Hetero-cyclic compound and organic light emitting device using the same
Park , et al. A
U.S. patent number 10,381,577 [Application Number 15/321,397] was granted by the patent office on 2019-08-13 for hetero-cyclic compound and organic light emitting device using the same. This patent grant is currently assigned to HEESUNG MATERIAL LTD.. The grantee listed for this patent is HEESUNG MATERIAL LTD.. Invention is credited to Dae-Hyuk Choi, Jin-Seok Choi, Dong-Jun Kim, Joo-Dong Lee, Yun-Ji Lee, Jae-Yeol Ma, Han-Kook Oh, Geon-Yu Park.










View All Diagrams
| United States Patent | 10,381,577 |
| Park , et al. | August 13, 2019 |
Hetero-cyclic compound and organic light emitting device using the same
Abstract
The present application provides a hetero-cyclic compound which may significantly improve the lifetime, efficiency, electrochemical stability, and thermal stability of an organic light emitting device, and an organic light emitting device in which the hetero-cyclic compound is contained in an organic compound layer.
| Inventors: | Park; Geon-Yu (Osan-si, KR), Oh; Han-Kook (Osan-si, KR), Lee; Yun-Ji (Osan-si, KR), Ma; Jae-Yeol (Yongin-si, KR), Kim; Dong-Jun (Yongin-si, KR), Choi; Jin-Seok (Suwon-si, KR), Choi; Dae-Hyuk (Yongin-si, KR), Lee; Joo-Dong (Seongnam-si, KR) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | HEESUNG MATERIAL LTD. (Yongin,
KR) |
||||||||||
| Family ID: | 58108986 | ||||||||||
| Appl. No.: | 15/321,397 | ||||||||||
| Filed: | July 27, 2016 | ||||||||||
| PCT Filed: | July 27, 2016 | ||||||||||
| PCT No.: | PCT/KR2016/008189 | ||||||||||
| 371(c)(1),(2),(4) Date: | December 22, 2016 | ||||||||||
| PCT Pub. No.: | WO2017/018795 | ||||||||||
| PCT Pub. Date: | February 02, 2017 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20170213988 A1 | Jul 27, 2017 | |
Foreign Application Priority Data
| Jul 27, 2015 [KR] | 10-2015-0106063 | |||
| May 11, 2016 [KR] | 10-2016-0057665 | |||
| May 13, 2016 [KR] | 10-2016-0059084 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 495/04 (20130101); C07D 333/76 (20130101); H01L 51/0071 (20130101); H01L 51/50 (20130101); H01L 51/0074 (20130101); C07D 487/04 (20130101); C09K 11/06 (20130101); H01L 51/00 (20130101); C07D 209/82 (20130101); C07D 491/04 (20130101); C09K 11/025 (20130101); C07D 409/14 (20130101); H01L 51/0072 (20130101); H01L 51/0067 (20130101); C09K 2211/1044 (20130101); H01L 51/0081 (20130101); C09K 2211/1007 (20130101); H01L 51/0085 (20130101); C09K 2211/1059 (20130101); H01L 51/5016 (20130101); H01L 2251/552 (20130101); C09K 2211/1092 (20130101) |
| Current International Class: | H01L 51/50 (20060101); C07D 209/82 (20060101); C07D 333/76 (20060101); C07D 495/04 (20060101); C09K 11/06 (20060101); C09K 11/02 (20060101); C07D 491/04 (20060101); C07D 487/04 (20060101); C07D 409/14 (20060101); H01L 51/00 (20060101) |
References Cited [Referenced By]
U.S. Patent Documents
| 4356429 | October 1982 | Tang |
| 2011/0006670 | January 2011 | Katakura et al. |
| 2011/0260138 | October 2011 | Xia |
| 2015/0014649 | January 2015 | Ma et al. |
| 2015/0179958 | June 2015 | Otsu |
| 2015/0322337 | November 2015 | Hattori et al. |
| 2016/0072078 | March 2016 | Lee et al. |
| 2017/0331052 | November 2017 | Park et al. |
| 2018/0037546 | February 2018 | Sugino |
| 3243820 | Nov 2017 | EP | |||
| 2009-267255 | Nov 2009 | JP | |||
| 2012-222268 | Nov 2012 | JP | |||
| 2013-16717 | Jan 2013 | JP | |||
| 2014-017494 | Jan 2014 | JP | |||
| 2016-51901 | Apr 2016 | JP | |||
| 2016-149473 | Aug 2016 | JP | |||
| 10-2014-0122929 | Oct 2014 | KP | |||
| 10-2011-0112098 | Oct 2011 | KR | |||
| 10-2013-0100236 | Sep 2013 | KR | |||
| WO 2011/004639 | Jan 2011 | WO | |||
| WO 2011/126224 | Oct 2011 | WO | |||
| WO 2013/168534 | Nov 2013 | WO | |||
| WO 2014/091958 | Jun 2014 | WO | |||
Other References
|
Bin, J.K. et al, "New sulfur-containing host materials for blue phosphorescent organic light-emitting diodes," J. Mater. Chem., 2012, vol. 22, pp. 21720-21726. cited by applicant . Office Action issued in Korean Patent Application No. 10-2016-0057665, dated Oct. 5, 2016. cited by applicant . Office Action issued in Korean Patent Application No. 10-2016-0059084, dated Aug. 8, 2016. cited by applicant . Extended European Search Report for European Application No. 16830828.6, dated Jan. 17, 2019. cited by applicant. |
Primary Examiner: Clark; Gregory D
Attorney, Agent or Firm: Birch, Stewart, Kolasch & Birch, LLP
Claims
The invention claimed is:
1. A hetero-cyclic compound represented by the following Chemical Formula 1: ##STR00296## in Chemical Formula 1, L1 and L2 are the same as or different from each other, and each independently a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group, Ar1 is selected from the group consisting of a substituted or unsubstituted pyridine group; a substituted or unsubstituted pyrimidine group; a substituted or unsubstituted triazine group; a substituted or unsubstituted quinoline group; a substituted or unsubstituted quinazoline group; a substituted or unsubstituted phenanthroline group; and a substituted or unsubstituted benzoimidazole group, Ar2 is represented by any one of the following Chemical Formulae 3 and 4, ##STR00297## in Chemical Formulae 3 and 4, Y1 to Y4 are the same as or different from each other, and each independently a substituted or unsubstituted C.sub.6 to C.sub.60 aromatic hydrocarbon ring; or a substituted or unsubstituted C.sub.2 to C.sub.60 aromatic hetero ring, R1 to R7 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, and R, R', and R'' are the same as or different from each other, and are each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
2. The hetero-cyclic compound of claim 1, wherein Chemical Formula 3 is represented by any one of the following structural formulae: ##STR00298## in the structural formulae, X1 to X6 are the same as or different from each other, and each independently NR, S, O, or CR'R'', R8 to R14 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and m is an integer of 0 to 8, and n, o, p, q, r, and s are each independently an integer of 0 to 6.
3. The hetero-cyclic compound of claim 1, wherein Chemical Formula 4 is represented by any one of the following structural formulae: ##STR00299## in the structural formulae, X7 and X8 are the same as or different from each other, and each independently NR, S, O, or CR'R'', R15 to R18 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, and R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and t is an integer of 0 to 7.
4. The hetero-cyclic compound of claim 1, wherein Chemical Formula 1 is represented by any one of the following Chemical Formulae 5 to 10: ##STR00300## ##STR00301## in Chemical Formulae 5 to 10, the definitions of R1 to R6, L1, and Ar1 are the same as those in Chemical Formula 1, X1, X4, and X5 are the same as or different from each other, and each independently NR, S, O, or CR'R'', R8, R9, R12, R13, and R16 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and m is an integer of 0 to 8, n, q, and r are each independently an integer of 0 to 6, and t is an integer of 0 to 7.
5. The hetero-cyclic compound of claim 1, wherein R1 to R6 of Chemical Formula 1 are each independently hydrogen or deuterium.
6. The hetero-cyclic compound of claim 2, wherein R8 to R14 of the structural formulae are each independently hydrogen; deuterium; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
7. The hetero-cyclic compound of claim 3, wherein R5 to R18 of the structural formulae are each independently hydrogen; deuterium; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
8. The organic light emitting device of claim 1, wherein Chemical Formula 1 is represented by any one of the following compounds: ##STR00302## ##STR00303## ##STR00304## ##STR00305## ##STR00306## ##STR00307## ##STR00308## ##STR00309## ##STR00310## ##STR00311## ##STR00312## ##STR00313## ##STR00314## ##STR00315## ##STR00316## ##STR00317## ##STR00318## ##STR00319## ##STR00320## ##STR00321## ##STR00322## ##STR00323## ##STR00324## ##STR00325## ##STR00326## ##STR00327## ##STR00328## ##STR00329## ##STR00330## ##STR00331## ##STR00332## ##STR00333## ##STR00334## ##STR00335## ##STR00336## ##STR00337## ##STR00338## ##STR00339## ##STR00340## ##STR00341## ##STR00342## ##STR00343## ##STR00344## ##STR00345## ##STR00346## ##STR00347## ##STR00348## ##STR00349## ##STR00350## ##STR00351## ##STR00352## ##STR00353## ##STR00354## ##STR00355## ##STR00356## ##STR00357## ##STR00358## ##STR00359##
9. An organic light emitting device comprising: a positive electrode; a negative electrode; and one or more organic material layers provided between the positive electrode and the negative electrode, wherein one or more layers of the organic material layers comprise the hetero-cyclic compound of claim 1.
10. The organic light emitting device of claim 9, wherein the organic material layer comprise at least one layer of a hole blocking layer, an electron injection layer, and an electron transport layer, and at least one layer of the hole blocking layer, the electron injection layer, and the electron transport layer comprises the hetero-cyclic compound.
11. The organic light emitting device of claim 9, wherein the organic material layer comprises a light emitting layer, and the light emitting layer comprises the hetero-cyclic compound.
12. The organic light emitting device of claim 9, wherein the organic material layer comprises one or more layers of a hole injection layer, a hole transport layer, and a layer which injects and transports holes simultaneously, and one layer of the layers comprises the hetero-cyclic compound.
13. The organic light emitting device of claim 9, wherein the organic material layer comprising the hetero-cyclic compound further comprises a compound represented by the following Chemical Formula 2: ##STR00360## in Chemical Formula 2, R1' to R4' are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, L1' is a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group, Ar1' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including at least one of S and O, Ar2' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and m', p' and q' are each independently an integer of 0 to 4, and n' is an integer of 0 to 2.
14. The organic light emitting device of claim 13, wherein Chemical Formula 2 is represented by any one of the following Chemical Formulae 11 to 22: ##STR00361## ##STR00362## in Chemical Formulae 11 to 22, the definitions of L1, Ar1, Ar2, R1 to R4, m, n, p, and q are the same as those in Chemical Formula 2.
