Liquid photo initiating compound and uses of the same
Chang , et al. A
U.S. patent number 10,385,145 [Application Number 16/281,143] was granted by the patent office on 2019-08-20 for liquid photo initiating compound and uses of the same. This patent grant is currently assigned to CHITEC TECHNOLOGY CO., LTD.. The grantee listed for this patent is CHITEC TECHNOLOGY CO., LTD.. Invention is credited to Wei-Chun Chang, Chingfan Chris Chiu, Huang-Min Wu.





View All Diagrams
| United States Patent | 10,385,145 |
| Chang , et al. | August 20, 2019 |
Liquid photo initiating compound and uses of the same
Abstract
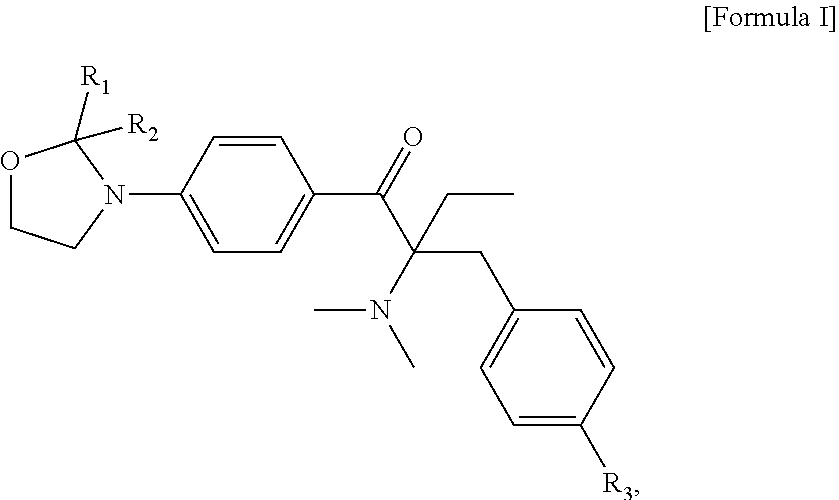
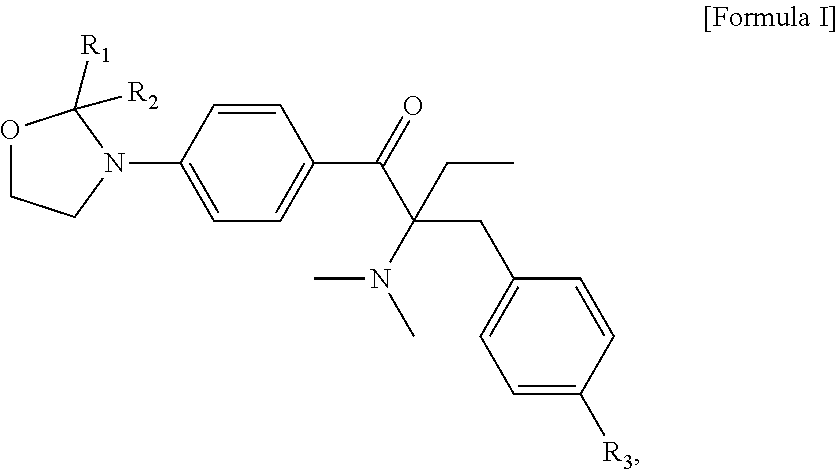
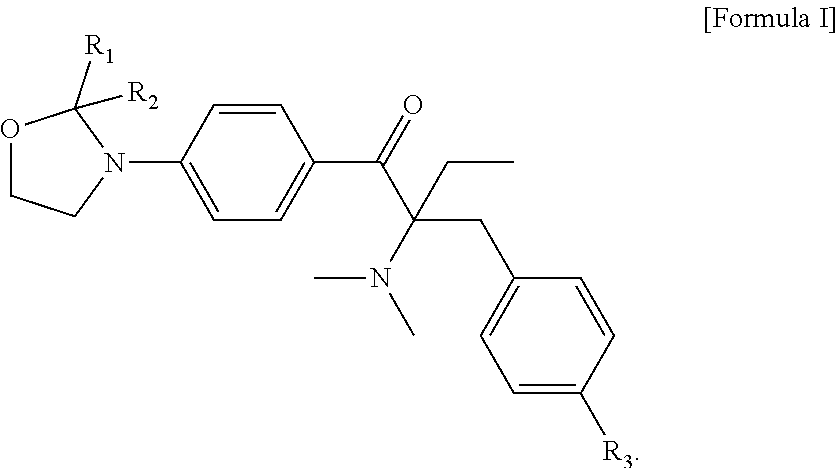
A liquid photo initiating compound and uses of the same are provided. The compound is represented by the following Formula I: ##STR00001## in Formula I, R.sub.1 and R.sub.2 are independently H or C.sub.1-C.sub.3 alkyl, and R.sub.3 is H or methyl.
| Inventors: | Chang; Wei-Chun (Taipei, TW), Chiu; Chingfan Chris (Taipei, TW), Wu; Huang-Min (Taipei, TW) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | CHITEC TECHNOLOGY CO., LTD.
(Taipei, TW) |
||||||||||
| Family ID: | 66182336 | ||||||||||
| Appl. No.: | 16/281,143 | ||||||||||
| Filed: | February 21, 2019 |
Foreign Application Priority Data
| Jul 17, 2018 [TW] | 107124654 A | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C08F 2/48 (20130101); C08F 2/50 (20130101); C08F 20/18 (20130101); C08K 5/5397 (20130101); C08K 5/08 (20130101); C08K 5/353 (20130101); C08K 5/06 (20130101); G03F 7/031 (20130101) |
| Current International Class: | C08F 2/46 (20060101); C08K 5/06 (20060101); C08G 61/04 (20060101); C08F 2/50 (20060101); C08K 5/08 (20060101); C08F 20/18 (20060101); C08K 5/353 (20060101); C08K 5/5397 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 5077402 | December 1991 | Desobry et al. |
| 7247659 | July 2007 | Kura et al. |
| 7732504 | June 2010 | Dietliker et al. |
| I277834 | Apr 2007 | TW | |||
| I564276 | Jan 2017 | TW | |||
Attorney, Agent or Firm: McClure, Qualey & Rodack, LLP
Claims
What is claimed is:
1. A liquid photo initiating compound, which is represented by the following Formula I: ##STR00015## wherein, in Formula I, R.sub.1 and R.sub.2 are independently H or C.sub.1-C.sub.3 alkyl, and R.sub.3 is H or methyl.
2. The compound of claim 1, wherein R.sub.1 and R.sub.2 are independently methyl, ethyl, n-propyl or isopropyl.
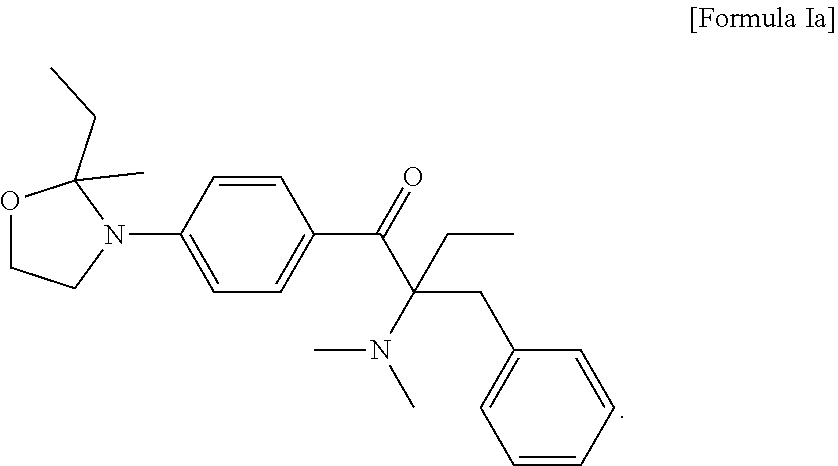
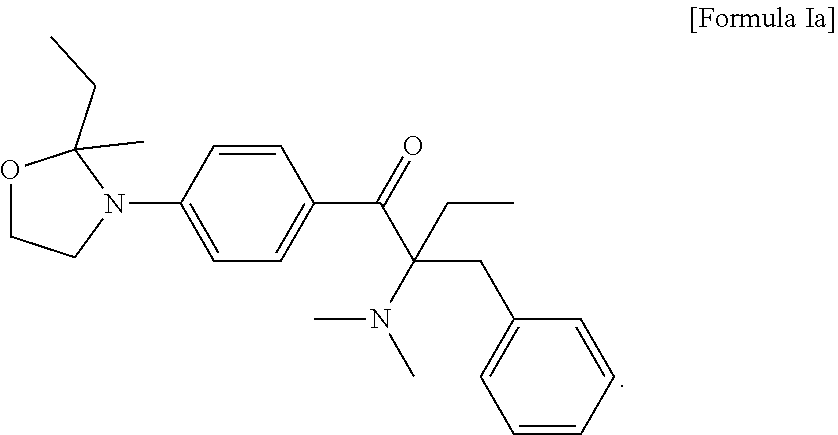
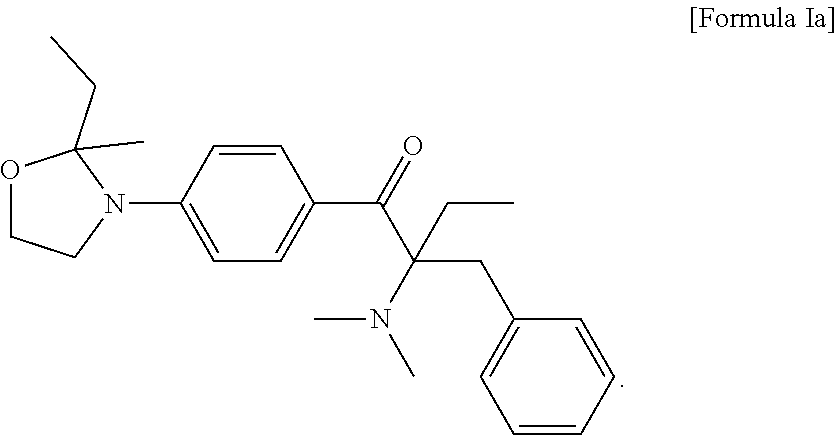
3. The compound of claim 2, which is represented by the following Formula Ia: ##STR00016##
4. A method of initiating a chemical reaction by using the liquid photo initiating compound of claim 1.
5. The method of claim 4, wherein, in Formula I, R.sub.1 and R.sub.2 are independently methyl, ethyl, n-propyl or isopropyl.
6. The method of claim 5, wherein the liquid photo initiating compound is represented by the following Formula Ia: ##STR00017##
7. A photopolymerizable composition, comprising: a first photoinitiator, which is the liquid photo initiating compound of claim 1; a photopolymerizable component; and an optional solvent.
8. The photopolymerizable composition of claim 7, wherein, in Formula I, R.sub.1 and R.sub.2 are independently methyl, ethyl, n-propyl or isopropyl.
9. The photopolymerizable composition of claim 8, wherein the liquid photo initiating compound is represented by the following Formula Ia: ##STR00018##
10. The photopolymerizable composition of claim 7, wherein the photopolymerizable component is an olefinic unsaturated monomer, an olefinic unsaturated oligomer, or a combination thereof.
11. The photopolymerizable composition of claim 10, wherein the photopolymerizable component is an acrylate-based monomer, an acrylate-based oligomer, or a combination thereof.
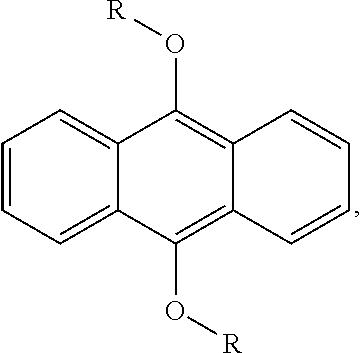
12. The photopolymerizable composition of claim 7, further comprising a second photoinitiator selected from the group consisting of acylphosphine oxides, 9,10-dialkyloxyanthracene, and combinations thereof.
