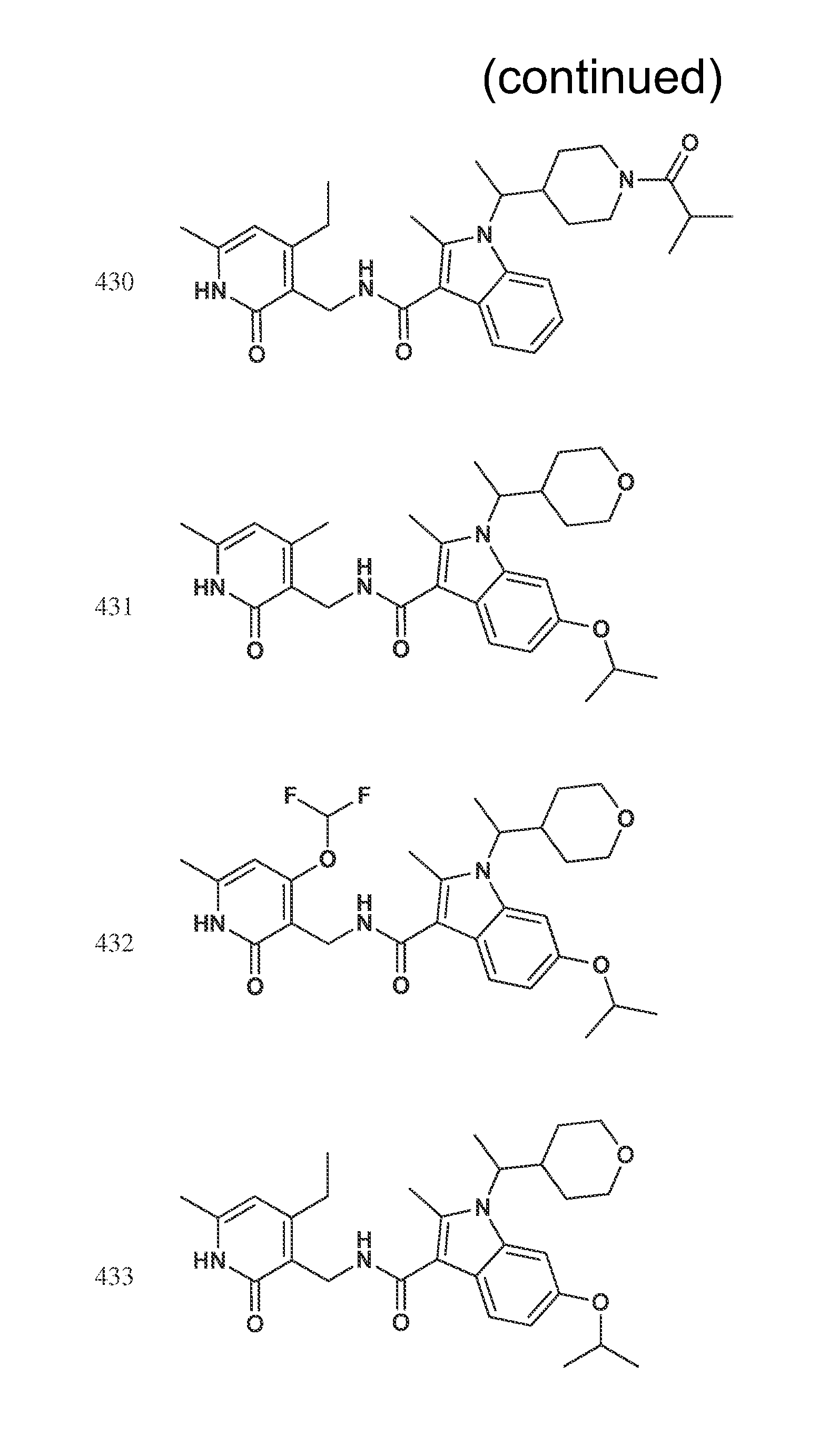
D00044