15. The organic light emitting device of claim 13, wherein Chemical Formula 2 is represented by any one of the following compounds: ##STR00363## ##STR00364## ##STR00365## ##STR00366## ##STR00367## ##STR00368## ##STR00369## ##STR00370## ##STR00371## ##STR00372## ##STR00373## ##STR00374## ##STR00375## ##STR00376## ##STR00377## ##STR00378## ##STR00379## ##STR00380## ##STR00381## ##STR00382## ##STR00383## ##STR00384## ##STR00385## ##STR00386## ##STR00387## ##STR00388## ##STR00389## ##STR00390## ##STR00391## ##STR00392## ##STR00393## ##STR00394## ##STR00395## ##STR00396## ##STR00397## ##STR00398## ##STR00399## ##STR00400##
16. A composition for an organic material layer of an organic light emitting device, comprising: both a hetero-cyclic compound represented by the following Chemical Formula 1 and a compound represented by the following Chemical Formula 2: ##STR00401## in Chemical Formulae 1 and 2, L1 and L2 are the same as or different from each other, and each independently a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group, Ar1 is a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group comprising at least one N, Ar2 is represented by any one of the following Chemical Formulae 3 and 4, ##STR00402## Y1 to Y4 are the same as or different from each other, and each independently a substituted or unsubstituted C.sub.6 to C.sub.60 aromatic hydrocarbon ring; or a substituted or unsubstituted C.sub.2 to C.sub.60 aromatic hetero ring, R1 to R7 and R1' to R4' are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, L1' is a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group, Ar1' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including at least one of S and O, Ar2' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, m', p' and q' are each independently an integer of 0 to 4, and n' is an integer of 0 to 2.
17. The composition for the organic material layer of the organic light emitting device of claim 16, wherein a weight ratio of the hetero-cyclic compound represented by Chemical Formula 1:the compound represented by Chemical Formula 2 in the composition is 1:10 to 10:1.
Description
TECHNICAL FIELD
This application claims priority to and the benefit of Korean Patent Application Nos. 10-2015-0106063, 10-2016-0057665, and 10-2016-0059084 filed in the Korean Intellectual Property Office on Jul. 27, 2015, May 11, 2016, and May 13, 2016, respectively, the entire contents of which are incorporated herein by reference.
The present application relates to a hetero-cyclic compound and an organic light emitting device using the same.
BACKGROUND ART
An electroluminescence device is a kind of self-emitting type display device, and has an advantage in that the viewing angle is wide, the contrast is excellent, and the response speed is fast.
An organic light emitting device has a structure in which an organic thin film is disposed between two electrodes. When a voltage is applied to an organic light emitting device having the structure, electrons and holes injected from the two electrodes combine with each other in an organic thin film to make a pair, and then, emit light while being extinguished. The organic thin film may be composed of a single layer or multi layers, if necessary.
A material for the organic thin film may have a light emitting function, if necessary. For example, as the material for the organic thin film, it is also possible to use a compound, which may itself constitute a light emitting layer alone, or it is also possible to use a compound, which may serve as a host or a dopant of a host-dopant-based light emitting layer. In addition, as a material for the organic thin film, it is also possible to use a compound, which may perform a function such as hole injection, hole transport, electron blocking, hole blocking, electron transport or electron injection.
In order to improve the performance, lifetime, or efficiency of the organic light emitting device, there is a continuous need for developing a material for an organic thin film.
DISCLOSURE
Technical Problem
It is necessary to perform studies on an organic light emitting device including a compound having a chemical structure, which may satisfy conditions required for a material which is available for the organic light emitting device, for example, appropriate energy levels, electrochemical stability, thermal stability, and the like, and may perform various functions required for the organic light emitting device according to the substituent.
Technical Solution
An exemplary embodiment of the present application provides a hetero-cyclic compound represented by the following Chemical Formula 1:
##STR00001##
In Chemical Formula 1,
L1 and L2 are the same as or different from each other, and each independently a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group,
Ar1 is a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including at least one N.
Ar2 is represented by any one of the following Chemical Formulae 3 and 4,
##STR00002##
in Chemical Formulae 3 and 4,
Y1 to Y4 are the same as or different from each other, and each independently a substituted or unsubstituted C.sub.6 to C.sub.60 aromatic hydrocarbon ring; or a substituted or unsubstituted C.sub.2 to C.sub.60 aromatic hetero ring,
R1 to R7 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, and
R, R', and R'' are the same as or different from each other, and are each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
Further, another exemplary embodiment of the present application provides an organic light emitting device including a positive electrode, a negative electrode, and one or more organic material layers provided between the positive electrode and the negative electrode, in which one or more layers of the organic material layers include the hetero-cyclic compound represented by Chemical Formula 1.
In addition, still another exemplary embodiment of the present application provides a composition for an organic material layer of an organic light emitting device, which includes both the hetero-cyclic compound represented by Chemical Formula 1 and a compound represented by the following Chemical Formula 2.
##STR00003##
In Chemical Formula 2,
R1' to R4' are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring,
L1' is a direct bond or a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group,
Ar1' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including at least one of S and O,
Ar2' is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group,
R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group,
m', p' and q' are each independently an integer of 0 to 4, and
n' is an integer of 0 to 2.
Advantageous Effects
A hetero-cyclic compound according to an exemplary embodiment of the present application may be used as a material for an organic material layer of an organic light emitting device. The hetero-cyclic compound may be used as a material for a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer, an electron injection layer, a charge generation layer, and the like in an organic light emitting device. In particular, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for an electron transport layer, a hole transport layer, or a light emitting layer of the organic light emitting device. In addition, when the hetero-cyclic compound represented by Chemical Formula 1 is used for the organic light emitting device, the driving voltage of the device may be lowered, the light efficiency of the device may be improved, and the lifetime characteristics of the device may be improved by the thermal stability of the compound.
Furthermore, the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by chemical Formula 2 may be used simultaneously as a material for a light emitting layer of an organic light emitting device. In addition, when the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2 are simultaneously used for the organic light emitting device, the driving voltage of the device may be lowered, the light efficiency of the device may be improved, and the lifetime characteristics of the device may be improved by the thermal stability of the compound.
DESCRIPTION OF DRAWINGS
FIGS. 1 to 3 each are views schematically illustrating a stacking structure of an organic light emitting device according to an exemplary embodiment of the present application.
FIG. 4 illustrates a measurement graph of LTPL of Compound 1-2 at a wavelength of 363 nm.
FIG. 5 illustrates a measurement graph of PL of Compound 1-2 at a wavelength of 238 nm.
FIG. 6 illustrates a UV absorption spectrum of Compound 1-2.
FIG. 7 illustrates a measurement graph of LTPL of Compound 1-11 at a wavelength of 339 nm.
FIG. 8 illustrates a measurement graph of PL of Compound 1-11 at a wavelength of 234 nm.
FIG. 9 illustrates a UV absorption spectrum of Compound 1-11.
FIG. 10 illustrates a measurement graph of LTPL of Compound 1-23 at a wavelength of 241 nm.
FIG. 11 illustrates a measurement graph of PL of Compound 1-23 at a wavelength of 241 nm.
FIG. 12 illustrates a UV absorption spectrum of Compound 1-23.
FIG. 13 illustrates a measurement graph of LTPL of Compound 1-27 at a wavelength of 340 nm.
FIG. 14 illustrates a measurement graph of PL of Compound 1-27 at a wavelength of 241 nm.
FIG. 15 illustrates a UV absorption spectrum of Compound 1-27.
FIG. 16 illustrates a measurement graph of LTPL of Compound 1-33 at a wavelength of 291 nm.
FIG. 17 illustrates a measurement graph of PL of Compound 1-33 at a wavelength of 239 nm.
FIG. 18 illustrates a UV absorption spectrum of Compound 1-33.
FIG. 19 illustrates a measurement graph of LTPL of Compound 1-39 at a wavelength of 259 nm.
FIG. 20 illustrates a measurement graph of PL of Compound 1-39 at a wavelength of 259 nm.
FIG. 21 illustrates a UV absorption spectrum of Compound 1-39.
FIG. 22 illustrates a measurement graph of LTPL of Compound 1-41 at a wavelength of 260 nm.
FIG. 23 illustrates a measurement graph of PL of Compound 1-41 at a wavelength of 260 nm.
FIG. 24 illustrates a UV absorption spectrum of Compound 1-41.
FIG. 25 illustrates a measurement graph of LTPL of Compound 1-65 at a wavelength of 361 nm.
FIG. 26 illustrates a measurement graph of PL of Compound 1-65 at a wavelength of 235 nm.
FIG. 27 illustrates a UV absorption spectrum of Compound 1-65.
FIG. 28 illustrates a measurement graph of LTPL of Compound 1-66 at a wavelength of 360 nm.
FIG. 29 illustrates a measurement graph of PL of Compound 1-66 at a wavelength of 307 nm.
FIG. 30 illustrates a UV absorption spectrum of Compound 1-66.
FIG. 31 illustrates a measurement graph of LTPL of Compound 1-67 at a wavelength of 361 nm.
FIG. 32 illustrates a measurement graph of PL of Compound 1-67 at a wavelength of 266 nm.
FIG. 33 illustrates a UV absorption spectrum of Compound 1-67.
FIG. 34 illustrates a measurement graph of LTPL of Compound 1-69 at a wavelength of 344 nm.
FIG. 35 illustrates a measurement graph of PL of Compound 1-69 at a wavelength of 308 nm.
FIG. 36 illustrates a UV absorption spectrum of Compound 1-69.
FIG. 37 illustrates a measurement graph of LTPL of Compound 1-70 at a wavelength of 344 nm.
FIG. 38 illustrates a measurement graph of PL of Compound 1-70 at a wavelength of 267 nm.
FIG. 39 illustrates a UV absorption spectrum of Compound 1-70.
FIG. 40 illustrates a measurement graph of LTPL of Compound 1-71 at a wavelength of 344 nm.
FIG. 41 illustrates a measurement graph of PL of Compound 1-71 at a wavelength of 241 nm.
FIG. 42 illustrates a UV absorption spectrum of Compound 1-71.
FIG. 43 illustrates a measurement graph of LTPL of Compound 1-78 at a wavelength of 361 nm.
FIG. 44 illustrates a measurement graph of PL of Compound 1-78 at a wavelength of 263 nm.
FIG. 45 illustrates a UV absorption spectrum of Compound 1-78.
FIG. 46 illustrates a measurement graph of LTPL of Compound 1-82 at a wavelength of 344 nm.
FIG. 47 illustrates a measurement graph of PL of Compound 1-82 at a wavelength of 307 nm.
FIG. 48 illustrates a UV absorption spectrum of Compound 1-82.
FIG. 49 illustrates a measurement graph of LTPL of Compound 1-84 at a wavelength of 363 nm.
FIG. 50 illustrates a measurement graph of PL of Compound 1-84 at a wavelength of 298 nm.
FIG. 51 illustrates a UV absorption spectrum of Compound 1-84.
FIG. 52 illustrates a measurement graph of LTPL of Compound 1-99 at a wavelength of 355 nm.
FIG. 53 illustrates a measurement graph of PL of Compound 1-99 at a wavelength of 355 nm.
FIG. 54 illustrates a UV absorption spectrum of Compound 1-99.