13. The photopolymerizable composition of claim 7, further comprising a photosensitizer selected from the group consisting of benzophenones, thioxanthones, Michler's ketones, anthraquinone, and combinations thereof.
14. The photopolymerizable composition of claim 7, further comprising an additive selected from the group consisting of pigments, amine synergists, and combinations thereof.
15. The photopolymerizable composition of claim 7, wherein the content of the first photoinitiator ranges from 0.1 wt % to 15 wt % based on the total weight of the photopolymerizable composition.
Description
CLAIM FOR PRIORITY
This application claims the benefit of Taiwan Patent Application No. 107124654 filed on Jul. 17, 2018, the subject matters of which are incorporated herein in their entirety by reference.
BACKGROUND OF THE INVENTION
Field of the Invention
The present invention relates to a liquid photo initiating compound and photopolymerizable compositions using the same.
Descriptions of the Related Art
A photoinitiator is a substance that forms free radicals via Norrish Type I photo-fragmentation after absorbing the energy of visible or UV light, which can initiate polymerization of monomers or oligomers to provide a curing effect.
In 1990, Ciba Specialty Corporation introduced Irgacure.RTM. 369, which is the first photoinitiator specifically designed for dark color UV ink (related patent: U.S. Pat. No. 5,077,402). Irgacure.RTM. 369 is in solid form and its structure is shown below.
##STR00002##
Irgacure.RTM. 369 has an outstanding photo speed and other advantages like odorless prior to and after curing. However, it suffers from poor solubility in most UV inks and thus requires additional grinding and heating processes to facilitate dissolution. Even though, Irgacure.RTM. 369 is prone to precipitate when dosage is over 4 wt % and when handled in cold areas. Irgacure.RTM. 369 has recently been shown to have reproductive toxicity and thus is under radar in EU to be prohibited from use in food contact applications.
In order to improve the solubility issue, Ciba Specialty Corporation introduced another solid photoinitiator product, i.e., Irgacure.RTM. 379 (related patent: U.S. Pat. No. 7,247,659 B2). Irgacure.RTM. 379 does have improved solubility in some monomers and resins than Irgacure.RTM. 369. Still, it is not easy to use in its solid form that requires grinding and heating processes in order to dissolve into an ink composition. Furthermore, Irgacure.RTM. 379 also has reproductive toxicity.
##STR00003##
In recent years, the demand for low viscosity UV inks such as digital UV and flexo UV inks has increased significantly. A photoinitiator in liquid form with high solubility is therefore highly desired to the low viscosity ink as these inks do not provide sufficient shearing force to perform the grinding and dispersion processes for solid photoinitiators.
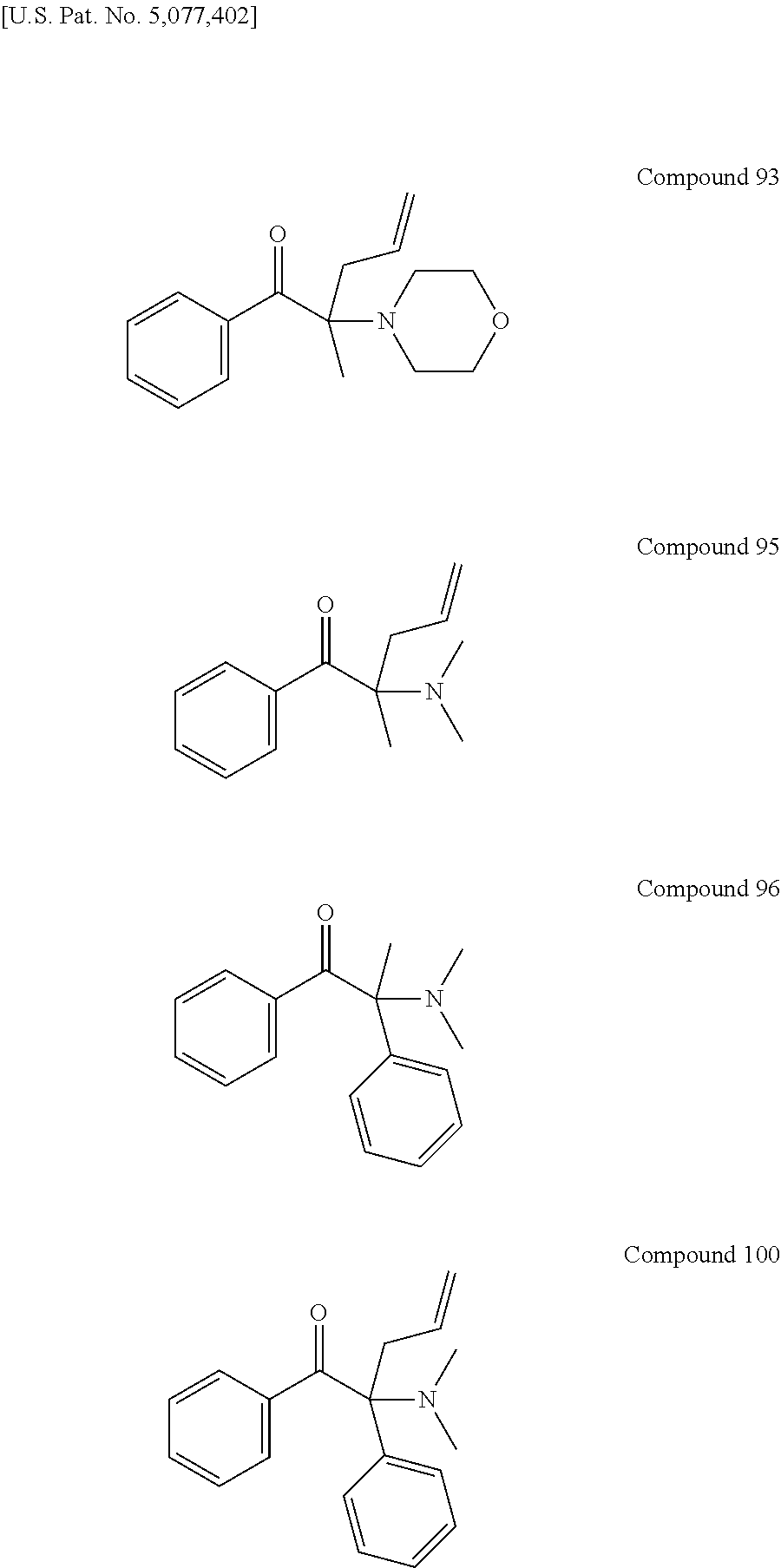
Examples of liquid photoinitiators include the following compounds 93, 95, 96 and 100 as disclosed in U.S. Pat. No. 5,077,402. But these compounds are not commercialized because their poor photospeeds as well as odor issues.
##STR00004##
TW 1277834 discloses a liquid photoinitiator in Example 3. Even though the liquid photoinitiator is odorless, it is not commercialized due to its poor photo speed which is about one tenth that of Irgacure.RTM. 369.
Example 3 of TW 1277834
##STR00005##
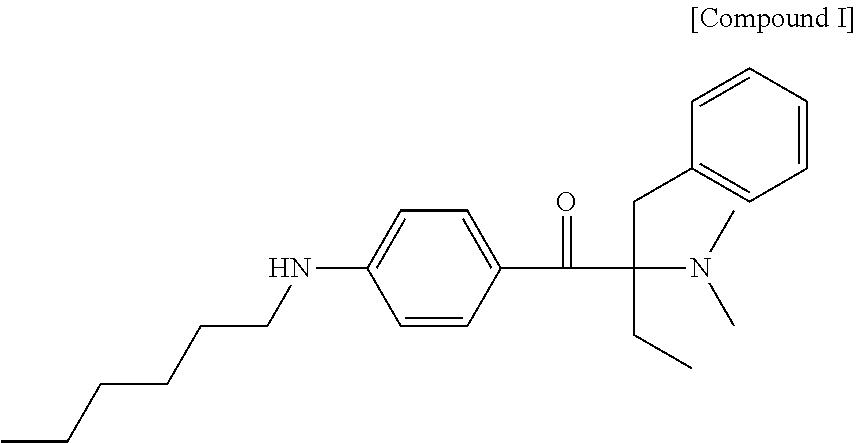
The following compound I is also a liquid photoinitiator and has a photo speed comparable to that of Irgacure.RTM. 369. Unfortunately, the compound I has a serious odor issue after curing.
##STR00006##
TW 1564276 also discloses a liquid photoinitiator, i.e., the following compound IIa, which is comparable to Irgacure.RTM. 369 in terms of photo speed and is odorless prior to or after curing. However, the compound IIa has high acute oral toxicity. Besides, it is in dark-brown color which is not satisfactory for some colors.
