Methods and devices for protecting catheter tips and stereotactic fixtures for microcatheters
Singh , et al. Oc
U.S. patent number 10,456,533 [Application Number 14/306,925] was granted by the patent office on 2019-10-29 for methods and devices for protecting catheter tips and stereotactic fixtures for microcatheters. This patent grant is currently assigned to Alcyone Lifesciences, Inc.. The grantee listed for this patent is ALCYONE LIFESCIENCES, INC.. Invention is credited to P J Anand, Deep Arjun Singh.








View All Diagrams
| United States Patent | 10,456,533 |
| Singh , et al. | October 29, 2019 |
Methods and devices for protecting catheter tips and stereotactic fixtures for microcatheters
Abstract
Methods and devices are disclosed herein that generally provide protection for devices (e.g., microcatheters) having small tips. Methods and devices are also disclosed herein that generally facilitate use of commercially-available stereotactic systems with devices (e.g., microcatheters) having small tips.
| Inventors: | Singh; Deep Arjun (Allston, MA), Anand; P J (Ayer, MA) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | Alcyone Lifesciences, Inc.
(Lowell, MA) |
||||||||||
| Family ID: | 52019843 | ||||||||||
| Appl. No.: | 14/306,925 | ||||||||||
| Filed: | June 17, 2014 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20140371711 A1 | Dec 18, 2014 | |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | Issue Date | ||
|---|---|---|---|---|---|
| 61835905 | Jun 17, 2013 | ||||
| 61984061 | Apr 25, 2014 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61B 90/11 (20160201); A61M 5/46 (20130101); A61B 17/3496 (20130101); A61B 2090/034 (20160201) |
| Current International Class: | A61M 5/46 (20060101); A61B 17/34 (20060101); A61B 90/00 (20160101); A61B 90/11 (20160101) |
| Field of Search: | ;606/108,130 |
References Cited [Referenced By]
U.S. Patent Documents
| 2830587 | April 1958 | Everett |
| 3460537 | August 1969 | Zeis |
| 3886948 | June 1975 | Hakim |
| 4146029 | March 1979 | Ellinwood, Jr. |
| 4692146 | September 1987 | Hilger |
| 4885945 | December 1989 | Chiodo |
| 4917686 | April 1990 | Bayston et al. |
| 4979284 | December 1990 | McMurtry et al. |
| 5088208 | February 1992 | Wells et al. |
| 5101548 | April 1992 | McMurtry et al. |
| 5190046 | March 1993 | Shturman |
| 5407431 | April 1995 | Botich et al. |
| 5415648 | May 1995 | Malay et al. |
| 5509910 | April 1996 | Lunn |
| 5590657 | January 1997 | Cain et al. |
| 5620479 | April 1997 | Diederich |
| 5624396 | April 1997 | McNamara et al. |
| 5695518 | December 1997 | Laerum |
| 5720720 | February 1998 | Laske et al. |
| 5782645 | July 1998 | Stobie et al. |
| 5843150 | December 1998 | Dreessen et al. |
| 5868711 | February 1999 | Kramer et al. |
| 5954687 | September 1999 | Baudino |
| 5963367 | October 1999 | Aksyuk et al. |
| 6061587 | May 2000 | Kucharczyk et al. |
| 6176842 | January 2001 | Tachibana et al. |
| 6193963 | February 2001 | Stern et al. |
| 6200291 | March 2001 | Di Pietro |
| 6224566 | May 2001 | Loeb |
| 6309634 | October 2001 | Bankiewicz et al. |
| 6454945 | September 2002 | Weigl et al. |
| 6464662 | October 2002 | Raghavan et al. |
| 6464687 | October 2002 | Ishikawa et al. |
| 6471993 | October 2002 | Shastri et al. |
| 6547779 | April 2003 | Levine et al. |
| 6599274 | July 2003 | Kucharczyk et al. |
| 6610235 | August 2003 | Lebouitz et al. |
| 6626902 | September 2003 | Kucharczyk et al. |
| 6706009 | March 2004 | Diermann et al. |
| 6803568 | October 2004 | Bousse et al. |
| 6953575 | October 2005 | Bankiewicz et al. |
| 7029697 | April 2006 | Segura et al. |
| 7048716 | May 2006 | Kucharczyk et al. |
| 7316676 | January 2008 | Peyman et al. |
| 7534613 | May 2009 | Bankiewicz et al. |
| 7549989 | June 2009 | Morgan et al. |
| 7588574 | September 2009 | Assell et al. |
| 7690325 | April 2010 | Henderson et al. |
| 7713269 | May 2010 | Auge, II et al. |
| 7771387 | August 2010 | Porter |
| 7842006 | November 2010 | Wang et al. |
| 7984929 | July 2011 | Gill |
| 8128600 | March 2012 | Gill |
| 8192366 | June 2012 | Mauge et al. |
| 8282566 | October 2012 | Mauge et al. |
| 8309355 | November 2012 | Bankiewicz et al. |
| 8347696 | January 2013 | Espinosa et al. |
| 8539905 | September 2013 | Cady et al. |
| 8602644 | December 2013 | Choi |
| 8790317 | July 2014 | Olbricht et al. |
| 8814853 | August 2014 | Bosel |
| 8992458 | March 2015 | Singh et al. |
| 9255245 | February 2016 | Bernick et al. |
| 9445838 | September 2016 | Wei et al. |
| 9844585 | December 2017 | Olbricht et al. |
| 9919129 | March 2018 | Singh et al. |
| 10065016 | September 2018 | Singh et al. |
| 2001/0005552 | June 2001 | Berg et al. |
| 2002/0055702 | May 2002 | Atala et al. |
| 2002/0055731 | May 2002 | Atala et al. |
| 2002/0099356 | July 2002 | Unger et al. |
| 2002/0138036 | September 2002 | Babaev |
| 2002/0193817 | December 2002 | Lal et al. |
| 2003/0009153 | January 2003 | Brisken et al. |
| 2003/0048969 | March 2003 | Hunter et al. |
| 2003/0093032 | May 2003 | Py et al. |
| 2003/0138403 | July 2003 | Drustrup |
| 2003/0148539 | August 2003 | van Dam et al. |
| 2003/0205947 | November 2003 | Klee et al. |
| 2003/0216685 | November 2003 | Porter |
| 2003/0216714 | November 2003 | Gill |
| 2004/0073114 | April 2004 | Oliver et al. |
| 2004/0106904 | June 2004 | Gonnelli et al. |
| 2004/0176732 | September 2004 | Frazier et al. |
| 2004/0186384 | September 2004 | Babaev |
| 2004/0220543 | November 2004 | Heruth et al. |
| 2004/0260241 | December 2004 | Yamamoto et al. |
| 2005/0035983 | February 2005 | Cruchon-Dupeyrat et al. |
| 2005/0125007 | June 2005 | Gill |
| 2005/0137134 | June 2005 | Gill et al. |
| 2005/0137531 | June 2005 | Prausnitz et al. |
| 2005/0143790 | June 2005 | Kipke et al. |
| 2005/0154297 | July 2005 | Gill |
| 2005/0177117 | August 2005 | Crocker et al. |
| 2005/0190999 | September 2005 | Hunter et al. |
| 2005/0236566 | October 2005 | Liu |
| 2005/0269251 | December 2005 | Cork et al. |
| 2005/0277862 | December 2005 | Anand |
| 2006/0003310 | January 2006 | Klauke et al. |
| 2006/0025752 | February 2006 | Broaddus et al. |
| 2006/0122677 | June 2006 | Vardiman |
| 2006/0135945 | June 2006 | Bankiewicz et al. |
| 2006/0211944 | September 2006 | Mauge et al. |
| 2006/0211945 | September 2006 | Mauge et al. |
| 2006/0211946 | September 2006 | Mauge et al. |
| 2007/0005017 | January 2007 | Alchas et al. |
| 2007/0016041 | January 2007 | Nita |
| 2007/0055180 | March 2007 | Deem et al. |
| 2007/0088295 | April 2007 | Bankiewicz |
| 2007/0123843 | May 2007 | Gill |
| 2007/0128083 | June 2007 | Yantz et al. |
| 2007/0163137 | July 2007 | Hunter et al. |
| 2007/0191767 | August 2007 | Hennessy et al. |
| 2007/0250054 | October 2007 | Drake |
| 2007/0276340 | November 2007 | Poston et al. |
| 2008/0004572 | January 2008 | Morris et al. |
| 2008/0091104 | April 2008 | Abraham |
| 2008/0275466 | November 2008 | Skakoon |
| 2008/0294096 | November 2008 | Uber, III et al. |
| 2008/0302960 | December 2008 | Meister et al. |
| 2009/0030373 | January 2009 | Gill |
| 2009/0048508 | February 2009 | Gill et al. |
| 2009/0071833 | March 2009 | Gorfinkel et al. |
| 2009/0088730 | April 2009 | Hoofnagle et al. |
| 2009/0112278 | April 2009 | Wingeier et al. |
| 2009/0124976 | May 2009 | Mittermeyer |
| 2009/0143659 | June 2009 | Li et al. |
| 2009/0143764 | June 2009 | Nelson |
| 2009/0198218 | August 2009 | Gill et al. |
| 2009/0224529 | September 2009 | Gill |