EXPLANATION OF REFERENCE NUMERALS AND SYMBOLS
100: Substrate 200: Positive electrode 300: Organic material layer 301: Hole injection layer 302: Hole transport layer 303: Light emitting layer 304: Hole blocking layer 305: Electron transport layer 306: Electron injection layer 400: Negative electrode
BEST MODE
Hereinafter, the present application will be described in detail.
A hetero-cyclic compound according to an exemplary embodiment of the present application is represented by Chemical Formula 1. More specifically, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for an organic material layer of an organic light emitting device by the structural characteristics of the core structure and the substituent as described above.
In Chemical Formulae 3 and 4, * denotes a position to be linked to L2 of Chemical Formula 1.
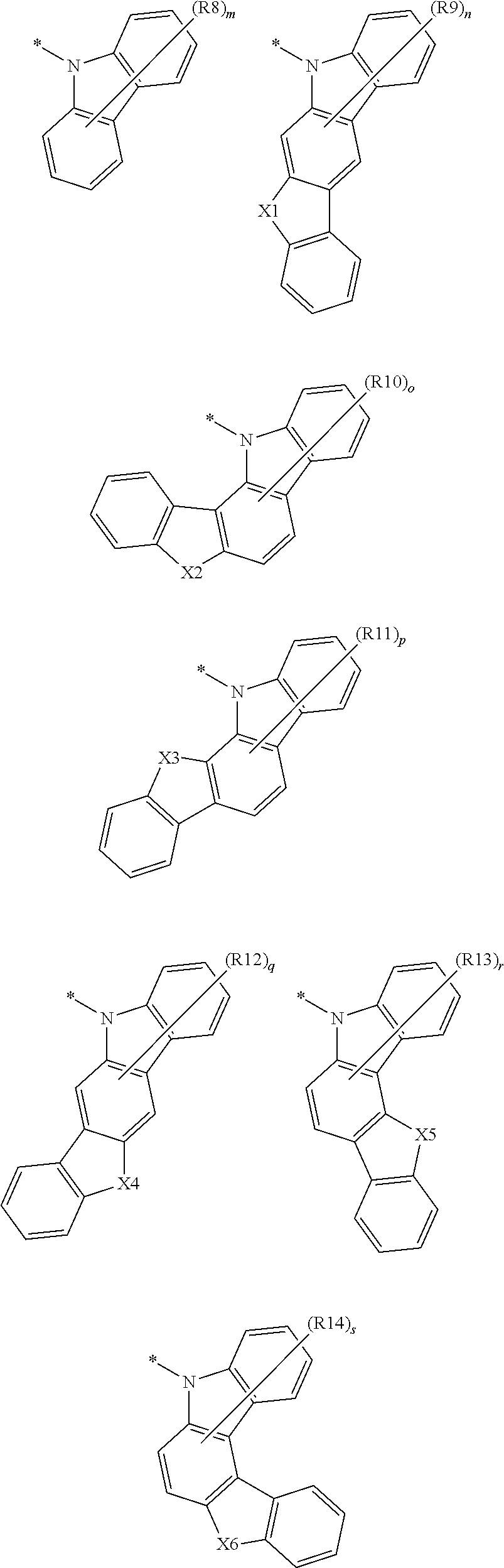
According to an exemplary embodiment of the present application, Chemical Formula 3 may be represented by any one of the following Chemical Formulae.
##STR00004##
In the structural formulae, X1 to X6 are the same as or different from each other, and each independently NR, S, O, or CR'R'',
R8 to R14 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring,
R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and
m is an integer of 0 to 8, and n, o, p, q, r, and s are each independently an integer of 0 to 6.
According to an exemplary embodiment of the present application, Chemical Formula 4 may be represented by any one of the following Chemical Formulae.
##STR00005##
In the structural formulae, X7 and X8 are the same as or different from each other, and each independently NR, S, O, or CR'R'',
R15 to R18 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group which is unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, or a C.sub.2 to C.sub.60 heteroaryl group, or two or more adjacent groups combine with each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring,
R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group, and
t is an integer of 0 to 7.
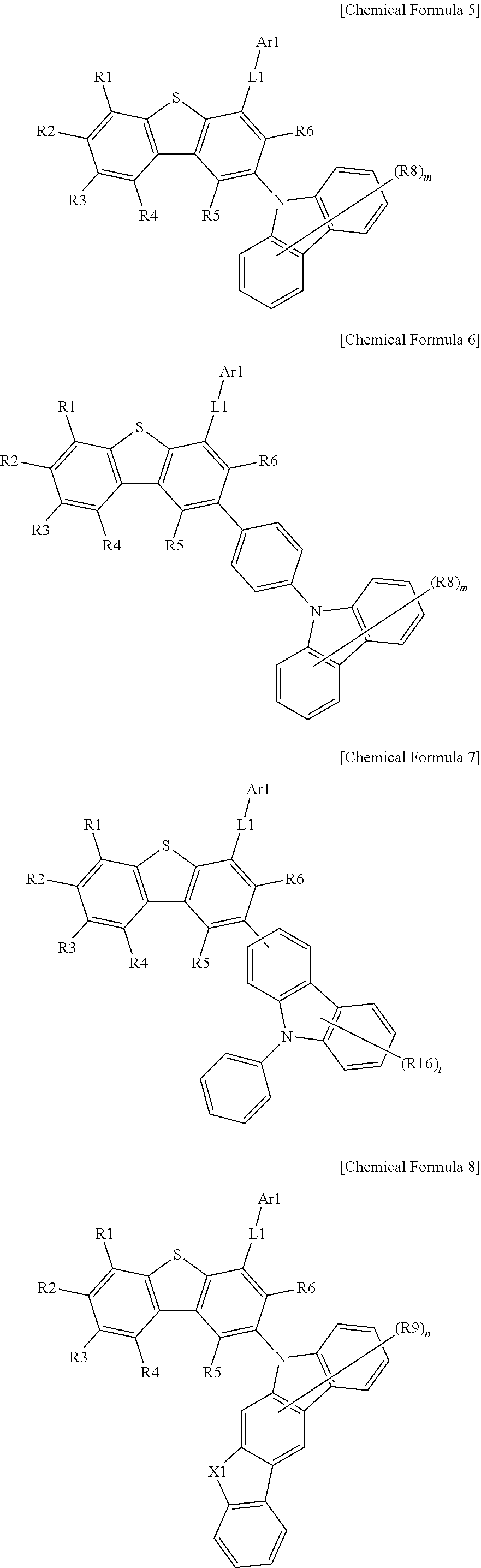
According to an exemplary embodiment of the present specification, Chemical Formula 1 may be represented by any one of the following Chemical Formulae 5 to 10.
##STR00006## ##STR00007##
In Chemical Formulae 5 to 10, the definitions of R1 to R6, R8, R9, R12, R13, R16, L1, Ar1, X1, X4, X5, m, n, q, r, and t are the same as those in Chemical Formula 1 and the structural formulae.
In an exemplary embodiment of the present application, R1 to R6 of Chemical Formula 1 may be each independently hydrogen or deuterium.
In an exemplary embodiment of the present application, R8 to R18 may be each independently hydrogen; deuterium; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
In an exemplary embodiment of the present application, R, R', and R'' of Chemical Formula 1 are the same as or different from each other, and each independently hydrogen; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; or a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group.
Further, the composition for an organic material layer of an organic light emitting device according to an exemplary embodiment of the present application may include both the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by Chemical Formula 2.
According to an exemplary embodiment of the present application, Chemical Formula 2 may be represented by any one of the following Chemical Formulae 11 to 22.
##STR00008## ##STR00009## ##STR00010## ##STR00011##
In Chemical Formulae 11 to 22, the definitions of L1, Ar1, Ar2, R1 to R4, m, n, p, and q are the same as those in Chemical Formula 2.
In an exemplary embodiment of the present application, when m', n', p', and q' of Chemical Formula 2 are each independently 2 or more, two or more R1' to R4' may be each the same as or different from each other.
In an exemplary embodiment of the present application, R1' to R4' of Chemical Formula 2 may be each independently hydrogen or deuterium.
In an exemplary embodiment of the present application, Ar1' of Chemical Formula 2 may be a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including S; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group including O.
In an exemplary embodiment of the present application, Ar1' of Chemical Formula 2 may be a phenyl group, a biphenyl group, a naphthyl group, a fluorene group in which an alkyl group is substituted, a dibenzothiophene group, or a dibenzofuran group.
In an exemplary embodiment of the present application, Ar2' of Chemical Formula 2 may be a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group.
In an exemplary embodiment of the present application, Ar2' of Chemical Formula 2 may be a phenyl group.
In the present application, the substituents of Chemical Formulae 1 and 2 will be more specifically described as follows.
In the present specification, "substituted or unsubstituted" means being unsubstituted or substituted with one or more substituents selected from the group consisting of deuterium; a halogen group; --CN; a C.sub.1 to C.sub.60 alkyl group; a C.sub.2 to C.sub.60 alkenyl group; a C.sub.2 to C.sub.60 alkynyl group; a C.sub.3 to C.sub.60 cycloalkyl group; a C.sub.2 to C.sub.60 heterocycloalkyl group; a C.sub.6 to C.sub.60 aryl group; a C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; a C.sub.1 to C.sub.20 alkylamine group; a C.sub.6 to C.sub.60 arylamine group; and a C.sub.2 to C.sub.60 heteroarylamine group, being unsubstituted or substituted with a substituent to which two or more substituents among the substituents are linked, or being unsubstituted or substituted with a substituent to which two or more substituents selected among the substituents are linked. For example, "the substituent to which two or more substituents are linked" may be a biphenyl group. That is, the biphenyl group may also be an aryl group, and may be interpreted as a substituent to which two phenyl groups are linked. The additional substituents may also be additionally substituted. R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
According to an exemplary embodiment of the present specification, the "substituted or unsubstituted" is unsubstituted or substituted with one or more substituents selected from the group consisting of deuterium, a halogen group, --CN, SiRR'R'', P(.dbd.O)RR', a C.sub.1 to C.sub.20 straight-chained or branch-chained alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group, and
R, R', and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a C.sub.1 to C.sub.60 alkyl group which is unsubstituted or substituted with deuterium, a halogen group, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group; a C.sub.3 to C.sub.60 cycloalkyl group which is unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group; a C.sub.6 to C.sub.60 aryl group which is unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group; or a C.sub.2 to C.sub.60 heteroaryl group which is unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group.
The term "substitution" means that a hydrogen atom bonded to a carbon atom of a compound is changed into another substituent, and a position to be substituted is not limited as long as the position is a position at which the hydrogen atom is substituted, that is, a position at which the substituent may be substituted, and when two or more are substituted, the two or more substituents may be the same as or different from each other.
In the present specification, the halogen may be fluorine, chlorine, bromine or iodine.