##STR00007##
Therefore, conventional liquid photoinitiators with individual defects for dark and black inks are far from satisfactory.
SUMMARY OF THE INVENTION
In view of the unsatisfaction of conventional photoinitiators, the present invention provides a photoinitiator derived from oxazolidine, which improves all the aforementioned defects associated conventional liquid photoinitiators for the black ink system as: (1) it becomes a liquid state at 50.degree. C.; (2) it has a photo speed comparable to that of Irgacure.RTM. 369; (3) it is odorless prior to and after curing; and (4) it does not have acute oral toxicity. Furthermore, its light color and high purity perfectly meet the requirements of the industry regarding liquid photoinitiators. Accordingly, the present invention involves at least the objectives described below.
It should be noted that neither U.S. Pat. No. 5,077,402 nor U.S. Pat. No. 7,732,504 B2 discloses the photoinitiator of the present invention, even though the photoinitiators disclosed in these patents may have an oxazolidine structure. Furthermore, persons having ordinary skill in the art cannot expect the advantages of the photoinitiator of the present invention based on the general teaching of U.S. Pat. Nos. 5,077,402 or 7,732,504 B2. The advantages include being liquid at a low temperature of 50.degree. C., high solubility in various monomers and oligomers, odorless, comparable photo speed to Irgacure.RTM. 369, and no acute oral toxicity.
An objective of the present invention is to provide a liquid photo initiating compound, which is represented by the following Formula .quadrature.:
##STR00008## wherein, in Formula I, R.sub.1 and R.sub.2 are independently H or C.sub.1-C.sub.3 alkyl, and R.sub.3 is H or methyl. Examples of C.sub.1-C.sub.3 alkyl include methyl, ethyl, n-propyl and isopropyl.
In some embodiments of the present invention, the aforementioned compound is represented by the following Formula Ia:
##STR00009##
Another objective of the present invention is to provide a method of initiating a chemical reaction by using the aforementioned liquid photo initiating compound.
Yet another objective of the present invention is to provide a photopolymerizable composition, comprising:
a first photoinitiator, which is the aforementioned liquid photo initiating compound;
a photopolymerizable component; and
an optional solvent.
In some embodiments of the present invention, the photopolymerizable component is an olefinic unsaturated monomer, an olefinic unsaturated oligomer, or a combination thereof. Examples of the olefinic unsaturated monomer include acrylate-based monomers. Examples of the olefinic unsaturated oligomer include acrylate-based oligomers.
In some embodiments of the present invention, the photopolymerizable composition further comprises a second photoinitiator selected from the group consisting of acylphosphine oxides, 9,10-dialkyloxyanthracene
##STR00010## wherein R is alkyl), and combinations thereof.
In some embodiments of the present invention, the photopolymerizable composition further comprises a photosensitizer selected from the group consisting of benzophenones, thioxanthones, Michler's ketones, anthraquinone, and combinations thereof.
In some embodiments of the present invention, the photopolymerizable composition further comprises an additive selected from the group consisting of pigments, amine synergists, and combinations thereof.
In some embodiments of the present invention, the content of the first photoinitiator ranges from 0.1 wt % to 15 wt % based on the total weight of the photopolymerizable composition.
To render the above objectives, technical features and advantages of the present invention more apparent, the present invention will be described in detail with reference to some embodiments hereinafter.
BRIEF DESCRIPTION OF THE DRAWINGS
Not applicable.
DESCRIPTION OF THE PREFERRED EMBODIMENT