| 2009/0279815 | November 2009 | Hunter et al. |
| 2009/0304314 | December 2009 | Derrick et al. |
| 2010/0030102 | February 2010 | Poston |
| 2010/0030148 | February 2010 | Alchas et al. |
| 2010/0042070 | February 2010 | Gill et al. |
| 2010/0042098 | February 2010 | Cross et al. |
| 2010/0098767 | April 2010 | Olbricht et al. |
| 2010/0121307 | May 2010 | Lockard et al. |
| 2010/0130884 | May 2010 | Linninger |
| 2010/0145304 | June 2010 | Cressman |
| 2010/0168583 | July 2010 | Dausch et al. |
| 2010/0185179 | July 2010 | Chan |
| 2010/0199788 | August 2010 | Ayliffe et al. |
| 2010/0217196 | August 2010 | Nelson |
| 2010/0217228 | August 2010 | Grahn et al. |
| 2010/0217236 | August 2010 | Gill et al. |
| 2010/0256549 | October 2010 | Kralick et al. |
| 2010/0298163 | November 2010 | Juncker et al. |
| 2010/0312193 | December 2010 | Stratton et al. |
| 2010/0318061 | December 2010 | Derrick et al. |
| 2010/0318064 | December 2010 | Derrick et al. |
| 2010/0324127 | December 2010 | Kay |
| 2011/0003330 | January 2011 | Durack |
| 2011/0009879 | January 2011 | Derrick et al. |
| 2011/0098580 | April 2011 | Mikhail et al. |
| 2011/0106054 | May 2011 | Osborne et al. |
| 2011/0137289 | June 2011 | Kunst |
| 2011/0178505 | July 2011 | Odland et al. |
| 2011/0184503 | July 2011 | Xu et al. |
| 2011/0200244 | August 2011 | Ashton et al. |
| 2011/0218494 | September 2011 | Gerrans et al. |
| 2011/0275994 | November 2011 | Iwase et al. |
| 2011/0282319 | November 2011 | Gill |
| 2011/0301235 | December 2011 | Erlanson et al. |
| 2012/0019270 | January 2012 | Amodei et al. |
| 2012/0041394 | February 2012 | Haider et al. |
| 2012/0046666 | February 2012 | Klein |
| 2012/0060847 | March 2012 | Stratton et al. |
| 2012/0065496 | March 2012 | Stratton et al. |
| 2012/0083739 | April 2012 | Nelson |
| 2012/0083742 | April 2012 | Nelson |
| 2012/0123391 | May 2012 | Gill et al. |
| 2012/0209110 | August 2012 | Bankiewicz et al. |
| 2012/0209303 | August 2012 | Frankhouser et al. |
| 2012/0257846 | October 2012 | Derrick et al. |
| 2012/0302959 | November 2012 | Fielder et al. |
| 2012/0310182 | December 2012 | Fielder et al. |
| 2012/0310215 | December 2012 | Stout |
| 2013/0019488 | January 2013 | McMurtry et al. |
| 2013/0035560 | February 2013 | Anand et al. |
| 2013/0035574 | February 2013 | Anand |
| 2013/0035660 | February 2013 | Anand |
| 2013/0046230 | February 2013 | Lewis, Jr. et al. |
| 2013/0072882 | March 2013 | Ogawa et al. |
| 2013/0079596 | March 2013 | Smith |
| 2013/0079779 | March 2013 | Smith |
| 2013/0204202 | August 2013 | Trombly et al. |
| 2013/0310767 | November 2013 | Solar et al. |
| 2014/0039459 | February 2014 | Folk et al. |
| 2014/0171760 | June 2014 | Singh et al. |
| 2014/0171902 | June 2014 | Singh et al. |
| 2014/0276417 | September 2014 | Nelson |
| 2014/0371712 | December 2014 | Olbricht et al. |
| 2015/0038949 | February 2015 | Singh et al. |
| 2015/0133887 | May 2015 | Singh et al. |
| 2016/0213312 | July 2016 | Singh et al. |
| 2016/0346505 | December 2016 | Gill et al. |
| 2018/0193595 | July 2018 | Singh et al. |
| 101123919 | Feb 2008 | CN | |||
| 101657189 | Feb 2010 | CN | |||
| 2 042 212 | Apr 2009 | EP | |||
| 2009-507531 | Feb 2009 | JP | |||
| 2009-526589 | Jul 2009 | JP | |||
| 2010-501233 | Jan 2010 | JP | |||
| 2011-212502 | Oct 2011 | JP | |||
| 95/05864 | Mar 1995 | WO | |||
| 97/00442 | Jan 1997 | WO | |||
| 97/17105 | May 1997 | WO | |||
| 97/40874 | Nov 1997 | WO | |||
| 97/48425 | Dec 1997 | WO | |||
| 98/52064 | Nov 1998 | WO | |||
| 99/52585 | Oct 1999 | WO | |||
| 00/51669 | Sep 2000 | WO | |||
| 02/068036 | Sep 2002 | WO | |||
| 02/085431 | Oct 2002 | WO | |||
| 2004/060465 | Jul 2004 | WO | |||
| 2006/015091 | Feb 2006 | WO | |||
| 2007/093778 | Aug 2007 | WO | |||
| 2007/104953 | Sep 2007 | WO | |||
| 2007/133545 | Nov 2007 | WO | |||
| 2008/100930 | Aug 2008 | WO | |||
| 2008/134509 | Nov 2008 | WO | |||
| 2010/006293 | Jan 2010 | WO | |||
| 2010/081072 | Jul 2010 | WO | |||
| 2011/098769 | Aug 2011 | WO | |||
| 2011/109735 | Sep 2011 | WO | |||
| 2012/145652 | Oct 2012 | WO | |||
| 2013/019830 | Feb 2013 | WO | |||
| 2014/016591 | Jan 2014 | WO | |||
Other References
|
Invitation to Pay Additonal Fees for Application No. PCT/US2014/049031, dated Nov. 24, 2014 (2 pages). cited by applicant . International Search Report and Written Opinion for Application No. PCT/2014/049031 dated Jan. 30, 2015 (16 pages). cited by applicant . Lewis et al., Design and characterization of a high-power ultrasound driver with ultralow-output impedance. Rev Sci Instrum. Nov. 2009;80(11):114704.1-114704.8. cited by applicant . Burmeister et al.; Improved Ceramic-Based Multisite Microelectrode for Rapid Measurements of L-Giutamate in the CNS; Journal of Neuroscience Methods 119 (2002) 163-171; Elsevier Science B.V. cited by applicant . International Search Report and Written Opinion for Application No. PCT/US2012/049100, dated Jan. 29, 2013. (12 pages). cited by applicant . International Search Report and Written Opinion for Application No. PCT/US2013/076084 dated Mar. 11, 2014 (13 Pages). cited by applicant . International Search Report for International Application No. PCT/US2011/027238, dated Nov. 15, 2011. cited by applicant . Olbricht, William L. et al., Microfluidic Probes in the Treatment of Brain-Related Diseases, Drug News and Perspectives, 2010, 23(8)--7 pages (Oct. 2010). cited by applicant . Saltzman et al.; Building Drug Delivery Into Tissue Engineering; Nature Reviews/Drug Discovery; 2002 Macmillan Magazines Ltd.; vol. 1; Mar. 2002; pp. 177-186. cited by applicant . International Search Report and Written Opinion for Application No. PCT/US2014/042726 dated Oct. 28, 2014 (13 Pages). cited by applicant . Extended European Search Report for Application No. 12819276.2, dated Mar. 23, 2015 (7 pages). cited by applicant . Extended European Search Report for Application No. 13865917.2, dated Aug. 17, 2016 (6 pages). cited by applicant . Extended European Search Report for Application No. 14814380.3, dated Nov. 11, 2016. (7 pages). cited by applicant . Chinese Office Action for Application No. 201280046268.8, dated May 27, 2015 (45 pages). cited by applicant . Debinski, W., et al., "Convection-enhanced Delivery for the Treatment of Brain Tumors," Expert Rev Neurother. Oct. 2009; 9(10): 1519-1527. cited by applicant . Fiandaca, M., et al., "Use of Convection-Enhanced Delivery with Liposomal Toxins in Neurooncology," Toxins 2011, 3 (4), 369-397. cited by applicant . Rapoport, S.I., "Osmotic opening of the blood-brain barrier: principles, mechanism, and therapeutic applications," Cell. Mol. Neurobiol. 20: 217-30 (2000). cited by applicant . Extended European Search Report for Application No. 14831460.2, dated Mar. 2, 2017 (7 pages). cited by applicant . Japanese Office Action for Application No. 2015-549618, dated Sep. 5, 2017 (12 pages). cited by applicant . Japanese Office Action for Application No. 2016-531883, dated Jun. 5, 2018 (10 pages). cited by applicant. |
Primary Examiner: Erezo; Darwin P
Assistant Examiner: Schwiker; Katherine H
Attorney, Agent or Firm: Nutter, McClennen & Fish LLP
Parent Case Text
CROSS-REFERENCE TO RELATED APPLICATIONS
This application claims priority to U.S. Provisional Application No. 61/835,905 filed on Jun. 17, 2013 and to U.S. Provisional Application No. 61/984,061 filed on Apr. 25, 2014, each of which is hereby incorporated herein by reference in its entirety.