In the present specification, the alkyl group includes a straight-chain or branched-chain having 1 to 60 carbon atoms, and may be additionally substituted with another substituent. The number of carbon atoms of the alkyl group may be 1 to 60, specifically 1 to 40, and more specifically 1 to 20. Specific examples thereof include a methyl group, an ethyl group, a propyl group, an n-propyl group, an isopropyl group, a butyl group, an n-butyl group, an isobutyl group, a tert-butyl group, a sec-butyl group, a 1-methyl-butyl group, a 1-ethyl-butyl group, a pentyl group, an n-pentyl group, an isopentyl group, a neopentyl group, a tert-pentyl group, a hexyl group, an n-hexyl group, a 1-methylpentyl group, a 2-methylpentyl group, a 4-methyl-2-pentyl group, a 3,3-dimethylbutyl group, a 2-ethylbutyl group, a heptyl group, an n-heptyl group, a 1-methylhexyl group, a cyclopentylmethyl group, a cyclohexylmethyl group, an octyl group, an n-octyl group, a tert-octyl group, a 1-methylheptyl group, a 2-ethylhexyl group, a 2-propylpentyl group, an n-nonyl group, a 2,2-dimethylheptyl group, a 1-ethyl-propyl group, a 1,1-dimethyl-propyl group, an isohexyl group, a 2-methylpentyl group, a 4-methylhexyl group, a 5-methylhexyl group, and the like, but are not limited thereto.
In the present specification, the alkenyl group includes a straight-chain or branched-chain having 2 to 60 carbon atoms, and may be additionally substituted with another substituent. The number of carbon atoms of the alkenyl group may be 2 to 60, specifically 2 to 40, and more specifically 2 to 20. Specific examples thereof include a vinyl group, a 1-propenyl group, an isopropenyl group, a 1-butenyl group, a 2-butenyl group, a 3-butenyl group, a 1-pentenyl group, a 2-pentenyl group, a 3-pentenyl group, a 3-methyl-1-butenyl group, a 1,3-butadienyl group, an allyl group, a 1-phenylvinyl-1-yl group, a 2-phenylvinyl-1-yl group, a 2,2-diphenylvinyl-1-yl group, a 2-phenyl-2-(naphthyl-1-yl)vinyl-1-yl group, a 2,2-bis(diphenyl-1-yl)vinyl-1-yl group, a stilbenyl group, a styrenyl group, and the like, but are not limited thereto.
In the present specification, the alkynyl group includes a straight-chain or branched-chain having 2 to 60 carbon atoms, and may be additionally substituted with another substituent. The number of carbon atoms of the alkynyl group may be 2 to 60, specifically 2 to 40, and more specifically 2 to 20.
In the present specification, the cycloalkyl group includes a monocycle or polycycle having 3 to 60 carbon atoms, and may be additionally substituted with another substituent. Here, the polycycle means a group in which a cycloalkyl group is directly linked to or fused with another cyclic group. Here, another cyclic group may also be a cycloalkyl group, but may also be another kind of cyclic group, for example, a heterocycloalkyl group, an aryl group, a heteroaryl group, and the like. The number of carbon atoms of the cycloalkyl group may be 3 to 60, specifically 3 to 40, and more specifically 5 to 20. Specific examples thereof include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a 3-methylcyclopentyl group, a 2,3-dimethylcyclopentyl group, a cyclohexyl group, a 3-methylcyclohexyl group, a 4-methylcyclohexyl group, a 2,3-dimethylcyclohexyl group, a 3,4,5-trimethylcyclohexyl group, a 4-tert-butylcyclohexyl group, a cycloheptyl group, a cyclooctyl group, and the like, but are not limited thereto.
In the present specification, the heterocycloalkyl group includes O, S, Se, N, or Si as a heteroatom, includes a monocycle or polycycle having 2 to 60 carbon atoms, and may be additionally substituted with another substituent. Here, the polycycle means a group in which a heterocycloalkyl group is directly linked to or fused with another cyclic group. Here, another cyclic group may also be a heterocycloalkyl group, but may also be another kind of cyclic group, for example, a cycloalkyl group, an aryl group, a heteroaryl group, and the like. The number of carbon atoms of the heterocycloalkyl group may be 2 to 60, specifically 2 to 40, and more specifically 3 to 20.
In the present specification, the aryl group includes a monocycle or polycycle having 6 to 60 carbon atoms, and may be additionally substituted with another substituent. Here, the polycycle means a group in which an aryl group is directly linked to or fused with another cyclic group. Here, another cyclic group may also be an aryl group, but may also be another kind of cyclic group, for example, a cycloalkyl group, a heterocycloalkyl group, a heteroaryl group, and the like. The aryl group includes a spiro group. The number of carbon atoms of the aryl group may be 6 to 60, specifically 6 to 40, and more specifically 6 to 25. Specific examples of the aryl group include a phenyl group, a biphenyl group, a triphenyl group, a naphthyl group, an anthryl group, a chrysenyl group, a phenanthrenyl group, a perylenyl group, a fluoranthenyl group, a triphenylenyl group, a phenalenyl group, a pyrenyl group, a tetracenyl group, a pentacenyl group, a fluorenyl group, an indenyl group, an acenaphthylenyl group, a benzofluorenyl group, a spirobifluorenyl group, a 2,3-dihydro-1H-indenyl group, a fused cyclic group thereof, and the like, but are not limited thereto.
In the present specification, the spiro group is a group including a spiro structure, and may have 15 to 60 carbon atoms. For example, the spiro group may include a structure in which a 2,3-dihydro-1H-indene group or a cyclohexane group is spiro-bonded to a fluorenyl group. Specifically, the following spiro group may include any one of the groups of the following structural formulae.
##STR00012##
In the present specification, the heteroaryl group includes S, O, Se, N, or Si as a heteroatom, includes a monocycle or polycycle having 2 to 60 carbon atoms, and may be additionally substituted with another substituent. Here, the polycycle means a group in which a heteroaryl group is directly linked to or fused with another cyclic group. Here, another cyclic group may also be a heteroaryl group, but may also be another kind of cyclic group, for example, a cycloalkyl group, a heterocycloalkyl group, an aryl group, and the like. The number of carbon atoms of the heteroaryl group may be 2 to 60, specifically 2 to 40, and more specifically 3 to 25. Specific examples of the heteroaryl group include a pyridyl group, a pyrrolyl group, a pyrimidyl group, a pyridazinyl group, a furanyl group, a thiophene group, an imidazolyl group, a pyrazolyl group, an oxazolyl group, an isoxazolyl group, a thiazolyl group, an isothiazolyl group, a triazolyl group, a furazanyl group, an oxadiazolyl group, a thiadiazolyl group, a dithiazolyl group, a tetrazolyl group, a pyranyl group, a thiopyranyl group, a diazinyl group, an oxazinyl group, a thiazinyl group, a dioxinyl group, a triazinyl group, a tetrazinyl group, a quinolyl group, an isoquinolyl group, a quinazolinyl group, an isoquinazolinyl group, a quinozolilyl group, a naphthyridyl group, an acridinyl group, a phenanthridinyl group, an imidazopyridinyl group, a diaza naphthalenyl group, a triazaindene group, an indolyl group, an indolizinyl group, a benzothiazolyl group, a benzoxazolyl group, a benzimidazolyl group, a benzothiophene group, a benzofuran group, a dibenzothiophene group, a dibenzofuran group, a carbazolyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a phenazinyl group, a dibenzosilole group, spirobi (dibenzosilole), a dihydrophenazinyl group, a phenoxazinyl group, a phenanthridyl group, an imidazopyridinyl group, a thienyl group, an indolo[2,3-a]carbazolyl group, an indolo[2,3-b]carbazolyl group, an indolinyl group, a 10,11-dihydro-dibenzo[b,f]azepin group, a 9,10-dihydroacridinyl group, a phenanthrazinyl group, a phenothiazinyl group, a phthalazinyl group, a naphthylidinyl group, a phenanthrolinyl group, a benzo[c][1,2,]thiadiazolyl group, a 5,10-dihydrodibenzo[b,e][1,4]azasilinyl, a pyrazolo[1,5-c]quinazolinyl group, a pyrido[1,2-b]indazolyl group, a pyrido[1,2-a]imidazo[1,2-e]indolinyl group, a 5,11-dihydroindeno[1,2-b]carbazolyl group, and the like, but are not limited thereto.
In the present specification, the amine group may be selected from the group consisting of a monoalkylamine group; a monoarylamine group; a monoheteroarylamine group; --NH.sub.2; a dialkylamine group; a diarylamine group; a diheteroarylamine group; an alkylarylamine group; an alkylheteroarylamine group; and an arylheteroarylamine group, and the number of carbon atoms thereof is not particularly limited, but is preferably 1 to 30. Specific examples of the amine group include a methylamine group, a dimethylamine group, an ethylamine group, a diethylamine group, a phenylamine group, a naphthylamine group, a biphenylamine group, a dibiphenylamine group, an anthracenylamine group, a 9-methyl-anthracenylamine group, a diphenylamine group, a phenylnaphthylamine group, a ditolylamine group, a phenyltolylamine group, a triphenylamine group, a biphenylnaphthylamine group, a phenylbiphenylamine group, a biphenylfluorenylamine group, a phenyltriphenylenylamine group, a biphenyltriphenylenylamine group, and the like, but are not limited thereto.
In the present specification, the arylene group means that there are two bonding positions in an aryl group, that is, a divalent group. The above-described description on the aryl group may be applied, except that the arylene groups are each a divalent group. Further, the heteroarylene group means that there are two bonding positions in a heteroaryl group, that is, a divalent group. The above-described description on the heteroaryl group may be applied, except that these are each a divalent group.
According to an exemplary embodiment of the present application, Chemical Formula 1 may be represented by any one of the following compounds, but is not limited thereto.
##STR00013## ##STR00014## ##STR00015## ##STR00016## ##STR00017## ##STR00018## ##STR00019## ##STR00020## ##STR00021## ##STR00022## ##STR00023## ##STR00024## ##STR00025## ##STR00026## ##STR00027## ##STR00028## ##STR00029## ##STR00030## ##STR00031## ##STR00032## ##STR00033## ##STR00034## ##STR00035## ##STR00036## ##STR00037## ##STR00038## ##STR00039## ##STR00040## ##STR00041## ##STR00042## ##STR00043## ##STR00044## ##STR00045## ##STR00046## ##STR00047## ##STR00048## ##STR00049## ##STR00050## ##STR00051## ##STR00052## ##STR00053## ##STR00054## ##STR00055## ##STR00056## ##STR00057## ##STR00058## ##STR00059## ##STR00060## ##STR00061## ##STR00062## ##STR00063## ##STR00064## ##STR00065## ##STR00066## ##STR00067## ##STR00068## ##STR00069## ##STR00070## ##STR00071## ##STR00072## ##STR00073## ##STR00074## ##STR00075## ##STR00076## ##STR00077## ##STR00078## ##STR00079## ##STR00080## ##STR00081## ##STR00082## ##STR00083## ##STR00084## ##STR00085## ##STR00086## ##STR00087## ##STR00088##
According to an exemplary embodiment of the present application, Chemical Formula 2 may be represented by any one of the following compounds, but is not limited thereto.