Hereinafter, some specific embodiments of the present invention will be described in detail. However, without departing from the spirit of the present invention, the present invention may be embodied in various embodiments and should not be limited to the specific embodiments described in the specification.
Unless it is additionally explained, the expressions "a,", "an", "the," or the like recited in the specification (especially in the claims) should include both the singular and the plural forms.
Unless it is additionally explained, the term such as "first", "second" or the like is used to distinguish different elements or components, not terms supplying a numerical limit.
Unless it is additionally explained, the term "alkyl" recited in the specification (especially in the claims) includes linear, branched and/or cyclic alkyl groups.
Liquid Photo Initiating Compound
The liquid photo initiating compound of the present invention is represented by the following Formula .quadrature.:
##STR00011##
In formula I, R.sub.1 and R.sub.2 are independently H or C.sub.1-C.sub.3 alkyl, and R.sub.3 is H or methyl. In some embodiments of the present invention, R.sub.1 and R.sub.2 are independently methyl, ethyl, n-propyl or isopropyl, and R.sub.3 is H. In the appended examples, the first photoinitiator is represented by the following Formula Ia:
##STR00012##
The synthesis of the liquid photo initiating compound of the present invention will be described in the appended examples.
Uses of Liquid Photo Initiating Compound
The liquid photo initiating compound of the present invention is liquid at a low temperature of 50.degree. C. Therefore, it can be used as a liquid photoinitiator to trigger polymerization and crosslinking of photopolymerizable monomers or oligomers and thus, provide a curing effect. The liquid photo initiating compound of the present invention surprisingly shows properties quite different from conventional photoinitiators and has many advantages. Specifically, the liquid photo initiating compound of the present invention has excellent photo speed performance, excellent solubility and light color and is in liquid state at a low temperature of 50.degree. C. When used in a photopolymerizable system, the influence of the liquid photo initiating compound of the present invention on viscosity is very minor. The liquid photo initiating compound of the present invention also has long storage stability. Therefore, the liquid photo initiating compound of the present invention is particularly useful for low viscosity UV inks (e.g., an ink with a viscosity of less than 1000 cP), light color UV inks, color resists, black matrixes and solder masks. Furthermore, the liquid photo initiating compound of the invention has high purity that can meet the chemical substance registration regulations of nations of the world.
Accordingly, the present invention also provides a photopolymerizable composition, comprising a first photoinitiator, a photopolymerizable component and an optional solvent, wherein the first photoinitiator is the compound represented by Formula I.
The species of the photopolymerizable component is not particularly limited and can be any substance that is photopolymerizable in the presence of a photoinitiator, including the substance that is generally used in photopolymerizable systems, like UV inks. In some embodiments of the present invention, the photopolymerizable component is selected from the group consisting of olefinic unsaturated monomers, olefinic unsaturated oligomers and combinations thereof. Examples of the olefinic unsaturated monomer include but are not limited to acrylate-based monomers, and examples of the olefinic unsaturated oligomer include but are not limited to acrylate-based oligomers.
The solvent is optional and can be any solvent that dissolves or disperses the components of the composition but does not react with the components, including those generally used in photopolymerizable systems such as UV inks. Examples of the solvent include but are not limited to water; aliphatic hydrocarbons, such as dichloromethane, trichloromethane, tetrachloromethane, n-hexane, and cyclohexane; aromatic hydrocarbons, such as toluene, benzene, and xylene; ketones, such as acetone, methyl ethyl ketone, isobutyl ketone and cyclohexanone; esters, such as ethyl acetate and butyl acetate; alcohols, such as methanol, ethanol, n-propanol and isopropanol; and ethers, such as dimethylether, diethylether, and methylethylether.