Claims
The invention claimed is:
1. A method of inserting a catheter into a patient, comprising: registering a stereotactic system to the patient; inserting the catheter having a tip protection device disposed over a distal tip thereof into a working channel of the stereotactic system until a depth stop on the tip protection device prevents further insertion, the tip protection device comprising an elongate body having a central lumen extending longitudinally therethrough configured to slidably receive the catheter, the tip protection device further including a locking mechanism comprising a set screw configured to intersect the central lumen, wherein the locking mechanism of the tip protection device is engaged with a portion of the catheter in the central lumen during insertion to hold the catheter in a fixed longitudinal position with respect to the tip protection device and, thereby, to prevent the catheter from advancing longitudinally any of distally and proximally in the tip protection device; releasing the locking mechanism of the tip protection and advancing the catheter distally into the patient; and engaging a locking mechanism of the stereotactic system with the tip protection device by tightening a set screw of the stereotactic system such that the set screw contacts an outer surface of the body of the tip protection device while the catheter is located within the central lumen of the tip protection device, thereby engaging the tip protection device onto the catheter to maintain a fixed longitudinal position between the catheter and the stereotactic system.
2. The method of claim 1, further comprising delivering a therapeutic agent through the catheter using convection-enhanced delivery.
3. The method of claim 1, further comprising, before said releasing and engaging, piercing a dura of the patient with a sharpened distal tip of the tip protection device, removing the tip protection device from the stereotactic system, snapping off the sharpened distal tip of the tip protection device, and reinserting the tip protection device through the stereotactic system.
4. The method of claim 1, further comprising inserting the tip protection device through a central lumen of a guide tube mounted in the stereotactic system such that a distal end of the tip protection device is received within a proximal portion of the central lumen of the guide tube.
5. The method of claim 4, wherein advancing the catheter comprises advancing a distal tip of the catheter through a distal portion of the central lumen of the guide tube, the distal portion of the central lumen of the guide tube having a diameter that is less than a diameter of the proximal portion of the central lumen of the guide tube, such that at least a portion of the catheter is disposed within the distal portion of the central lumen of the guide tube.
6. The method of claim 4, further comprising inserting the guide tube through a guide stop adapter and a guide block adapter mounted in the stereotactic system.
7. The method of claim 1, further comprising, before said releasing and engaging, piercing a tissue of the patient with a sharpened distal tip of the tip protection device, and subsequently separating the sharpened distal tip from the tip protection device at a perforated snap portion.
8. A method of inserting a catheter into a patient, comprising: registering a stereotactic system to the patient; inserting the catheter having a tip protection device disposed over a distal tip thereof into a working channel of the stereotactic system until a depth stop on the tip protection device prevents further insertion; releasing a locking mechanism of the tip protection device that prevents the catheter from moving longitudinally any of distally and proximally within the tip protection device, where the releasing step includes releasing the locking mechanism by loosening a set screw of the tip protection device thereby laterally withdrawing the set screw from a central lumen of the tip protection device in which the catheter is disposed such that the catheter is free to move longitudinally with respect to the tip protection device and the working channel, and advancing the catheter distally into the patient; and engaging a locking mechanism of the stereotactic system over the tip protection device, thereby tightening the tip protection device onto the catheter to fix a longitudinal position between the catheter, the tip protection device, and the stereotactic system.
9. The method of claim 8, further comprising delivering a therapeutic agent through the catheter using convection-enhanced delivery.
10. The method of claim 8, further comprising, before said releasing and engaging, piercing a dura of the patient with a sharpened distal tip of the tip protection device, removing the tip protection device from the stereotactic system, snapping off the sharpened distal tip of the tip protection device, and reinserting the tip protection device through the stereotactic system.
11. The method of claim 8, further comprising inserting the tip protection device through a central lumen of a guide tube mounted in the stereotactic system such that a distal end of the tip protection device is received within a proximal portion of the central lumen of the guide tube.
12. The method of claim 11, wherein advancing the catheter comprises advancing a distal tip of the catheter through a distal portion of the central lumen of the guide tube, the distal portion of the central lumen of the guide tube having a diameter that is less than a diameter of the proximal portion of the central lumen of the guide tube, such that at least a portion of the catheter is disposed within the distal portion of the central lumen of the guide tube.
13. The method of claim 11, further comprising inserting the guide tube through a guide stop adapter and a guide block adapter mounted in the stereotactic system.
14. The method of claim 8, wherein the tip protection device comprises an elongate body having a central lumen extending longitudinally therethrough configured to slidably receive the catheter, wherein the locking mechanism of the tip protection device is engaged with a portion of the catheter in the central lumen during insertion of the catheter into the stereotactic system to hold the catheter in a fixed longitudinal position with respect to the tip protection device.
15. The method of claim 8, further comprising, before said releasing and engaging, piercing a tissue of the patient with a sharpened distal tip of the tip protection device, and subsequently separating the sharpened distal tip from the tip protection device at a perforated snap portion.
16. A method of inserting a catheter into a patient, comprising: registering a stereotactic system to the patient; inserting the catheter having a tip protection device disposed over a distal tip thereof into a working channel of the stereotactic system until a depth stop on the tip protection device prevents further insertion, the tip protection device comprising an elongate body having a central lumen extending longitudinally therethrough configured to slidably receive the catheter, the tip protection device further including a locking mechanism comprising a set screw configured to intersect the central lumen, wherein the locking mechanism of the tip protection device is engaged with a portion of the catheter in the central lumen during insertion to hold the catheter in a fixed longitudinal position with respect to the tip protection device; piercing a dura of the patient with a sharpened distal tip of the tip protection device; removing the tip protection device from the stereotactic system and snapping off the sharpened distal tip of the tip protection device at a perforated snap portion; reinserting the tip protection device through the stereotactic system; releasing the locking mechanism of the tip protection device and advancing the catheter distally into the patient; and engaging a locking mechanism of the stereotactic system with the tip protection device by tightening a set screw of the stereotactic system such that the set screw contacts an outer surface of the body of the tip protection device while the catheter is located within the central lumen of the tip protection device, thereby engaging the tip protection device onto the catheter to maintain a fixed longitudinal position between the catheter and the stereotactic system.
Description
FIELD
Methods and devices for protecting catheter tips and stereotactic fixtures for microcatheters are disclosed herein.