##STR00089## ##STR00090## ##STR00091## ##STR00092## ##STR00093## ##STR00094## ##STR00095## ##STR00096## ##STR00097## ##STR00098## ##STR00099## ##STR00100## ##STR00101## ##STR00102## ##STR00103## ##STR00104## ##STR00105## ##STR00106## ##STR00107## ##STR00108## ##STR00109## ##STR00110## ##STR00111## ##STR00112## ##STR00113## ##STR00114## ##STR00115## ##STR00116## ##STR00117## ##STR00118## ##STR00119##
Further, it is possible to synthesize a compound having inherent characteristics of a substituent introduced by introducing various substituents into the structures of Chemical Formulae 1 and 2. For example, it is possible to synthesize a material which satisfies conditions required for each organic material layer by introducing a substituent usually used for a hole injection layer material, a material for transporting holes, a light emitting layer material, an electron transport layer material, and a charge generation layer material, which are used for preparing an organic light emitting device, into the core structure.
In addition, it is possible to finely adjust an energy band gap by introducing various substituents into the structures of Chemical Formulae 1 and 2, and meanwhile, it is possible to improve characteristics at the interface between organic materials and diversify the use of material.
Meanwhile, the hetero-cyclic compound has a high glass transition temperature (Tg) and thus has excellent thermal stability. The increase in thermal stability becomes an important factor which provides driving stability to a device.
The hetero-cyclic compound according to an exemplary embodiment of the present application may be prepared by a multi-step chemical reaction. Some intermediate compounds are first prepared, and a compound of Chemical Formula 1 or 2 may be prepared from the intermediate compounds. More specifically, the hetero-cyclic compound according to an exemplary embodiment of the present application may be prepared based on the Preparation Examples to be described below.
Furthermore, another exemplary embodiment of the present application provides a composition for an organic material layer of an organic light emitting device, which includes both the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by Chemical Formula 2.
The specific contents on the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by Chemical Formula 2 are the same as those described above.
The weight ratio of the hetero-cyclic compound represented by Chemical Formula 1:the compound represented by Chemical Formula 2 in the composition may be 1:10 to 10:1, 1:8 to 8:1, 1:5 to 5:1, and 1:2 to 2:1, but is not limited thereto.
The composition may be used when an organic material for an organic light emitting device is formed, and particularly, may be more preferably used when a host of a light emitting layer is formed.
The composition is in a form in which two or more compounds are simply mixed, materials in a powder state may also be mixed before an organic material layer of an organic light emitting device is formed, and it is possible to mix compounds in a liquid state at a temperature which is equal to or more than a suitable temperature. The composition is in a solid state at a temperature which is equal to or less than the melting point of each material, and may be maintained as a liquid if the temperature is adjusted.
Another exemplary embodiment of the present application provides an organic light emitting device including the hetero-cyclic compound represented by Chemical Formula 1.
Further, the organic light emitting device according to an exemplary embodiment of the present application includes a positive electrode, a negative electrode, and one or more organic material layers provided between the positive electrode and the negative electrode, in which one or more layers of the organic material layers include the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by Chemical Formula 2.
The organic light emitting device according to an exemplary embodiment of the present application may be manufactured by typical methods and materials for manufacturing an organic light emitting device, except that the one or more organic material layers are formed by using the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2, which are described above.
The compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2 may be formed as an organic material layer by not only a vacuum deposition method, but also a solution application method when an organic light emitting device is manufactured. Here, the solution application method means spin coating, dip coating, inkjet printing, screen printing, a spray method, roll coating, and the like, but is not limited thereto.
Specifically, the organic light emitting device according to an exemplary embodiment of the present application includes a positive electrode, a negative electrode, and one or more organic material layers provided between the positive electrode and the negative electrode, in which one or more of the organic material layers include the hetero-cyclic compound represented by Chemical Formula 1.
Further, the organic light emitting device according to an exemplary embodiment of the present application includes a positive electrode, a negative electrode, and an one or more organic material layers provided between the positive electrode and the negative electrode, in which one or more layers of the organic material layers include the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2.
FIGS. 1 to 3 exemplify the stacking sequence of the electrodes and the organic material layers of the organic light emitting device according to an exemplary embodiment of the present application. However, the scope of the present application is not intended to be limited by these drawings, and the structure of the organic light emitting device known in the art may also be applied to the present application.
According to FIG. 1, an organic light emitting device in which a positive electrode 200, an organic material layer 300, and a negative electrode 400 are sequentially stacked on a substrate 100 is illustrated. However, the organic light emitting device is not limited only to such a structure, and as in FIG. 2, an organic light emitting device in which a negative electrode, an organic material layer, and a positive electrode are sequentially stacked on a substrate may also be implemented.
FIG. 3 exemplifies a case where an organic material layer is a multilayer. An organic light emitting device according to FIG. 3 includes a hole injection layer 301, a hole transport layer 302, a light emitting layer 303, a hole blocking layer 304, an electron transport layer 305, and an electron injection layer 306. However, the scope of the present application is not limited by the stacking structure as described above, and if necessary, the other layers except for the light emitting layer may be omitted, and another necessary functional layer may be further added.
The organic light emitting device according to the present specification may be manufactured by materials and methods known in the art, except that one or more layers in the organic material layers include the hetero-cyclic compound represented by Chemical Formula 1, or include both the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2.
The hetero-cyclic compound represented by Chemical Formula 1 may alone constitute one or more layers of the organic material layers of the organic light emitting device. However, the hetero-cyclic compound represented by Chemical Formula 1 may be mixed with another material, if necessary, to constitute an organic material layer.
The hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for an electron transport layer, a hole blocking layer, or a light emitting layer, and the like in the organic light emitting device. As an example, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for an electron transport layer, a hole transport layer, or a light emitting layer of the organic light emitting device.
Furthermore, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for a light emitting layer in the organic light emitting device. As an example, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material for a phosphorescent host of a light emitting layer in the organic light emitting device.
Further, the organic material layer including the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2 may additionally include another material, if necessary.
The hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by chemical Formula 2 may be used as a material for a charge generation layer in the organic light emitting device.
The hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by chemical Formula 2 may be used as a material for an electron transport layer, a hole blocking layer, and a light emitting layer, and the like in the organic light emitting device. As an example, the hetero-cyclic compound represented by Chemical Formula 1 and the hetero-cyclic compound represented by Chemical Formula 2 may be used as a material for an electron transport layer, a hole transport layer, or a light emitting layer of the organic light emitting device.
Furthermore, the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by chemical Formula 2 may be used as a material for a light emitting layer in the organic light emitting device. As an example, the hetero-cyclic compound represented by Chemical Formula 1 and the compound represented by chemical Formula 2 may be used as a material for a phosphorescent host of a light emitting layer in the organic light emitting device.
In the organic light emitting device according to an exemplary embodiment of the present application, materials other than the hetero-cyclic compound of Chemical Formula 1 and the hetero-cyclic compound of Chemical Formula 2 will be exemplified below, but these materials are provided only for exemplification and are not for limiting the scope of the present application, and may be replaced with materials publicly known in the art.
As a material for the positive electrode, materials having a relatively high work function may be used, and a transparent conductive oxide, a metal or a conductive polymer, and the like may be used. Specific examples of the positive electrode material include: a metal, such as vanadium, chromium, copper, zinc, and gold, or alloys thereof; a metal oxide, such as zinc oxide, indium oxide, indium tin oxide (ITO), and indium zinc oxide (IZO); a combination of metal and oxide, such as ZnO:Al or SnO.sub.2:Sb; a polymer, such as poly(3-methyl compound), poly[3,4-(ethylene-1,2-dioxy)compound] (PEDT), polypyrrole, and polyaniline, and the like, but are not limited thereto.
As a material for the negative electrode, materials having a relatively low work function may be used, and a metal, a metal oxide, or a conductive polymer, and the like may be used. Specific examples of the negative electrode material include: a metal, such as magnesium, calcium, sodium, potassium, titanium, indium, yttrium, lithium, gadolinium, aluminum, silver, tin, and lead, or alloys thereof; a multi-layered structural material, such as LiF/Al or LiO.sub.2/Al, and the like, but are not limited thereto.
As a hole injection material, a publicly-known hole injection material may also be used, and it is possible to use, for example, a phthalocyanine compound such as copper phthalocyanine disclosed in U.S. Pat. No. 4,356,429 or starburst-type amine derivatives described in the document [Advanced Material, 6, p. 677 (1994)], for example, tris(4-carbazoyl-9-ylphenyl)amine (TCTA), 4,4',4''-tris[phenyl(m-tolyl)amino]triphenylamine (m-MTDATA), 1,3,5-tris[4-(3-methylphenylphenylamino)phenyl]benzene (m-MTDAPB), polyaniline/dodecylbenzenesulfonic acid or poly(3,4-ethylenedioxythiophene)/poly(4-styrenesulfonate), which is a soluble conductive polymer, polyaniline/camphor sulfonic acid or polyaniline/poly(4-styrene-sulfonate), and the like.
As the hole transport material, a pyrazoline derivative, an arylamine-based derivative, a stilbene derivative, a triphenyldiamine derivative, and the like may be used, and a low-molecular weight or polymer material may also be used.
As the electron transport material, it is possible to use an oxadiazole derivative, anthraquinodimethane and a derivative thereof, benzoquinone and a derivative thereof, naphthoquinone and a derivative thereof, anthraquinone and a derivative thereof, tetracyanoanthraquinodimethane and a derivative thereof, a fluorenone derivative, diphenyldicyanoethylene and a derivative thereof, a diphenoquinone derivative, a metal complex of 8-hydroxyquinoline and a derivative thereof, and the like, and a low-molecular weight material and a polymer material may also be used.
As the electron injection material, for example, LiF is representatively used in the art, but the present application is not limited thereto.
As the light emitting material, a red, green, or blue light emitting material may be used, and if necessary, two or more light emitting materials may be mixed and used. In this case, two or more light emitting materials are deposited or used as an individual supply source, or pre-mixed to be deposited and used as one supply source. Further, as the light emitting material, a fluorescent material may also be used, but a phosphorescent material may also be used. As the light emitting material, it is also possible to use alone a material which emits light by combining holes and electrons each injected from the positive electrode and the negative electrode, but materials in which a host material and a dopant material work together to emit light may also be used.
When hosts of the light emitting material are mixed and used, the same series hosts may also be mixed and used, and different series hosts may also be mixed and used. For example, two or more materials selected from n-type host materials or p-type host materials may be used as a host material for a light emitting layer.
The organic light emitting device according to an exemplary embodiment of the present application may be a top emission type, a bottom emission type, or a dual emission type according to the material to be used.
The hetero-cyclic compound according to an exemplary embodiment of the present application may be operated by a principle which is similar to the principle applied to an organic light emitting device, even in an organic electronic device including an organic solar cell, an organic photoconductor, an organic transistor, and the like.
Mode for Invention
Hereinafter, the present specificastion will be described in more detail through the Examples, but these Examples are provided only for exemplifying the present application, and are not intended to limit the scope of the present application.
EXAMPLES
<Preparation Example 1> Preparation of Compound 1-11-2
##STR00120##
1) Preparation of Compound 1-11-2
5.0 g (19.0 mM) of 2-bromodibenzo[b,d]thiophene, 2.6 g (15.8 mM) of 9H--carbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 4.7 g (85%) of Target Compound 1-11-2.