In the photopolymerizable composition of the present invention, the amount of the first photoinitiator is not particularly limited and can be optionally adjusted by persons having ordinary skill in the art. To obtain a better photocuring efficacy, the amount of the photoinitiator is usually from 0.1 wt % to 15 wt %, such as 1 wt %, 2 wt %, 3 wt %, 4 wt %, 5 wt %, 6 wt %, 7 wt %, 8 wt %, or 9 wt %, based on the total weight of the photopolymerizable composition. However, the present invention is not limited thereto.
In addition to the first photoinitiator, the photopolymerizable composition of the present invention can optionally further include other components that are advantageous to the efficacy of photoinitiation, like photosensitizing components or other photo initiating components. Therefore, in some embodiments of the present invention, the photopolymerizable composition further comprises a second photoinitiator, a photosensitizer, or a combination thereof.
The second photoinitiator can be any photo initiating component other than the first photoinitiator. Examples of the second photoinitiator include but are not limited to acylphosphine oxides, 9,10-dialkyloxyanthracene, and combinations thereof. The species of the photosensitizer is not particularly limited. Examples of the photosensitizer include but are not limited to benzophenones, thioxanthones, Michler's ketones, anthraquinone, and combinations thereof. In the appended examples, 2,4-diethylthioxanthone (product name: Chivacure.RTM. DETX) is used as a second photosensitizer in the photopolymerizable composition. The amount of the second photoinitiator or the photosensitizer is not particularly limited and can be optionally adjusted by persons having ordinary skill in the art depending on the need.
The photopolymerizable composition of the present invention may optionally further comprise one or more additives to impart desired properties to the cured product of the composition. For example, a pigment can be added to provide color to the cured product, and an amine synergist can be added to improve curing performance, for example, to improve the surface curing speed of composition and to enhance the mirror surface effect after curing. Examples of the pigment include titanium oxide, carbon black, cadmium red, molybdate red, chrome yellow, cadmium yellow, titanium yellow, chromium oxide, cobalt titanate green, cerulean blue, copper blue, azo pigments, phthalocyanine pigments, quinacridone based pigments, isoindolinone based pigments, perylene based pigments, thio indigo based pigments, and metal complex pigments. As used herein, an amine synergist is well-known to persons having ordinary skill in the art and refers to a reactive amine that can promote the photoinitiation, such as a low-molecular weight tertiary amine. Examples of the amine synergist include but are not limited to methyldiethanolamine, dibutylethanolamine, triethylamine and triethanolamine. Commercially available products of the amine synergist include Chivacure.RTM. 115, Chivacure.RTM. EPD, Chivacure.RTM. OPD, etc. available from Chitec Technology Co., Ltd.
The present invention is further illustrated by the following embodiments.
EXAMPLES
Example 1: Preparation of Liquid Photo Initiating Compound Represented by Formula Ia
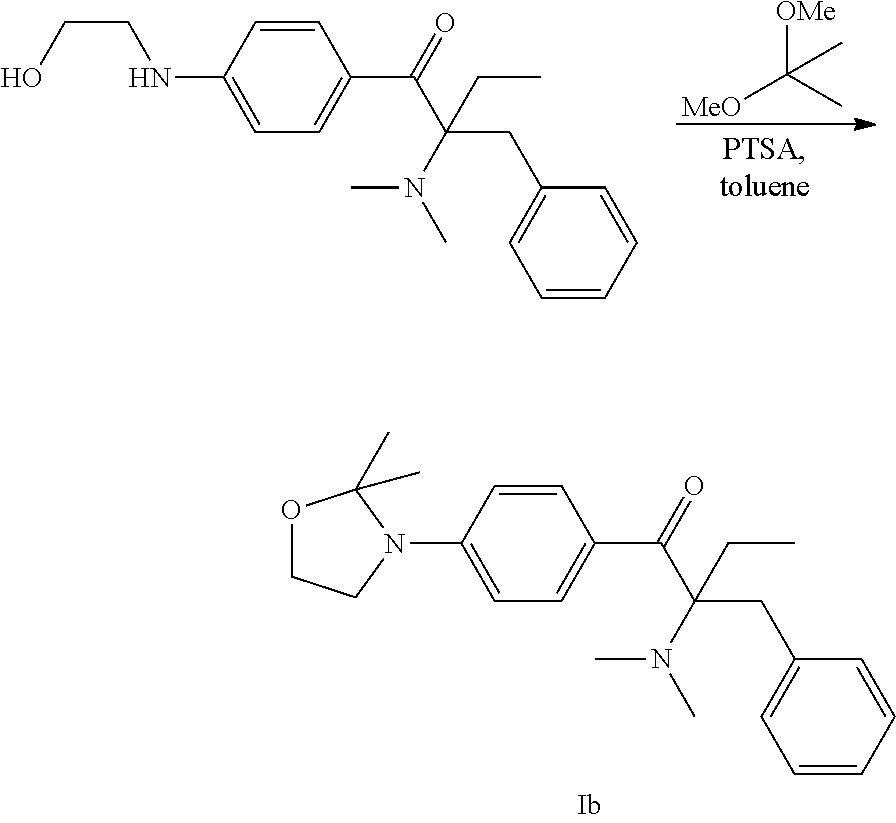
34 g 2-(dimethylamino)-1-[4-[(2-hydroxyethyl)amino]phenyl]-2-(phenylmethy- l)-1-butanone (CAS 862589-48-8); the synthesis method of it may refer to TW 1277834), 13.0 g 2,2-dimethoxybutane (CAS 3453-99-4), 0.17 g PTSA (p-toluenesulfonic acid), and 100 mL toluene were added in sequence to a 250 mL three-necked flask at room temperature and then the reaction as shown below was carried out at reflux. The by-product methanol produced by this reaction was removed by Dean-Stark evaporator.