BACKGROUND
In convection-enhanced delivery (CED), drugs are infused locally into tissue through a needle, cannula, or microcatheter inserted into the tissue. Transport of the infused material is dominated by convection, which enhances drug penetration into the target tissue compared with diffusion-mediated delivery or systemic delivery.
The devices used to perform CED, as well as devices used in several other fields, can include a very small, thin tip (e.g., a microfabricated tip). For example, as shown in FIG. 1, a microcatheter 100 can include a catheter body 102 with a microfabricated tip 104 at the distal end thereof. The tip can be damaged or broken during handling and/or during a surgical procedure. For example, the tip can either break during handling as a user hits the catheter tip against an object, or the surgeon may break the tip while inserting it in the brain through a stereotactic system. Stereotactic systems generally have a lumen with a small inside diameter (ID) to snugly fit the catheter. For example, as shown in FIG. 2, an exemplary stereotactic system 200 has a small-diameter central lumen 202 extending therethrough. The surgeon is required to "aim" the small catheter into the tight lumen to get the catheter loaded into the stereotactic system. Catheters with small tips may get damaged as the surgeon may hit the tip against the stereotactic system while manually trying to align the catheter to the small lumen. In addition, stereotactic systems are generally sized for larger instruments and cannot adequately support and protect catheters with small diameters or small tips.
A need exists for methods and devices for protecting catheter tips and stereotactic fixtures for microcatheters.
SUMMARY
Methods and devices are disclosed herein that generally provide protection for devices (e.g., microcatheters) having small tips. Methods and devices are also disclosed herein that generally facilitate use of commercially-available stereotactic systems with devices (e.g., microcatheters) having small tips.
In some embodiments, a tip protection device includes an elongate body having a central lumen extending longitudinally therethrough, the lumen being sized and configured to slidably receive a catheter, and a locking mechanism configured to selectively maintain the elongate body in a fixed longitudinal position relative to a catheter inserted through the central lumen.
The locking mechanism can include a screw. The elongate body can include an increased-diameter portion configured to act as a depth stop when the elongate body is inserted through a lumen of a stereotactic system. The elongate body can be formed from at least one of silastic, poly-urethane, poly-ester, PTFE, E-PTFE, stainless steel, polycarbonate, PVC, Delrin, aluminum, PEEK, plastic, metal, and titanium. The elongate body can be fabricated using at least one of extrusion, molding, and machining. The elongate body can include a sharpened distal tip. The distal tip can be separable from the elongate body along a perforated snap portion. The elongate body can include a distal cylindrical portion having a first diameter and a proximal cylindrical portion having a second diameter that is greater than the first diameter. The central lumen can have a diameter of about 0.5 mm to about 4.0 mm.
In some embodiments, a system includes a tip protection device (e.g., of the type described above) and a depth stop comprising a cylindrical body portion having a central lumen extending longitudinally therethrough and a locking mechanism configured to selectively engage a catheter inserted through the cylindrical body portion.
In some embodiments, a system includes a tip protection device (e.g., of the type described above) and a guide tube that includes an elongate body having a central lumen extending longitudinally therethrough, the central lumen including a proximal portion having a first diameter and a distal portion having a second diameter that is less than the first diameter, the proximal portion being sized to receive a reduced diameter distal portion of the tip protection device and the distal portion being sized to receive at least a portion of a catheter inserted through the tip protection device.
The elongate body of the guide tube can include a proximal portion having an outside diameter which is greater than an outside diameter of a distal portion of the elongate body of the guide tube. A distal end of the guide tube can be tapered. The system can include a guide stop adapter comprising a cylindrical disc having an inside diameter sized to receive the distal portion of the guide tube therethrough and an outside diameter sized to fit within a guide stop of a stereotactic system, and a guide block adapter comprising a cylindrical sleeve having an inside diameter sized to receive the distal portion of the guide tube therethrough and an outside diameter sized to fit within a guide block of a stereotactic system. The guide tube can have a length sufficient to span a distance between the guide block of the stereotactic system and a skull of a patient to which the stereotactic system is registered.
In some embodiments, a method of inserting a catheter into a patient includes registering a stereotactic system to the patient, inserting a catheter having a tip protection device disposed over a distal tip thereof into a working channel of the stereotactic system until a depth stop on the tip protection device prevents further insertion, releasing a locking mechanism of the tip protection device and advancing the catheter distally into the patient, and engaging a locking mechanism of the stereotactic system with the tip protection device, thereby engaging the tip protection device with the catheter to maintain a fixed longitudinal position between the catheter and the stereotactic device.
The method can include delivering a therapeutic agent through the catheter using convection-enhanced delivery. The method can include, before said releasing and engaging, piercing the dura of the patient with a sharpened distal tip of the tip protection device, removing the tip protection device from the stereotactic frame, snapping off the sharpened distal tip of the tip protection device, and reinserting the tip protection device through the stereotactic frame. The method can include inserting the tip protection device through a central lumen of a guide tube mounted in the stereotactic system such that a distal end of the tip protection device is received within a proximal portion of the central lumen of the guide tube. Advancing the catheter can include advancing a distal tip of the catheter through a distal portion of the central lumen of the guide tube, the distal portion of the central lumen of the guide tube having a diameter that is less than a diameter of the proximal portion of the central lumen of the guide tube, such that at least a portion of the catheter is disposed within the distal portion of the central lumen of the guide tube. The method can include inserting the guide tube through a guide stop adapter and a guide block adapter mounted in the stereotactic system.
The present invention further provides devices, systems, and methods as claimed.
BRIEF DESCRIPTION OF THE DRAWINGS
The invention will be more fully understood from the following detailed description taken in conjunction with the accompanying drawings, in which:
FIG. 1 is a perspective view of an exemplary microcatheter;
FIG. 2 is a perspective view of an exemplary stereotactic system;
FIG. 3 is a perspective view of an exemplary tip protector;
FIG. 4 is a perspective view of the tip protector of FIG. 3 shown with the microcatheter of FIG. 1;
FIG. 5 is another perspective view of the tip protector of FIG. 3 shown with the microcatheter of FIG. 1;
FIG. 6 is another perspective view of the tip protector of FIG. 3 shown with the microcatheter of FIG. 1;
FIG. 7 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, and an exemplary stereotactic system;
FIG. 8 is another perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, and an exemplary stereotactic system;
FIG. 9 is another perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, and an exemplary stereotactic system;
FIG. 10 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, and an exemplary depth stop;
FIG. 11 is perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, and an exemplary stereotactic system;
FIG. 12 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, and another exemplary stereotactic system;
FIG. 13 is a perspective view of an exemplary guide tube;
FIG. 14 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the guide tube of FIG. 13, and another exemplary stereotactic system;
FIG. 15 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, and the guide tube of FIG. 13;
FIG. 16 is another perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, and the guide tube of FIG. 13;
FIG. 17 is a perspective view of another exemplary stereotactic system;
FIG. 18 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, the guide tube of FIG. 13, an exemplary guide stop adapter, and an exemplary guide block adapter;
FIG. 19 is another perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, the guide tube of FIG. 13, and the guide stop and guide block adapters of FIG. 18; and
FIG. 20 is a perspective view of the tip protector of FIG. 3, the microcatheter of FIG. 1, the depth stop of FIG. 10, the guide tube of FIG. 13, the guide stop and guide block adapters of FIG. 18, and an exemplary stereotactic system.
DETAILED DESCRIPTION
Methods and devices are disclosed herein that generally provide protection for devices (e.g., microcatheters) having small tips. Methods and devices are also disclosed herein that generally facilitate use of commercially-available stereotactic systems with devices (e.g., microcatheters) having small tips.
Certain exemplary embodiments will now be described to provide an overall understanding of the principles of the structure, function, manufacture, and use of the methods, systems, and devices disclosed herein. One or more examples of these embodiments are illustrated in the accompanying drawings. Those skilled in the art will understand that the methods, systems, and devices specifically described herein and illustrated in the accompanying drawings are non-limiting exemplary embodiments and that the scope of the present invention is defined solely by the claims. The features illustrated or described in connection with one exemplary embodiment may be combined with the features of other embodiments. Such modifications and variations are intended to be included within the scope of the present invention.