2) Preparation of Compound 1-11-1
7.4 mL (18.6 mM) of 2.5 M n-BuLi was added dropwise to a mixed solution containing 5 g (14.3 mM) of Compound 1-11-2 and 100 mL of THF at -78.degree. C., and the resulting mixture was stirred at room temperature for 1 hour. 4.8 mL (42.9 mM) of trimethyl borate (B(OMe).sub.3) was added dropwise to the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:MeOH=100:3) and recrystallized with DCM to obtain 3.9 g (70%) of Target Compound 1-11-1.
3) Preparation of Compound 1-11
7.5 g (19.0 mM) of Compound 1-11-1, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/EtOH/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 7.7 g (70%) of Target Compound 1-11.
Target Compound A was prepared and synthesized in the same manner as in the preparation in Preparation Example 1, except that Intermediate A in the following Table 1 was used instead of 9H-carbazole, and Intermediate B in the following Table 1 was used instead of 2-chloro-4,6-diphenyl-1,3,5-triazine in Preparation Example 1.
TABLE-US-00001 TABLE 1 Compound No. Intermediate A Intermediate B 1-2 ##STR00121## ##STR00122## 1-12 ##STR00123## 1-17 ##STR00124## 1-23 ##STR00125## 1-27 ##STR00126## 1-33 ##STR00127## 1-36 ##STR00128## 1-39 ##STR00129## ##STR00130## 1-40 ##STR00131## 1-41 ##STR00132## 1-42 ##STR00133## 1-46 ##STR00134## ##STR00135## 1-65 ##STR00136## ##STR00137## 1-66 ##STR00138## 1-67 ##STR00139## 1-68 ##STR00140## 1-69 ##STR00141## 1-70 ##STR00142## 1-71 ##STR00143## 1-72 ##STR00144## 1-76 ##STR00145## ##STR00146## 1-77 ##STR00147## 1-78 ##STR00148## 1-79 ##STR00149## 1-82 ##STR00150## ##STR00151## 1-83 ##STR00152## 1-84 ##STR00153## 1-85 ##STR00154## 1-86 ##STR00155## 1-91 ##STR00156## ##STR00157## 1-92 ##STR00158## 1-93 ##STR00159## 1-94 ##STR00160## 1-96 ##STR00161## ##STR00162## 1-98 ##STR00163## 1-99 ##STR00164## 1-100 ##STR00165## 1-109 ##STR00166## ##STR00167## 1-110 ##STR00168## 1-111 ##STR00169## 1-112 ##STR00170## 1-113 ##STR00171## 1-117 ##STR00172## ##STR00173## 1-118 ##STR00174## 1-119 ##STR00175## 1-125 ##STR00176## ##STR00177## 1-126 ##STR00178## 1-127 ##STR00179## 1-128 ##STR00180## 1-138 ##STR00181## ##STR00182## 1-176 ##STR00183## ##STR00184## 1-177 ##STR00185## 1-178 ##STR00186## 1-179 ##STR00187## 1-180 ##STR00188## 1-181 ##STR00189## Compound No. Target Compound A Total yield 1-2 ##STR00190## 41% 1-12 ##STR00191## 42% 1-17 ##STR00192## 43% 1-23 ##STR00193## 46% 1-27 ##STR00194## 45% 1-33 ##STR00195## 43% 1-36 ##STR00196## 41% 1-39 ##STR00197## 48% 1-40 ##STR00198## 49% 1-41 ##STR00199## 47% 1-42 ##STR00200## 45% 1-46 ##STR00201## 48% 1-65 ##STR00202## 44% 1-66 ##STR00203## 46% 1-67 ##STR00204## 47% 1-68 ##STR00205## 44% 1-69 ##STR00206## 46% 1-70 ##STR00207## 44% 1-71 ##STR00208## 43% 1-72 ##STR00209## 42% 1-76 ##STR00210## 45% 1-77 ##STR00211## 46% 1-78 ##STR00212## 47% 1-79 ##STR00213## 48% 1-82 ##STR00214## 49% 1-83 ##STR00215## 43% 1-84 ##STR00216## 44% 1-85 ##STR00217## 45% 1-86 ##STR00218## 43% 1-91 ##STR00219## 46% 1-92 ##STR00220## 47% 1-93 ##STR00221## 47% 1-94 ##STR00222## 44% 1-96 ##STR00223## 46% 1-98 ##STR00224## 43% 1-99 ##STR00225## 43% 1-100 ##STR00226## 42% 1-109 ##STR00227## 41% 1-110 ##STR00228## 42% 1-111 ##STR00229## 42% 1-112 ##STR00230## 45% 1-113 ##STR00231## 45% 1-117 ##STR00232## 46% 1-118 ##STR00233## 49% 1-119 ##STR00234## 46% 1-125 ##STR00235## 43% 1-126 ##STR00236## 44% 1-127 ##STR00237## 45% 1-128 ##STR00238## 42% 1-138 ##STR00239## 47% 1-176 ##STR00240## 48% 1-177 ##STR00241## 49% 1-178 ##STR00242## 44% 1-179 ##STR00243## 44% 1-180 ##STR00244## 43% 1-181 ##STR00245## 46%
<Preparation Example 2> Preparation of Compound 1-64
##STR00246##
1) Preparation of Compound 1-64-2
5.0 g (19.0 mM) of 2-bromodibenzo[b,d]thiophene, 5.5 g (19.0 mM) of (9-phenyl-9H-carbazol-3-yl)boronic acid, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/EtOH/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with hexane to obtain 5.7 g (70%) of Target Compound 1-64-2.
2) Preparation of Compound 1-64-1
7.4 mL (18.6 mM) of 2.5 M n-BuLi was added dropwise to a mixed solution containing 6.1 g (14.3 mM) of Compound 1-64-2 and 100 mL of THF at -78.degree. C., and the resulting mixture was stirred at room temperature for 1 hour. 4.8 mL (42.9 mM) of trimethyl borate (B(OMe).sub.3) was added dropwise to the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:MeOH=100:3) and recrystallized with DCM to obtain 4.7 g (70%) of Target Compound 1-64-1.
3) Preparation of Compound 1-64
8.9 g (19.0 mM) of Compound 1-64-1, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/EtOH/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 8.7 g (70%) of Target Compound 1-64.
Target Compound B was prepared and synthesized in the same manner as in the preparation in Preparation Example 2, except that Intermediate C in the following Table 2 was used instead of (9-phenyl-9H-carbazol-3-yl)boronic acid, and Intermediate D in the following Table 2 was used instead of 2-chloro-4,6-diphenyl-1,3,5-triazine in Preparation Example 2.
TABLE-US-00002 TABLE 2 Compound No. Intermediate C Intermediate D 1-80 ##STR00247## ##STR00248## 1-95 ##STR00249## 1-114 ##STR00250## 1-139 ##STR00251## ##STR00252## 1-140 ##STR00253## ##STR00254## 1-155 ##STR00255## ##STR00256## 1-156 ##STR00257## 1-157 ##STR00258## 1-158 ##STR00259## 1-160 ##STR00260## 1-162 ##STR00261## 1-163 ##STR00262## 1-164 ##STR00263## 1-165 ##STR00264## 1-170 ##STR00265## 1-172 ##STR00266## 1-174 ##STR00267## ##STR00268## Compound No. Target Compound B Total yield 1-80 ##STR00269## 34% 1-95 ##STR00270## 35% 1-114 ##STR00271## 37% 1-139 ##STR00272## 36% 1-140 ##STR00273## 38% 1-155 ##STR00274## 40% 1-156 ##STR00275## 39% 1-157 ##STR00276## 39% 1-158 ##STR00277## 37% 1-160 ##STR00278## 36% 1-162 ##STR00279## 37% 1-163 ##STR00280## 33% 1-164 ##STR00281## 34% 1-165 ##STR00282## 36% 1-170 ##STR00283## 37% 1-172 ##STR00284## 33% 1-174 ##STR00285## 38%
<Preparation Example 3> Synthesis of Compound 2-2
##STR00286##
1) Preparation of Compound 2-2-2 (Ref 1)
4.2 g (15.8 mM) of 2-bromodibenzo[b,d]thiophene, 6.5 g (15.8 mM) of 9-phenyl-9H,9'H-3,3'-bicarbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 7.9 g (85%) of Target Compound 2-2-2.
2) Preparation of Compound 2-2-1
7.4 mL (18.6 mmol) of 2.5 M n-BuLi was added dropwise to a mixed solution containing 8.4 g (14.3 mmol) of Compound 2-2-1 and 100 mL of THF at -78.degree. C., and the resulting mixture was stirred at room temperature for 1 hour. 4.8 mL (42.9 mmol) of trimethyl borate was added dropwise to the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:MeOH=100:3) and recrystallized with DCM to obtain 3.9 g (70%) of Target Compound 2-2-1.
3) Preparation of Compound 2-2
6.7 g (10.5 mM) of Compound 2-2-1, 2.1 g (10.5 mM) of iodobenzene, 606 mg (0.52 mM) of Pd(PPh.sub.3).sub.4, and 2.9 g (21.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/EtOH/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 4.9 g (70%) of Target Compound 2-2.
<Preparation Example 4> Synthesis of Compound 2-3
Target Compound 2-3 (83%) was obtained by performing the preparation in the same manner as in the preparation of Compound 2-2, except that 4-iodo-1,1'-biphenyl was used instead of iodobenzene in the preparation of Compound 2-2.
<Preparation Example 5> Synthesis of Compound Ref 2
##STR00287##
1) Preparation of Compound ref 2-2
88.0 mL (157.8 mM) of 1.8 M LDA was added dropwise to a mixed solution containing 30.0 g (121.4 mM) of 2-bromodibenzofuran and 300 mL of THF at -78.degree. C., and the resulting mixture was stirred at room temperature for 1 hour. 11.0 g (42.9 mmol) of iodine was put into the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM) and recrystallized with MeOH to obtain 23.1 g (51%) of Target Compound ref 2-2.
2) Preparation of Compound Ref 2-1
3.9 g (10.5 mM) of Compound ref 2-2, 1.3 g (10.5 mM) of phenylboronic acid, 606 mg (0.52 mM) of Pd(PPh.sub.3).sub.4, and 2.9 g (21.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/EtOH/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 2.4 g (70%) of Target Compound ref 2-1.
3) Preparation of Compound ref 2
5.1 g (15.8 mM) of Compound ref 2-1, 6.5 g (15.8 mM) of 9-phenyl-9H,9'H-3,3'-bicarbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 8.7 g (85%) of Target Compound ref 2.
<Preparation Example 6> Preparation of Compound Ref 3
##STR00288##
5.5 g (19.0 mmol) of (3-(9H-carbazol-9-yl)phenyl)boronic acid, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/ethanol/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 6.3 g (70%) of Target Compound ref 3.
<Preparation Example 7> Synthesis of Compound 2-7
Compound 2-7 was obtained by using 2-bromo-9,9-dimethyl-9H-fluorene instead of iodobenzene in the preparation of Compound 2-2 (yield 69%).
<Preparation Example 8> Synthesis of Compound 2-9
Compound 2-9 was obtained by using 2-bromodibenzo[b,d]thiophene instead of iodobenzene in the preparation of Compound 2-2 (yield 72%).
<Preparation Example 9> Synthesis of Compound 2-11
Compound 2-11 was obtained by using 2-bromodibenzo[b,d]furan instead of iodobenzene in the preparation of Compound 2-2 (yield 68%).