##STR00013##
The reaction was monitored by High Performance Liquid Chromatography (HPLC). After the reaction was completed, the reaction product was cooled to room temperature and washed with 17 g pure water for three times. The organic layer was collected and then concentrated under vacuum to obtain the compound represented by Formula Ia (hereinafter "photo initiating compound Ia") as a yellowish and viscous liquid. The yield is 95%.
The photo initiating compound Ia was subjected to nuclear magnetic resonance analysis, and the results are as follows:
TABLE-US-00001 Nuclear .sup.1H NMR (500 MHz, CDCl.sub.3): 0.70 (t, J = 7.5 Hz, 3H), magnetic 0.82 (t, J = 7.5 Hz, 3H), 1.85-1.89 (m, 1H), 1.94-1.99 resonance (m, 1H), 2.04-2.09 (m, 1H), 2.13-2.18 (m, 1H), 2.36 analysis: (s, 6H), 3.16-3.23 (m, 2H), 3.18 (d, J = 14.0 Hz, 1H), 3.22 (d, J = 14.0 Hz, 1H), 4.06-4.10 (m, 1H), 4.12-4.26 (m, 1H), 6.60 (d, J = 9.0 Hz, 2H), 7.16-7.28 (m, 6H), 8.34 (d, J = 9.0 Hz, 2H) .sup.13C NMR (500 MHz, CDCl.sub.3): 7.7, 9.8, 23.8, 27.8, 31.0, 35.5, 39.1, 48.4, 63.2, 73.8, 96.2, 111.5, 125.8, 125.9, 127.9, 131.3, 132.2, 139.6, 147.1, 201.6
Example 2: Preparation of a Liquid Photo Initiating Compound Represented by Formula Ib
34 g 2-(dimethylamino)-1-[4-[((2-hydroxyethyl)amino]phenyl]-2-(phenylmeth- yl)-1-butanone, 12.0 g 2,2-dimethoxypropane (CAS 77-76-9), 0.17 g PTSA (p-toluenesulfonic acid), and 100 mL toluene were added to a 250 mL three-necked flask at room temperature in sequence and the reaction as shown below was carried out at a condition the same as the previous example.
##STR00014##
The reaction was monitored by High Performance Liquid Chromatography (HPLC). After the reaction was completed, the reaction product was extracted with 17 g pure water for three times. The organic layer was collected and then concentrated under vacuum to obtain the compound represented by Formula Ib (hereinafter referred to as "photo initiating compound Ib") as a yellowish and viscous liquid. The yield is 93%.
The photo initiating compound Ib was subjected to nuclear magnetic resonance analysis, and the results are as follows:
TABLE-US-00002 Nuclear .sup.1H NMR (500 MHz, CDCl.sub.3): 0.69 (t, J = 7.5Hz, 3H), magnetic 1.64 (s, 6H), 1.83-1.90 (m, 1H), 2.02-2.17 (m, 1H), resonance 2.36 (s, 6H), 3.17 (d, J = 14.0 Hz, 1H), 3.22 (d, J = analysis: 14.0 Hz, 1H), 3.53 (t, J = 6.5 Hz, 2H), 4.11 (t, J = 6.5 Hz, 2H), 6.60 (d, J = 9.0 Hz, 2H), 7.15-7.28 (m, 6H), 8.35 (d, J = 9.0 Hz, 2H) .sup.13C NMR (500 MHz, CDCl.sub.3): 9.8, 25.4, 27.8, 35.5, 39.1, 47.8, 62.9, 73.9, 93.9, 111.8, 125.8, 125.9, 127.9, 131.3, 132.2, 139.6, 147.0, 201.6
Example 3: Solubility Test
Mixtures I to III were prepared according to the composition provided below, and then 6 parts by weight of photoinitiator Trgacure.RTM. 369 (available from IGM), photoinitiator R-gen.RTM. 919 (available from Chitec Technology Co., Ltd.) or photo initiating compound Ia was added to each of the mixtures I to III. The obtained mixtures were ultrasonicated for 1 hour at room temperature, and the dissolution of each of Irgacure.RTM. 369, R-gen.RTM. 919 and photo initiating compound Ia was observed and described in the following Table 1.
TABLE-US-00003 Mixture I: a mixture of 50 parts by weight of 1,6-hexanediol diacrylate (product name: EM221, Eternal Materials Co., Ltd.) and 50 parts by weight of polyester tetraacrylate (product name: Oligomer 6325-100, Eternal Materials Co., Ltd.) with a viscosity of 41.2 cP Mixture II: a mixture of 50 parts by weight of trimethylolpropane triacrylate (product name: EM231, Eternal Materials Co., Ltd.) and 50 parts by weight of polyester tetraacrylate (product name: Oligomer 6325-100, Eternal Materials Co., Ltd.) with a viscosity of 349.5 cP Mixture III: a mixture of 50 parts by weight of ethoxylated pentaerythritol tetraacrylate (product name: EM2411, Eternal Materials Co., Ltd.) and 50 parts by weight of polyester tetraacrylate (product name: Oligomer 6325-100, Eternal Materials Co., Ltd.) with a viscosity of 433.0 cP