In some embodiments, a tip protector is provided in the form of a sleeve. The sleeve can be formed by cutting a length of tubing or using extrusion, molding, and/or machining processes. The sleeve can include a central lumen extending longitudinally therethrough, defined by a relatively thin wall. The sleeve can be slid over the catheter (or similar small-tip device) to protect the catheter tip from breakage or damage during handling or use. The tip protector can be configured to sit over the catheter or other device such that it covers and protects the micro-tip. The tip protector can be secured on the catheter using a set-screw or a snap feature, or other feature that can easily be un-deployed to slide the catheter through the sleeve as needed. The tip protector can be packaged and shipped with the catheter (e.g., with the protector pre-installed over the tip of the catheter).
In use, the surgeon/user can align the catheter to the stereotactic system using the sleeve as a reference. Once aligned, the tip protector and catheter can be slid inside the stereotactic system (the tip protector can be sized to fit existing systems). Once inside the stereotactic system, the user can loosen the set-screw on the tip protector to slide the catheter further into the brain or other tissue. The proximal end of the tip protector can have a large outside diameter (OD) stop or collar that does not allow it to slide inside the stereotactic system as the catheter is being inserted into the brain. Once the catheter is inserted into the brain, the stereotactic system set-screw can be tightened over the tip-protector sleeve (due to the thin wall) onto the catheter to fix it in place and prevent the catheter from sliding. The tip protector can be MRI compatible so that it does not interfere with MR imaging.
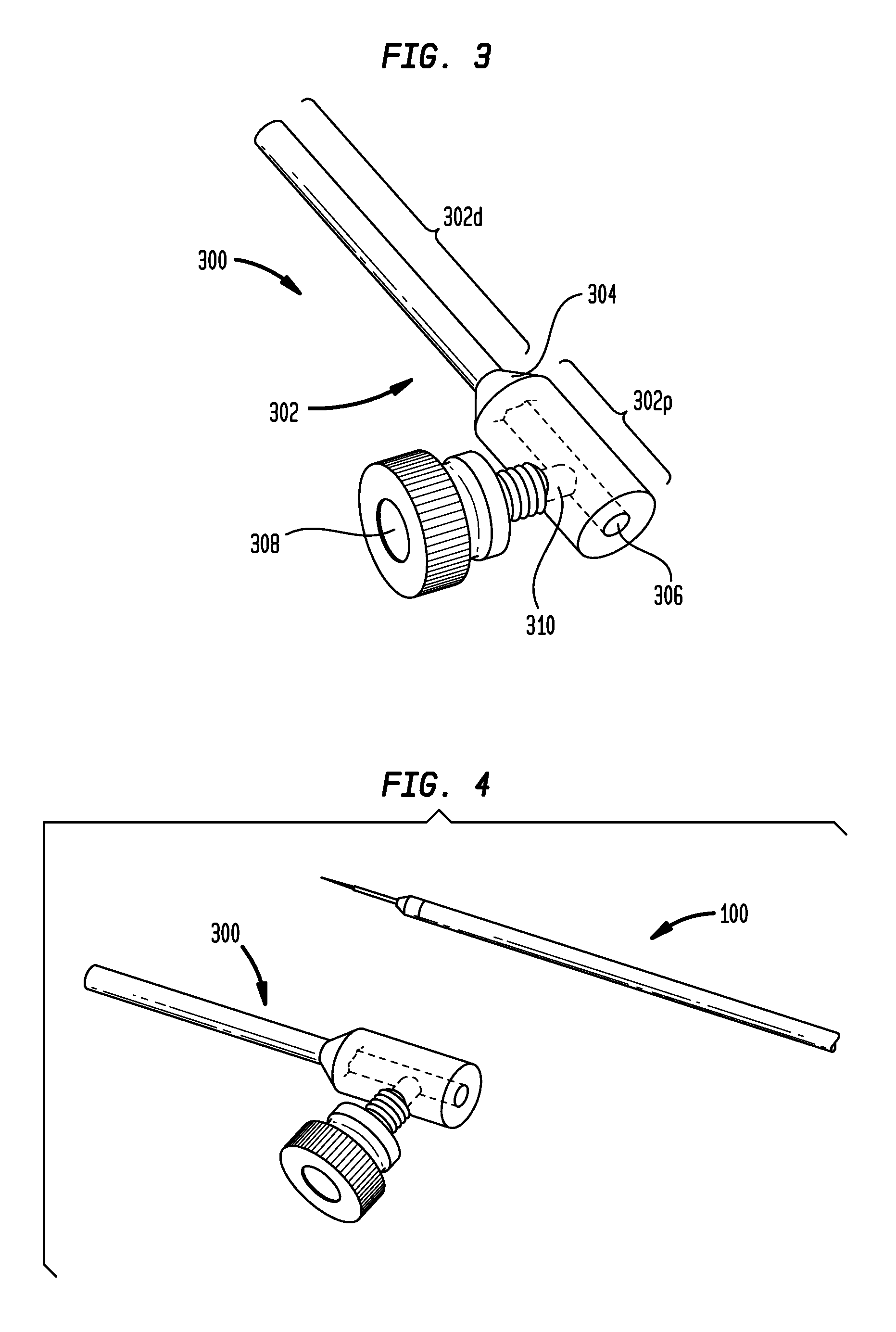
FIG. 3 illustrates an exemplary embodiment of a tip protector 300 having a set screw and a large diameter stop portion. As shown, the tip protector 300 includes an elongate sleeve 302. The distal portion 320d of the sleeve 302 has an outside diameter which is less than the outside diameter of the proximal portion 302p of the sleeve. The outside diameter of the sleeve 302 can be curved, ramped, stepped, or tapered at the junction of the proximal and distal portions to provide a transition or shoulder 304. This transition 304 can act as a shoulder or stop surface to limit the degree to which the tip protector 300 can be advanced distally through a cylindrical opening, such as the opening of a stereotactic system. A central cylindrical lumen 306 extends through the tip protector 300 from a proximal end of the sleeve 302 to a distal end of the sleeve. The lumen 306 is sized and configured to receive at least a portion of a catheter therethrough. The tip protector 300 also includes a set screw 308 threadably mounted in a channel 310 which extends perpendicular to the central lumen 306. Rotation of the set screw 308 in a first direction can be effective to advance the set screw within the channel 310 such that it extends into the central lumen 306 and engages a catheter or other instrument inserted therethrough. Rotation of the set screw 308 in a second, opposite direction can be effective to withdraw the set screw within the channel 310 such that it does not extend into the central lumen 306 and does not engage a catheter or other instrument inserted therethrough.
The tip protector 300 is shown in FIG. 4 with an exemplary embodiment of a microcatheter 100 for convection-enhanced delivery. It will be appreciated that any of a variety of microcatheters or other instruments can be used with the tip protector. Exemplary microcatheter devices are disclosed in the following references, the entire contents of each of which are hereby incorporated by reference herein: U.S. Publication No. 2013/0035560 entitled MULTI-DIRECTIONAL MICROFLUIDIC DRUG DELIVERY DEVICE; U.S. Publication No. 2013/0035574 entitled MICROFLUIDIC DRUG DELIVERY DEVICES WITH VENTURI EFFECT; U.S. Publication No. 2013/0035660 entitled MULTIDIRECTIONAL MICROFLUIDIC DRUG DELIVERY DEVICES WITH CONFORMABLE BALLOONS; U.S. application Ser. No. 14/132,762 entitled SYSTEMS AND METHODS FOR REDUCING OR PREVENTING BACKFLOW IN A DELIVERY SYSTEM; U.S. Publication No. 2010/0098767 entitled CONVECTION ENHANCED DELIVERY APPARATUS, METHOD, AND APPLICATION; and U.S. Publication No. 2013/0046230 entitled ULTRASOUND-ASSISTED CONVECTION ENHANCED DELIVERY OF COMPOUNDS IN VIVO WITH A TRANSDUCER CANNULA ASSEMBLY.