<Preparation Example 10> Preparation of Compound Ref 4
##STR00289##
5.5 g (19.0 mmol) of (3-(9H-carbazol-9-yl)phenyl)boronic acid, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/ethanol/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 6.3 g (70%) of Compound ref 4.
<Preparation Example 11> Preparation of Compound Ref 5
##STR00290##
5.8 g (19.0 mM) of (3-(dibenzo[b,d]thiophene-4-yl)phenyl)boronic acid, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/ethanol/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 6.5 g (70%) of Compound ref 5.
<Preparation Example 12> Synthesis of Compound Ref 6
##STR00291##
1) Preparation of Compound ref 6-2
11.4 mL (22.8 mM) of 2.0 M lithium diisopropylamine was added dropwise to a mixed solution containing 4.7 g (19.0 mM) of 2-bromodibenzo[b,d]furan and 100 mL of THF at -78.degree. C., and the resulting mixture was stirred at -78.degree. C. for 1 hour. 4.8 mL (42.9 mM) of trimethyl borate was added dropwise to the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:MeOH=100:3) and recrystallized with DCM to obtain 3.9 g (70%) of Compound ref 6-2.
2) Preparation of Compound Ref 6-1
5.5 g (19.0 mM) of Compound ref 6-2, 5.9 g (19.0 mM) of 2-bromo-4,6-diphenylpyrimidine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/ethanol/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with hexane to obtain 6.3 g (70%) of Compound ref 6-1.
3) Preparation of Compound Ref 6
9.1 g (19.0 mM) of Compound ref 6-1, 4.5 g (15.8 mM) of 7,7-dimethyl-5,7-dihydroindeno[2,1-b]carbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 9.1 g (85%) of Compound ref 6.
<Preparation Example 13> Synthesis of Compound Ref 7
##STR00292##
1) Preparation of Compound Ref 7-2
5.0 g (19.0 mM) of 4-bromodibenzo[b,d]thiophene, 2.6 g (15.8 mM) of 9H-carbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 4.7 g (85%) of Compound ref 7-2.
2) Preparation of Compound Ref 7-1
7.4 mL (18.6 mM) of 2.5 M n-BuLi was added dropwise to a mixed solution containing 5.0 g (14.3 mM) of Compound ref 7-2 and 100 mL of THF at -78.degree. C., and the resulting mixture was stirred at room temperature for 1 hour. 4.8 mL (42.9 mM) of trimethyl borate was added dropwise to the reaction mixture, and the resulting mixture was stirred at room temperature for 2 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:MeOH=100:3) and recrystallized with DCM to obtain 3.9 g (70%) of Target Compound ref 7-1.
3) Preparation of Compound Ref 7
7.5 g (19.0 mM) of Compound ref 7-1, 5.1 g (19.0 mM) of 2-chloro-4,6-diphenyl-1,3,5-triazine, 1.1 g (0.95 mM) of Pd(PPh.sub.3).sub.4, and 5.2 g (38.0 mM) of K.sub.2CO.sub.3 were dissolved in 100/20/20 mL of toluene/ethanol/H.sub.2O, and then the resulting solution was refluxed for 12 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 7.7 g (70%) of Compound ref 7.
<Preparation Example 14> Synthesis of Compound Ref 8
##STR00293##
5.0 g (19.0 mM) of 2-bromodibenzo[b,d]thiophene, 4.5 g (15.8 mM) of 7,7-dimethyl-5,7-dihydroindeno[2,1-b]carbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 7.3 g (85%) of Compound ref 8.
<Preparation Example 15> Synthesis of Compound Ref 9
##STR00294##
4.2 g (15.8 mM) of 2-bromodibenzo[b,d]thiophene, 6.5 g (15.8 mM) of 9-phenyl-9H,9'H-3,3'-bicarbazole, 3.0 g (15.8 mM) of CuI, 1.9 mL (15.8 mM) of trans-1,2-diaminocyclohexane, and 3.3 g (31.6 mM) of K.sub.3PO.sub.4 were dissolved in 100 mL of 1,4-oxane, and then the resulting solution was refluxed for 24 hours. After the reaction was completed, distilled water and DCM were added thereto at room temperature, extraction was performed, the organic layer was dried over MgSO.sub.4, and then the solvent was removed by a rotary evaporator. The reactant was purified by column chromatography (DCM:Hex=1:3) and recrystallized with methanol to obtain 7.9 g (85%) of Compound ref 9.
Compounds were prepared in the same manner as in the Preparation Examples, and the synthesis confirmation results thereof are shown in Tables 3 to 23.
TABLE-US-00003 TABLE 3 HOMO = -5.5 - (E.sub.ox(Compound 1-2) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(404 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.81 Compound 1-2 1.45 -6.14 3.07 -3.07
TABLE-US-00004 TABLE 4 HOMO = - 5.5 - (E.sub.ox(Compound 1-11) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(432 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.80 Compound 1-11 1.48 -6.18 2.87 -3.31
TABLE-US-00005 TABLE 5 HOMO = -5.5 - (E.sub.ox(Compound 1-23) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(367 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.82 Compound 1-23 1.38 -6.06 3.38 -2.68
TABLE-US-00006 TABLE 6 HOMO = -5.5 - (E.sub.ox(Compound 1-27) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(369 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.80 Compound 1-27 1.37 -6.07 3.36 -2.71
TABLE-US-00007 TABLE 7 HOMO = -5.5 - (E.sub.ox(Compound 1-33) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(378 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.83 Compound 1-33 1.45 -6.11 3.28 -3.82
TABLE-US-00008 TABLE 8 HOMO = -5.5 - (E.sub.ox(Compound 1-39) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(433 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.76 Compound 1-39 1.41 -6.15 2.86 -3.29
TABLE-US-00009 TABLE 9 HOMO = -5.5 - (E.sub.ox(Compound 1-41) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(366 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.78 Compound 1-41 1.40 -6.12 3.39 -2.73
TABLE-US-00010 TABLE 10 HOMO = -5.5 - (E.sub.ox(Compound 1-65) - E.sub.ox(NPB)) (eV) Band gap = 1240/UV absorption edge(416 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.79 Compound 1-65 1.22 -5.93 2.98 -2.95
TABLE-US-00011 TABLE 11 HOMO = -5.5 - (E.sub.ox (Compound 1-66) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (425 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.77 Compound 1-66 1.19 -5.93 2.92 -3.01
TABLE-US-00012 TABLE 12 HOMO = -5.5 - (E.sub.ox (Compound 1-67) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (452 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.76 Compound 1-67 1.16 -5.89 2.74 -3.15
TABLE-US-00013 TABLE 13 HOMO = -5.5 - (E.sub.ox (Compound 1-69) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (371 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.78 Compound 1-69 1.13 -5.86 3.34 -2.52
TABLE-US-00014 TABLE 14 HOMO = -5.5 - (E.sub.ox (Compound 1-70) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (371 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.76 Compound 1-70 1.14 -5.88 3.34 -2.54
TABLE-US-00015 TABLE 15 HOMO = -5.5 - (E.sub.ox (Compound 1-71) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (371 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.77 Compound 1-71 1.15 -5.88 3.34 -2.54
TABLE-US-00016 TABLE 16 HOMO = -5.5 - (E.sub.ox (Compound 1-78) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (438 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.78 Compound 1-78 1.30 -6.02 2.83 -3.19
TABLE-US-00017 TABLE 17 HOMO = -5.5 - (E.sub.ox (Compound 1-82) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (426 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.78 Compound 1-82 1.09 -5.81 2.91 -2.90
TABLE-US-00018 TABLE 18 HOMO = -5.5 - (E.sub.ox (Compound 1-84) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (463 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.77 Compound 1-84 1.08 -5.80 2.68 -3.12
TABLE-US-00019 TABLE 19 HOMO = -5.5 - (E.sub.ox (Compound 1-99) - E.sub.ox (NPB)) (eV) Band gap = 1240/UV absorption edge (364 nm) (eV) E.sub.ox HOMO Band gap LUMO NPB 0.83 Compound 1-99 1.36 -6.03 3.41 -2.62
TABLE-US-00020 TABLE 20 Td (signal value: 95%) Tg Compound 1-2 442.04.degree. C. -- Compound 1-11 426.38.degree. C. -- Compound 1-23 473.62.degree. C. 141.91.degree. C. Compound 1-27 464.07.degree. C. -- Compound 1-33 478.83.degree. C. 153.06.degree. C. Compound 1-39 460.32.degree. C. 151.88.degree. C. Compound 1-41 484.24.degree. C. 157.88.degree. C. Compound 1-65 467.84.degree. C. 179.13.degree. C. Compound 1-66 463.00.degree. C. 179.38.degree. C. Compound 1-67 461.79.degree. C. 185.05.degree. C. Compound 1-69 492.45.degree. C. 179.99.degree. C. Compound 1-70 482.96.degree. C. 185.05.degree. C. Compound 1-71 461.10.degree. C. 169.72.degree. C. Compound 1-78 462.82.degree. C. 180.63.degree. C. Compound 1-82 495.17.degree. C. -- Compound 1-84 492.52.degree. C. 188.83.degree. C. Compound 1-99 523.67.degree. C. 176.50.degree. C.