TABLE-US-00004 TABLE 1 Dissolution of Irgacure .RTM. 369, R-gen .RTM. 919 and photo initiating compound Ia after one-hour ultrasonication at room temperature Photoinitiator Mixture I Mixture II Mixture III Irgacure .RTM. 369 Completely Only a small part Only a small part dissolved was dissolved was dissolved R-gen .RTM. 919 Completely Completely Completely dissolved dissolved dissolved Photo initiating Completely Completely Completely compound Ia dissolved dissolved dissolved
As shown in Table 1, the photo initiating compound Ia completely dissolved in each of the three mixtures after one-hour ultrasonication at room temperature. The results indicate that the photo initiating compound Ia is much better than the solid photoinitiator Irgacure.RTM. 369 and comparable to R-gen.RTM. 919 in terms of solubility.
Example 4: Viscosity Variation Test
4 parts by weight of photoinitiators Irgacure.RTM. 369, R-gen.RTM. 919, and photo initiating compound Ia were individually added to each of the mixtures I to III, and the obtained mixtures were exposed to a 0.degree. C. atmosphere for 7 days. The viscosities before and after the exposure were measured and tabulated in the following Table 2.
TABLE-US-00005 TABLE 2 Viscosities (cP) before and after exposure to 0.degree. C. atmosphere for 7 days Mixture I Mixture II Mixture III Photoinitiator Initial 7 days Initial 7 days Initial 7 days None 41.2 50.1 349.5 401.0 433.0 548.0 Irgacure.RTM. 369 52.7 60.4 385.0 Precipitate 487.0 Precipitate R-gen.RTM. 919 42.2 51.0 354.0 408.0 445.5 544.0 Photo initiating 41.6 51.2 351.3 406.7 433.1 547.8 compound Ia
As shown in Table 2, the photo initiating compound IA has the lowest influence on the viscosity of the mixture. In addition, after the exposure to a 0.degree. C. atmosphere for 7 days, the viscosity variation of the mixture with the photo initiating compound Ia is similar to that of the mixture without photoinitiator, and no precipitate was observed in the mixture with the photo initiating compound Ia. The results indicate that the photo initiating compound Ia is better than Irgacure.RTM. 369 in terms of solubility and resin compatibility and therefore is very useful.
Example 5: Photo Speed Performance Test
The black flexo formulation containing Oligomer 6325-100, EM2411 and carbon black (viscosity: 1750 cP) was mixed with a photoinitiator to carry out the photo speed performance test. Specifically, 100 parts by weight of the black flexo formulation and 2 parts by weight of Chivacure.RTM. DETX (available from Chitec Technology Co., Ltd.) were mixed, and then 6 parts by weight of Irgacure.RTM. 369, R-gen.RTM. 998 (available from Chitec Technology Co., Ltd.), R-gen.RTM. 919, or photo initiating compound Ia was added to the obtained mixture. The resultant mixture was coated on a printable pearlescent film (wet film thickness: 25.15 .mu.m); and the coated printable pearlescent film was exposed to an UV light (Fusion F300, D-bulb, 300 W/inch) for curing. The photo speed performance was observed and tabulated in the following Table 3.
TABLE-US-00006 TABLE 3 Photo speed performance of photoinitiator Photo initiating compound Photoinitiator Irgacure .RTM. 369 R-gen .RTM. 998 R-gen .RTM. 919 Ia Photo speed 120 70 120 120 (m/minute)
Theoretically, photoinitiators with electron withdrawing groups, such as the photo initiating compound Ia, usually have a lower photo speed due to the blue shift effect. However, as shown in Table 3, the photo initiating compound Ia surprisingly has an outstanding photo speed which is comparable to those of Irgacure.RTM. 369 and R-gen.RTM. 919 and significantly better than that of R-gen.RTM. 998.
Example 6: Photoinitiator Dosage Test