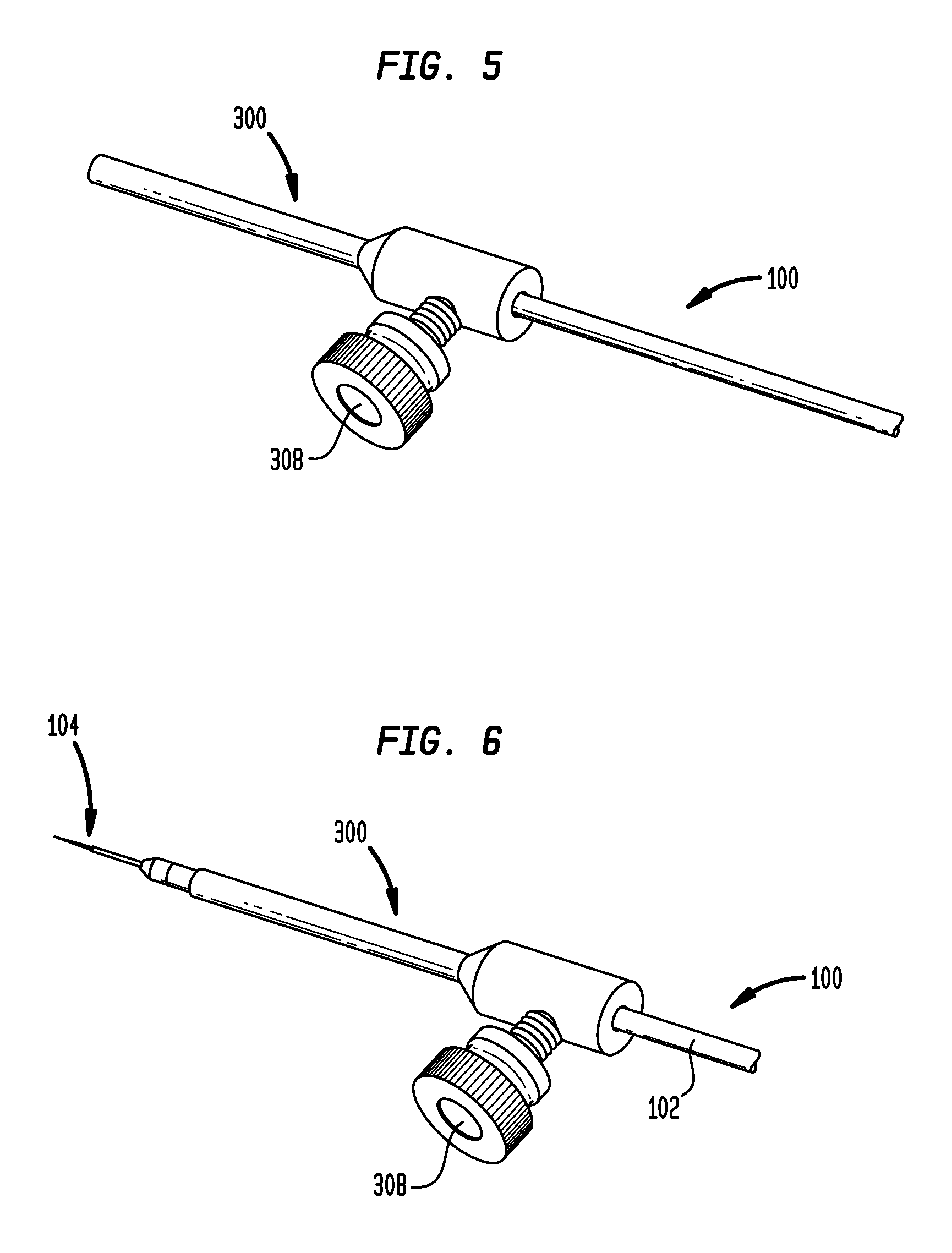
As shown in FIG. 5, the microcatheter 100 can be inserted through the central lumen 306 of the tip protector 300. The set screw 308 can be tightened to secure the tip protector 300 to the microcatheter 100 and prevent longitudinal translation of the tip protector relative to the microcatheter. The microcatheter 100 can also be positioned such that the tip 104 of the microcatheter protrudes from a distal end of the tip protector 300, as shown in FIG. 6. This relative positioning of the tip protector 300 and the microcatheter 100 would typically be used only after the microcatheter is inserted into the stereotactic system, although the user may wish to slide the tip 104 out of the tip protector before insertion to confirm fluid flow during priming, etc., and then retract the tip back into the tip protector before inserting the catheter through the stereotactic system.
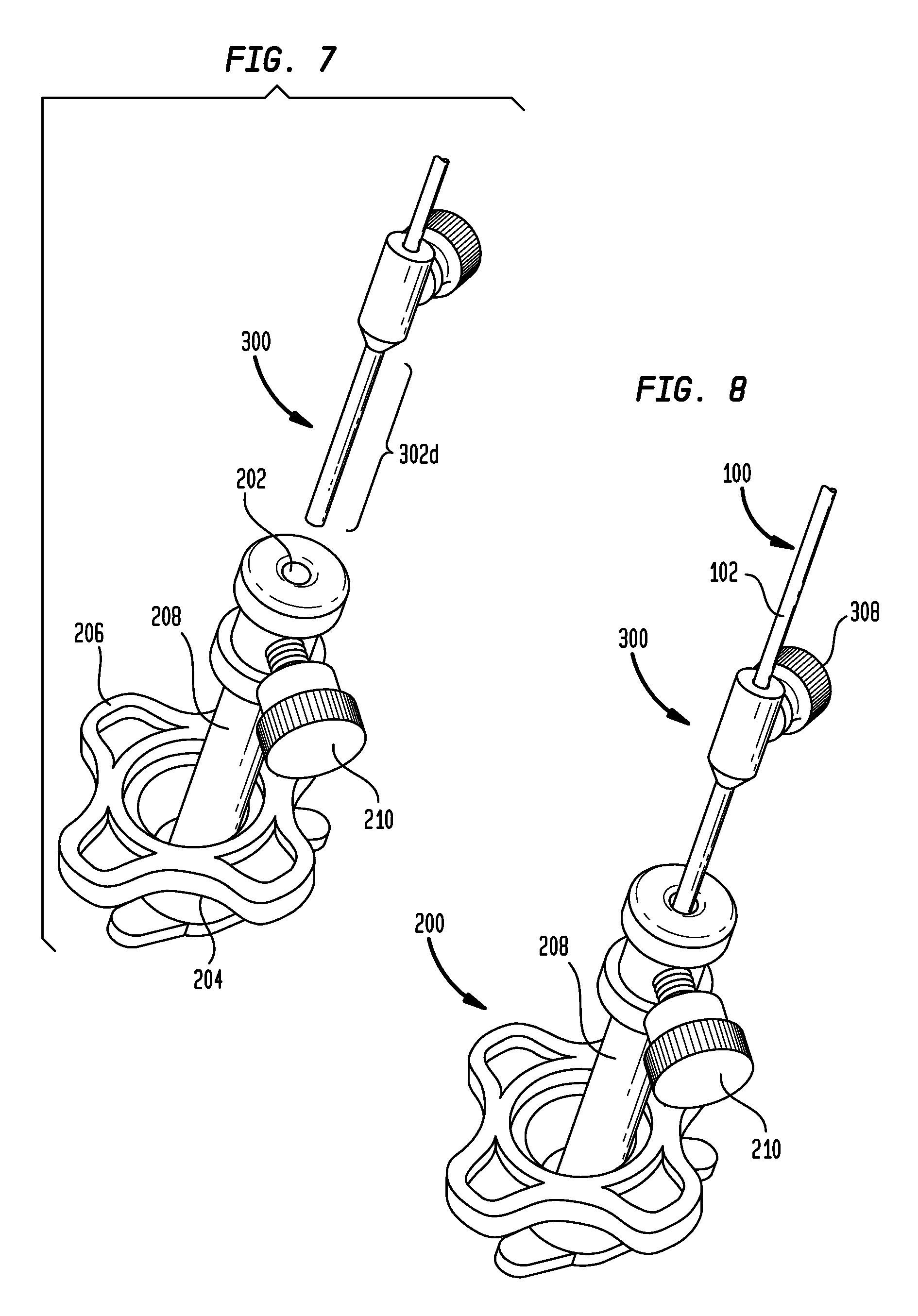
The tip protector can be used with any of a variety of stereotactic systems. For example, as shown in FIGS. 7-9, the tip protector 300 can be used with a stereotactic system 200 of the type available from MEDTRONIC, INC. under the NAVIGUS brand. The illustrated stereotactic system 200 includes a base 204 with a locking ring 206 and a stem 208 that can be positioned at various angles with respect to the base. The stem 208 includes a set screw 210 to secure a device inserted through an inner lumen 202 of the stem. In use, the base 204 is installed over a portion of the patient (e.g., a burr hole formed in the patient's skull).