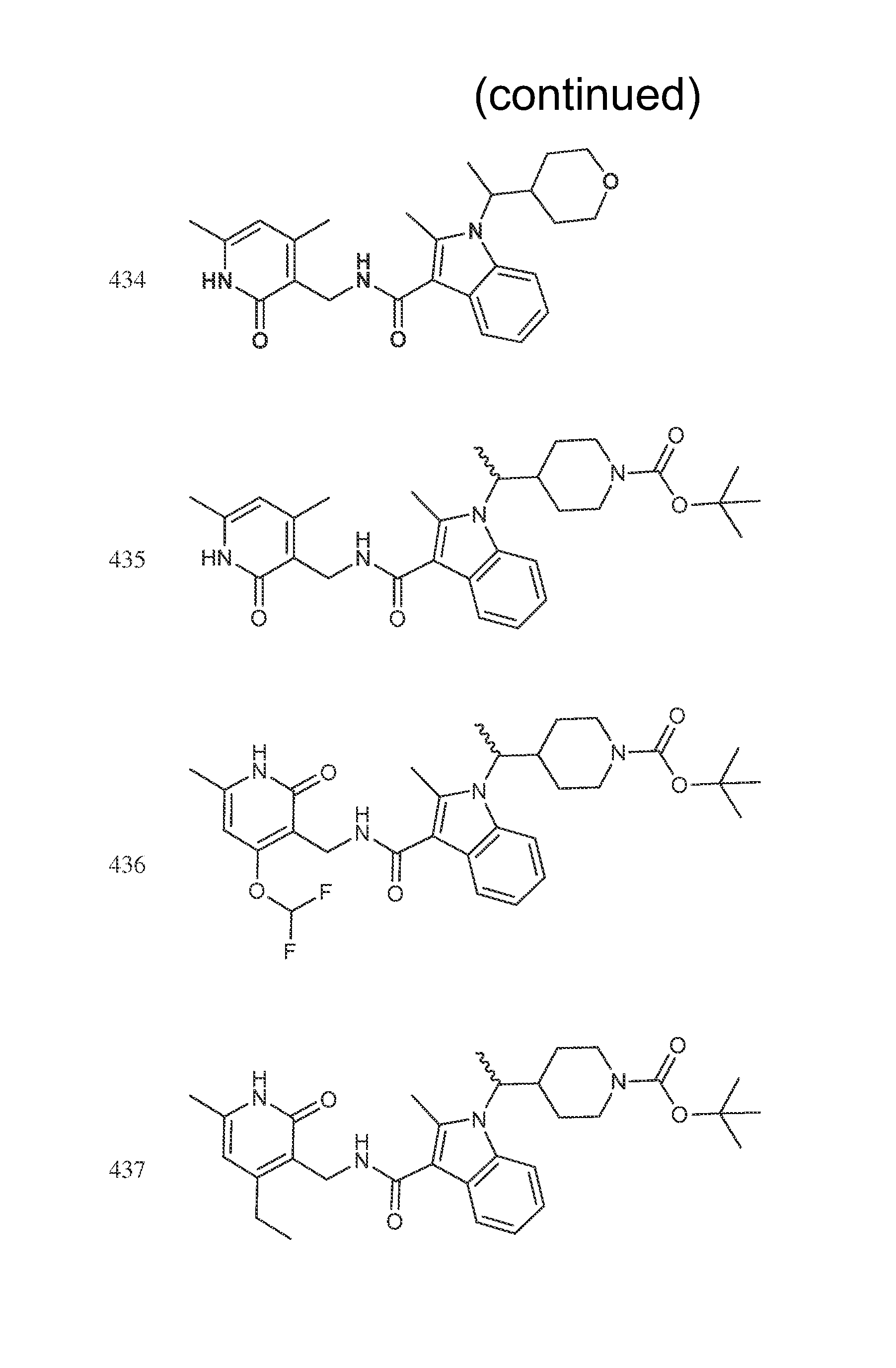
D00045
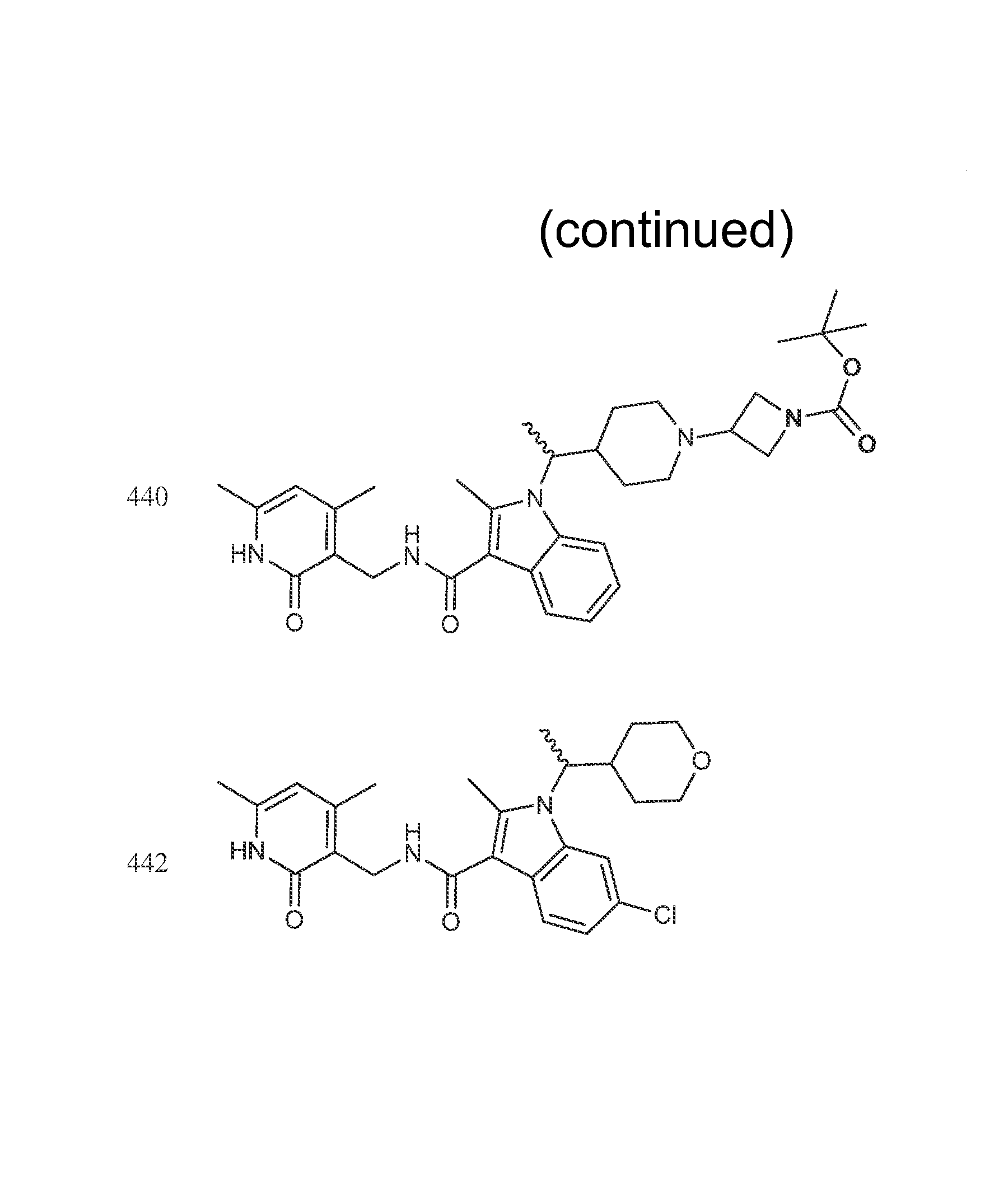
P00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.