The commercially available black flexo ink without a photoinitiator was mixed with a photoinitiator to carry out the photo speed performance test, and the effect of the amount of photo initiating compound Ia on the photo speed performance was evaluated. Specifically, 100 parts by weight of the black flexo formulation and 2 parts by weight of Chivacure.RTM. DETX (photoinitiator; Chitec Technology Co., Ltd.) were mixed, and then a specific amount of Irgacure.RTM. 369 or photo initiating compound Ia as shown in Table 4 was added to the obtained mixture. The resultant mixture was coated on a PET film with primer (means for improving adhesion) (dry film thickness: 5 .mu.m). The coated PET film was exposed to an UV light (wavelength: 365 nm; 4 W/inch) for curing. The photo speed performance in each case was observed and tabulated in the following Table 4.
TABLE-US-00007 TABLE 4 Photo speed performance of photoinitiator in different amounts Amounts Curing performance (parts by (times of UV light treatment) weight) 1 2 3 4 5 Irgacure.RTM. 369 4 X X X .DELTA. O 8 Incomplete dissolution Photo 4 X X X .DELTA. O initiating 8 .DELTA. O compound Ia 12 O
TABLE-US-00008 Description for curing results: X Not tack-free .DELTA. Tack-free but not through-cure .largecircle. Tack-free and through-cure
As shown in Table 4, the tested black flexo ink is an ink system that is difficult to be photocured, and the photocuring efficiency can be improved by increasing the amount of the photoinitiator. However, when 8 parts by weight of Irgacure.RTM. 369 was added, like the result of the solubility test, the photoinitiator could not be completely dissolved in the ink system, making it impossible to carry out a photo speed test. By contrast, when the amount of the photo initiating compound Ia was increased, no solubility issue was observed even if the amount reaches 12 parts by weight. Furthermore, increasing the amount of the photo initiating compound Ia from 4 parts by weight to 8 parts by weight can significantly improve the photocuring speed; and only one time of UV light treatment was required to achieve sufficient curing when the amount of the photo initiating compound Ia was 12 parts by weight.
Example 7: Acute Oral Toxicity Test
The median lethal dose (LD50) test was carried out for each of Irgacure.RTM. 369, R-gen.RTM. 998, R-gen.RTM. 919 and photo initiating compound Ia in accordance with Test Guidelines 423: acute oral toxicity, Organization for Economic Co-operation and Development (OECD).
The results are tabulated in the following Table 5.
TABLE-US-00009 TABLE 5 Median lethal dose (LD50) of photoinitiator Photo initiating compound Photoinitiator Irgacure .RTM. 369 R-gen .RTM. 998 R-gen .RTM. 919 Ia LD50 >5000 2500 500 >5000 (mg/kg)
As shown in Table 5, the median lethal dose (LD50) of the photo initiating compound Ia is comparable with that of Irgacure.RTM. 369, both exceeding 5000 mg/kg, which is considered as no acute oral toxicity.
As can be seen from the results of Examples 1 to 7, the liquid photo initiating compound of the present invention has excellent photo speed performance, excellent solubility, light color and no acute oral toxicity. The liquid photo initiating compound of the present invention is in liquid state at a low temperature of 50.degree. C. When being used in a photopolymerizable system, the influence of the liquid photo initiating compound of the present invention on viscosity is very minor. The liquid photo initiating compound of the present invention also has long storage stability. Therefore, the liquid photo initiating compound of the present invention is particularly useful for low viscosity UV inks, light color UV inks, color resists, black matrixes and solder masks.
The above examples are used to illustrate the principle and efficacy of the present invention and show the inventive features thereof. People skilled in this field may proceed with a variety of modifications and replacements based on the disclosures and suggestions of the invention as described without departing from the principle and spirit thereof. Therefore, the scope of protection of the present invention is that as defined in the claims as appended.
* * * * *
C00001

C00002

C00003

C00004

C00005

C00006

C00007

C00008

C00009

C00010

C00011

C00012

C00013

C00014
C00015

C00016
C00017
C00018

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.