As shown in FIG. 7, the microcatheter 100 can be aligned to the small hole 202 in the stem 208 without damaging the catheter tip by using the tip protector 300 as a visual and contact reference. The outside diameter of the distal portion 302d of the tip protector 300 can be sized to substantially match the inside diameter of the central lumen 202 of the stem 208 such that the tip protector fits snugly within the stereotactic system 200, as shown in FIG. 8. As shown in FIG. 9, the tip protector 300 can be advanced distally within the stem 208 until the shoulder portion 304 of the tip protector engages the proximal end of the stem and prevents further insertion. The large diameter stop portion 302p on the tip protector 300 can thus prevent the tip protector from being advanced too far through the stereotactic system 200. The distal portion 302d of the tip protector 300 can have a length that substantially corresponds to the length of the stem 208, such that the distal end of the tip protector is aligned with the distal end of the stereotactic system 200 when the tip protector is inserted up to the shoulder portion 304.
Once the tip protector 300 is inserted through the stem 208, the set screw 308 of the tip protector can be loosened to allow the microcatheter 100 to be translated longitudinally relative to the tip protector and the stem, such that the catheter tip can be advanced distally into the patient or retracted proximally from the patient. The set screw 210 on the stem 208 can be tightened over the tip protector 300 to secure the catheter 100 and the tip protector with respect to the stem.
The terminal distal end of the tip protector 300 can also be made to be sharp and, when the tip protector is fully-advanced into the stereotactic system 200, the distal tip of the tip protector can extend into the skull and past the dura to ensure the dura and corresponding anatomies are pierced and will not interfere with the catheter micro-tip 104 during insertion. For example, the distal tip of the tip protector 300 can be pointed or otherwise sharpened and can extend a few millimeters beyond the skull when inserted through the stereotactic system 200. The length of the tip protector 300 can thus be selected based on the stereotactic system with which it will be used to achieve the desired degree of protrusion. In an exemplary method of use, the catheter 100 and that elongated, sharp-tipped protector 300 can be inserted through the stereotactic system 200 such that the distal tip of the tip protector extends through the skull and a few millimeters past the dura, thereby opening, tearing, and/or piercing the dura. The catheter 100 and the tip protector 300 can then be removed and the sharp tip of the tip protector can be broken or snapped off (e.g., along a perforated snap section or frangible portion) to expose the lumen 306 of the tip protector. The tip protector 300 and the catheter 100 can then be re-inserted and used as described above.
As shown in FIG. 10, a depth stop 312 can be included for setting the desired insertion depth of the microcatheter 100 and preventing over-insertion. The illustrated depth stop 312 includes a collar 314 that can be longitudinally slidable with respect to the catheter 100 and can include a thumb screw 316 for engaging the catheter to secure the collar in a fixed longitudinal position with respect thereto. In particular, the set screw 316 can be selectively positioned such that a tip of the set screw extends into a central lumen 318 of the collar 314 to engage the microcatheter 100 disposed therein. As the microcatheter 100 is advanced distally through the tip protector 300, the collar 314 eventually contacts the proximal end of the tip protector, preventing further insertion of the catheter. The depth stop 312 can thus be slid along the catheter 100 and locked in place to set the maximum insertion depth. The catheter 100 can also include depth markings on the catheter body 102 to help a user place the depth stop 312 at the desired calculated depth.
In some embodiments, the tip protector can have a standard length to allow easy depth registration between the tip protector, the catheter, and the stereotactic system. In some embodiments, the distal portion of the tip protector is approximately 5 cm in length and the proximal, increased-diameter portion of the tip protector is approximately 1 cm in length such that the tip protector has an overall length of approximately 6 cm. Accordingly, a catheter with marked depth graduations on its exterior sidewall can be advanced into the tip protector to the 6 cm marking, indicating that the distal end of the catheter is aligned (i.e., not protruding or recessed) with the distal end of the tip protector. Similarly, the tip protector can be fully-advanced into a stereotactic system having a 5 cm stem length, such that the distal end of the tip protector is aligned (i.e., not protruding or recessed) with the distal end of the stereotactic system.
In some embodiments, the central lumen of the tip protector can have an inside diameter that corresponds to (e.g., is substantially equal to or slightly greater than) the outside diameter of the catheter. For example, the central lumen of the tip protector can have a diameter of about 0.5 mm to about 4.0 mm. In some embodiments, the central lumen of the tip protector can have a diameter of about 1.5 mm. In some embodiments, the central lumen of the tip protector can have a diameter of about 3.0 mm.
While an exemplary microcatheter 100 and an exemplary stereotactic system 200 are shown and described above, it will be appreciated that the tip protector 300 can be sized or otherwise configured to work with any of a variety of catheters or other small-tipped devices, and can likewise be sized or otherwise configured to work with any of a variety of stereotactic systems, stems, collets, sleeves, frames, etc. In addition, one or more fixtures, adapters, guides, or other accessories can be included to facilitate use of the tip protector and/or a microcatheter with a particular stereotactic system.
Exemplary stereotactic systems include the NAVIGUS system available from MEDTRONIC, INC. and the VARIOGUIDE system available from BRAINLAB. Both of these systems are "frameless," meaning they are mounted directly or close to the patient's head, and do not need the functional "frame" per conventional stereotactic procedures.
As shown above and in FIG. 11, the tip protector 300 can be sized to be received within the inner lumen of the NAVIGUS system 200. For example, the distal portion 302d of the tip protector 300 can have a length that is equal to the length of the stem 208, such that when the tip protector is inserted up to the proximal, increased-diameter portion 302p, the distal end of the tip protector is aligned with the distal end or center of the pivoting stem, which typically serves as the depth reference point in the system 200. This can advantageously allow for simple depth registration between the catheter 100, the tip protector 300, and the system 200. In some embodiments, the distal portion 302d of the tip protector 300 has a length of approximately 5 cm.
Similarly, as shown in FIG. 12, the length of the tip protector 300 can be selected to correspond with the diameter of the circular guide block 400 of the VARIOGUIDE system, thereby facilitating depth registration between the catheter 100, the tip protector 300, and the system.
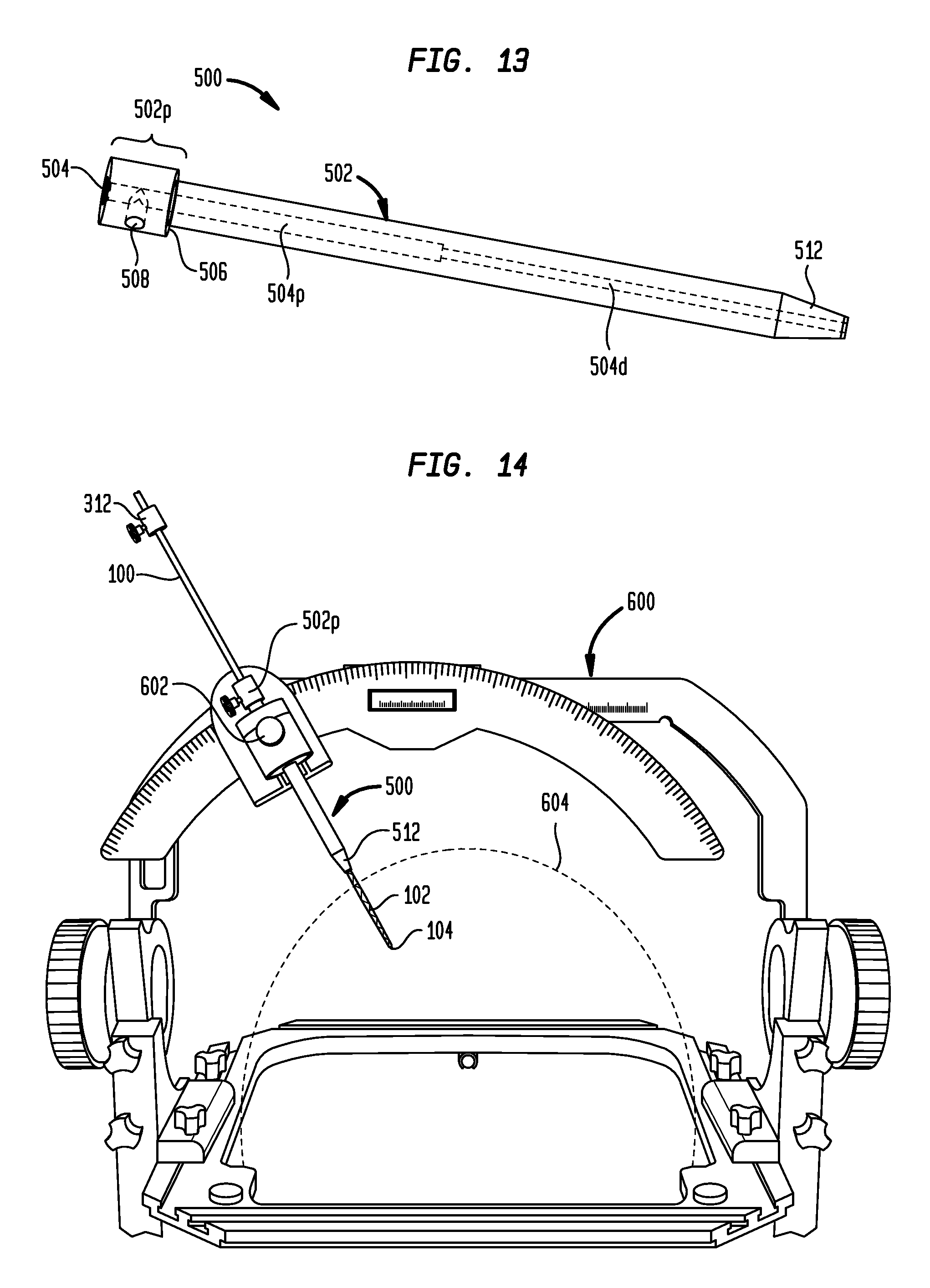
In some embodiments, a guide tube can be provided to facilitate coupling of the tip protector 300 and/or the catheter 100 to the stereotactic system. FIG. 13 illustrates an exemplary embodiment of a guide tube 500. As shown, the guide tube 500 has an elongate body 502 with a central lumen 504 extending longitudinally therethrough. The inside diameter of the central lumen 504 is stepped, such that the lumen includes an increased-diameter proximal portion 504p sized to receive the distal end 302d of the tip protector 300 and a decreased-diameter distal portion 504d sized to snugly receive a portion of the catheter 100 that protrudes from the distal end of the tip protector. The guide tube 500 also has a proximal end 502p with an enlarged outside diameter such that a shoulder 506 is defined on the exterior of the guide tube. In use, the guide tube 500 can be inserted distally through the guide block of a stereotactic system until the shoulder 506 engages the guide block. The tip protector 300 can be inserted into the central lumen 504 of the guide tube 500 such that the tip protector is supported and stabilized in the stereotactic system. A lateral opening 508 can be included in the proximal end of the guide tube 500 to receive a set screw 510 for locking the tip protector 300 in place within the central lumen 504. The enlarged proximal end 502p of the guide tube 500 can have a standard length (e.g., 1 cm) to aid in depth registration. The guide tube 500 can also include a tapered distal tip 512 for easy insertion of the guide tube into the guide block of the stereotactic system.
FIG. 14 illustrates an exemplary frame-based stereotactic system--the CRW frame available from INTEGRA LIFESCIENCES. As shown, the guide tube 500 can be sized to fit the existing guide block 602 of the frame 600 and help guide the catheter 100 from the guide block to the top of the skull 604. For example, the guide tube 500 can have an outside diameter of 0.25'' to correspond with the inside diameter of the frame's guide block 602. The guide tube 500 can thus allow the frame 600 to be used with microcatheters 100 that would not otherwise fit in the guide block 602. In some embodiments, a kit including a plurality of guide tubes having different lengths can be provided. Accordingly, as the arc of the frame 600 is moved towards or away from the patient's skull 604 depending on target depth, a guide tube having an appropriate length can be selected such that the guide tube supports the full length or a significant portion of the full length of the microcatheter 100 extending between the guide block 602 and the skull 604.
FIG. 15 is a schematic illustration of a microcatheter 100 coupled to a tip protector 300 and depth stop 312 and aligned with the guide tube 500. FIG. 16 is a schematic illustration of the microcatheter 100 and tip protector 300 inserted through the guide tube 500. As shown, the tip protector 300 can be advanced until the enlarged proximal end 302p of the tip protector engages the proximal end surface of the guide tube 500.
FIG. 17 illustrates another exemplary frame-based stereotactic system--the LEKSELL frame available from ELEKTA. As shown, the system 700 includes an upper guide stop 702 mounted to the arc 704 of the frame. The system 700 also includes a lower guide block 706 mounted to an arm 708 that extends down from the arc 704 towards the patient's skull 710. In some embodiments, a guide stop adapter 802 and a guide block adapter 804 can be provided to facilitate coupling of the guide tube 500, tip protector 300, and/or the catheter 100 to the LEKSELL system 700 or to other similar systems.
FIG. 18 illustrates a microcatheter 100 (with a depth stop 312 and a tip protector 300), a guide tube 500, a guide stop adapter 802, and a guide block adapter 804. These components are illustrated in an assembled configuration in FIG. 19 and installed in the LEKSELL frame 700 in FIG. 20.
As shown, the guide stop adapter 802 can be a cylindrical disc having an inside diameter sized to receive the guide tube 500 and an outside diameter sized to fit within the guide stop 702 of the stereotactic system 700. The guide stop adapter 802 can include an enlarged proximal end 802p that defines an exterior shoulder 806.
The guide block adapter 804 can be a cylindrical sleeve having an inside diameter sized to receive the guide tube 500 and an outside diameter sized to fit within the guide block 706 of the stereotactic system 700. The guide block adapter 804 can include an enlarged proximal end 804p that defines an exterior shoulder 808. Lateral openings 810, 812 can be formed in the guide stop adapter 802 and/or the guide block adapter 804 to receive set screws 814, 816 for locking the guide tube 500 in position. In use, the guide stop adapter 802 and the guide block adapter 804 can be fitted to the guide stop 702 and guide block 706, respectively, of the stereotactic frame 700 and adjusted to the desired heights. The guide tube 500 can then be inserted through the adapters 802, 804, and can be secured in a fixed longitudinal position by tightening the set screws 814, 816 of the guide stop adapter 802 and the guide block adapter 804. The microcatheter 100 and attached tip protector 300 can then be inserted through the guide tube 500. The set screw of the guide tube 500 can be tightened to secure the tip protector 300 to the guide tube, before or after advancing the microcatheter 100 relative to the tip protector to the desired depth. The set screw 308 of the tip protector 300 can also be tightened to secure the microcatheter 100 in a fixed longitudinal position relative to the tip protector.
It will be appreciated that similar adapters can be made to fit other frames to facilitate stereotactic use of the tip protectors and microcatheters disclosed herein. The systems and methods disclosed herein can facilitate precision-targeted drug delivery (e.g., via convection-enhanced delivery) using a stereotactic system and a microcatheter. In an exemplary embodiment, a stereotactic system is registered to a patient, for example using MR images. A microcatheter and associated tip protector can be coupled to the stereotactic system using one or more guide tubes, guide block adapters, and/or guide stop adapters as disclosed herein and aimed towards a target site in the patient. The microcatheter can then be advanced into the patient under stereotactic guidance until one or more fluid outlet ports of the microcatheter are positioned at the target site. Drug-containing fluid can then be infused under positive pressure to deliver the drug through the catheter to the target site via convection-enhanced delivery.
The tip protectors, depth stops, fixtures, adapters, guides, and other components or devices disclosed herein can be manufactured or produced using any of a variety of techniques, including extrusion, molding, machining, and combinations thereof. The tip protectors, depth stops, fixtures, adapters, guides, and other components or devices disclosed herein can be formed from a variety of materials, including silastic, poly-urethane, poly-ester, PTFE, E-PTFE, stainless steel, titanium, polycarbonate, PVC, Delrin, aluminum, PEEK, plastic, metal, and combinations thereof.
Although the invention has been described by reference to specific embodiments, it should be understood that numerous changes may be made within the spirit and scope of the inventive concepts described. Accordingly, it is intended that the invention not be limited to the described embodiments, but that it have the full scope defined by the language of the following claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.