TABLE-US-00021 TABLE 21 Compound .sup.1H NMR (CDCl.sub.3, 200 Mz) 1-2 .delta. = 9.26 (1H, d), 8.51 (1H, d), 8.41~8.39 (4H, m), 8.23 (2H, d), 8.18 (1H, d), 8.13 (1H, s), 8.06 (1H, d), 7.62~7.43 (12H, m), 7.34 (2H, t) 1-11 .delta. = 9.32 (1H, d), 8.90~8.88 (4H, m), 8.61 (1H, d), 8.25 (2H, d), 8.21 (1H, d), 8.12 (1H, d), 7.65~7.45 (12H, m), 7.37 (2H, t) 1-12 .delta. = 8.55 (1H, d), 8.45~8.36 (4H, m), 8.19 (1H, d), 7.93~8.00 (4H, m), 7.73~7.77 (4H, m), 7.35~7.61 (12H, m), 7.20~7.20 (2H, m) 1-17 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.30 (2H, d), 7.90~8.12 (6H, m), 7.78 (1H, t), 7.47~7.69 (10H, m), 7.25~7.33 (3H, m) 1-23 .delta. = 8.55 (1H, d), 8.45~8.38 (2H, m), 8.23~8.19 (2H, m), 8.00~7.93 (8H, m), 7.77~7.73 (2H, m), 7.58~7.49 (11H, m), 7.35 (1H, t), 7.20~7.16 (2H, m) 1-27 .delta. = 9.27 (1H, s), 8.89 (1H, d), 8.79 (4H, m), 8.41 (1H, d), 8.21 (3H, m), 8.05 (1H, d), 7.93 (1H, d), 7.87 (1H, d), 7.77 (1H, t), 7.64~7.46 (12H, m), 7.32 (2H, t) 1-33 .delta. = 8.92 (2H, d), 8.36 (1H, d), 8.32 (4H, m), 8.21~8.16 (3H, m), 8.07 (1H, s), 8.01 (2H, d), 7.94 (1H, d), 7.61~7.44 (12H, m), 7.32 (2H, t) 1-36 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.36 (4H, d), 8.19 (1H, d), 8.00~7.93 (5H, m), 7.77 (1H, s), 7.58~7.49 (10H, m), 7.35 (1H, t), 7.25~7.16 (4H, m) 1-39 .delta. = 9.35 (1H, s), 8.90 (4H, d), 8.64 (1H, s), 8.30~8.20 (3H, m), 8.13 (1H, d), 7.71 (1H, s), 7.66~7.45 (14H, m), 7.38~7.33 (3H, m) 1-40 .delta. = 9.38 (1H, s), 9.24 (1H, s), 8.87 (3H, d), 8.64 (1H, s), 8.30~8.21 (3H, m), 8.08 (1H, d), 7.90 (1H, d), 7.80 (2H, d), 7.72~7.32 (19H, d) 1-41 .delta. = 9.30 (1H, s), 8.90 (1H, d), 8.80 (4H, d), 8.43 (1H, s), 8.26~8.20 (3H, m), 8.03 (1H, d), 7.93 (1H, d), 7.87 (1H, s), 7.77 (1H, t), 7.72 (1H, s), 7.67 (1H, d), 7.60~7.47 (11H, m), 7.41~7.29 (4H, m) 1-42 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.36~8.31 (5H, m), 8.00~7.91 (6H, m), 7.77~7.74 (4H, m), 7.56~7.35 (12H, m), 7.25 (2H, d), 7.16 (1H, t) 1-46 .delta. = 8.62 (1H, d), 8.45 (1H, d), 8.36~8.31 (5H, m), 8.22 (1H, m), 8.00 (1H, s), 7.93~7.91 (2H, m), 7.77~7.74 (7H, m), 7.50~7.41 (14H, m) 1-64 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.36 (4H, d), 8.22 (1H, s), 7.99~7.89 (5H, m), 7.77 (1H, d), 7.62~7.49 (13H, m), 7.35 (1H, t), 7.16 (1H, t) 1-65 .delta. = 9.33 (1H, d), 8.59 (1H, d), 8.53 (1H, s), 8.42 (4H, m), 8.28 (1H, d), 8.22 (1H, d), 8.15 (1H, s), 8.09 (1H, d), 7.90 (1H, d), 7.59~7.51 (10H, m), 7.50~7.34 (4H, m), 7.17 (1H, m), 1.53~1.50 (6H, d) 1-66 .delta. = 8.96 (2H, d), 8.60 (1H, s), 8.53 (1H, s), 8.46 (1H, s), 8.29~8.23 (4H, m), 8.19 (1H, s), 8.10 (1H, d), 7.90 (1H, d), 7.66~7.59 (4H, m), 7.56~7.51 (4H, m), 7.47~7.38 (6H, m), 7.31 (1H, m), 1.53~1.50 (6H, d) 1-67 .delta. = 9.39 (1H, s), 8.90 (4H, d), 8.68 (1H, s), 8.54 (1H, s), 8.30 (1H, d), 8.25 (1H, d), 8.15 (1H, d), 7.91 (1H, d), 7.64~7.52 (10H, m), 7.47~7.35 (4H, m), 7.29 (1H, t), 1.53~1.50 (6H, d) 1-68 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.24 (1H, d), 7.94~7.88 (3H, m), 7.80~7.74 (4H, m), 7.57~7.49 (7H, m), 7.38~7.33 (3H, m), 7.24~7.16 (4H, m), 7.05 (1H, m), 1.69 (6H, s) 1-69 .delta. = 9.26 (1H, s), 8.84 (1H, d), 8.49 (1H, s), 8.43 (1H, s), 8.31 (4H, m), 8.25~8.20 (2H, m), 8.07 (1H, s), 7.95 (2H, t), 7.88 (2H, s), 7.73 (1H, t), 7.55 (10H, m), 7.45~7.28 (5H, m), 1.53~1.50 (6H, d) 1-70 .delta. = 9.26 (1H, s), 8.84 (1H, d), 8.49 (1H, s), 8.43 (1H, s), 8.31 (4H, m), 8.25~8.20 (2H, m), 8.07 (1H, s), 7.71 (1H, t), 7.55 (10H, m), 7.45~7.28 (5H, m), 1.53~1.50 (6H, d) 1-71 .delta. = 8.54 (1H, s), 8.33 (1H, s), 8.30 (1H, d), 8.15 (1H, d), 7.96 (1H, d), 7.89 (4H, t), 7.78 (1H, s), 7.59~7.50 (3H, m), 7.44~7.25 (11H, m), 7.23~7.14 (4H, m), 6.85 (1H, t), 1.53~1.50 (6H, d) 1-72 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.24~8.23 (2H, m), 8.00~7.88 (10H, m), 7.77~7.74 (2H, m), 7.56~7.49 (10H, m), 7.38~7.35 (2H, m), 7.25 (2H, d), 7.16 (1H, t), 1.69 (6H, s) 1-78 .delta. = 9.36 (1H, s), 8.89 (2H, d), 8.64 (1H, s), 8.27~8.21 (3H, m), 8.13 (1H, d), 7.79 (1H, s), 7.67~7.50 (9H, m), 7.47~7.35 (4H, m), 7.30~7.17 (2H, m), 1.69 (6H, s) 1-82 .delta. = 9.31 (1H, s), 8.92 (1H, s), 8.57 (1H, s), 8.43 (4H, m), 8.35 (1H, d), 8.31 (1H, d), 8.18 (2H, m), 8.05 (1H, d), 7.63~7.47 (11H, m), 7.45~7.28 (7H, m), 7.20 (1H, m) 1-83 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.35 (2H, d), 8.23~8.19 (2H, m), 8.00~7.93 (5H, m), 7.77 (1H, s), 7.62~7.49 (16H, m), 7.40~7.35 (2H, m), 7.20~7.16 (2H, m) 1-84 .delta. = 9.39 (1H, s), 8.92 (1H, s), 8.90 (4H, m), 8.62 (1H, s), 8.37~8.33 (2H, m), 8.19 (1H, d), 8.11 (1H, d), 7.67~7.47 (11H, m), 7.45~7.28 (7H, m), 7.30 (2H, m), 7.20 (1H, m) 1-93 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.28 (4H, d), 8.12 (1H, d), 7.90~7.98 (3H, m), 7.29~7.69 (21H, m) 1-96 .delta. = 8.55 (1H, d), 8.45 (2H, m), 8.23 (1H, s), 7.90~8.08 (5H, m), 7.79 (4H, d), 7.69 (1H, s), 7.25~7.52 (13H, m) 1-98 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.36 (4H, d), 8.05~7.93 (5H, m), 7.77 (1H, s), 7.56~7.49 (10H, m), 7.35~7.33 (2H, m), 7.16 (1H, t) 1-99 .delta. = 9.25 (1H, s), 8.85 (1H, d), 8.45 (1H, s), 8.38 (1H, d), 8.32 (4H, m), 8.22 (3H, m), 8.07 (1H, s), 8.00 (2H, t), 7.94 (2H, m), 7.76~7.66 (3H, m), 7.66~7.47 (12H, m) 1-100 .delta. = 8.55 (1H, d), 8.45 (2H, m), 8.28 (4H, d), 8.24 (1H, d), 7.90~8.05 (5H, m), 7.69~7.70 (2H, m), 7.23~7.57 (15H, m) 1-110 9.31 (1H, s), 9.29 (1H, s), 8.81 (4H, d), 8.30~8.58 (7H, m), 7.30~7.79 (13H, m) 1-117 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.28 (4H, d), 7.89~7.94 (5H, m), 7.66~7.69 (2H, m), 7.25~7.52 (12H, m), 7.13 (1H, d) 1-119 .delta. = 8.57 (1H, d), 8.46 (1H, d), 8.28 (4H, d), 8.24 (1H, d), 7.89~7.98 (5H, m), 7.66~7.70 (3H, m), 7.25~7.57 (14H, m), 7.13 (1H, d) 1-126 .delta. = 8.56 (1H, d), 8.45 (1H, d), 8.28 (4H, d), 7.89~7.98 (5H, m), 7.66~7.69 (2H, m), 7.25~7.53 (13H, m) 1-163 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.36 (4H, d), 8.22~8.19 (2H, m), 7.99~7.91 (8H, m), 7.58~7.49 (10H, m), 7.35 (1H, t), 7.20~7.16 (2H, m) 1-170 .delta. = 8.55 (1H, d), 8.45 (2H, m), 8.28 (4H, d), 7.94~8.05 (5H, m), 7.75~7.79 (3H, m), 7.68 (2H, d), 7.25~7.53 (13H, m) 1-172 .delta. = 8.57 (1H, d), 8.45 (1H, d), 8.28 (4H, d), 7.89~7.98 (5H, m), 7.66~7.79 (6H, m), 7.25~7.52 (12H, m) 1-176 .delta. = 8.58 (1H, d), 8.45 (1H, d), 8.16 (1H, d), 7.79~7.94 (9H, m), 7.25~7.69 (12H, m) 1-177 .delta. = 8.55 (1H, d), 8.45~8.46 (2H, m), 8.16 (1H, d), 7.83~8.05 (9H, m), 7.69 (1H, s), 7.25~7.58 (11H, m) 1-178 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.09~8.16 (2H, m), 7.79~7.98 (7H, m), 7.24~7.69 (13H, m), 1.72 (6H, s) 1-179 .delta. = 8.57 (1H, d), 8.47 (1H, d), 8.09~8.16 (3H, m), 7.79~7.98 (7H, m), 7.69 (1H, s), 7.24~7.61 (12H, m), 1.72 (6H, s) 1-180 .delta. = 8.54 (1H, d), 8.44~8.46 (2H, m), 8.16 (1H, d), 7.79~8.05 (9H, m), 7.69 (1H, s), 7.25~7.60 (11H, m) 1-181 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.16 (1H, d), 7.79~7.98 (9H, m), 7.25~7.69 (13H, m) 2-2 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.30 (1H, d), 8.19 (1H, d), 8.13 (1H, d), 8.00~7.89 (6H, m), 7.77 (2H, m), 7.62~7.35 (15H, m), 7.20~7.16 (2H, m) 2-3 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.30 (1H, d), 8.19 (1H, d), 8.13 (1H, d), 8.00~7.89 (6H, m), 7.77 (4H, m), 7.62~7.41 (13H, m), 7.25~7.16 (6H, m) 2-7 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.30 (1H, d), 8.19~8.09 (3H, m), 8.00~7.89 (8H, m), 7.78~7.77 (3H, m), 7.62~7.49 (10H, m), 7.38~7.16 (5H, m), 1.69 (6H, s) 2-9 .delta. = 8.55 (1H, d), 8.45 (2H, d), 8.30 (1H, d), 8.19~8.12 (4H, m), 8.00~7.89 (8H, m), 7.77 (2H, m), 7.62~7.49 (11H, m), 7.35 (1H, t), 7.21~7.16 (2H, m) 2-11 .delta. = 8.55 (1H, d), 8.45 (1H, d), 8.30 (1H, d), 8.19 (1H, d), 8.13 (1H, d), 8.00~7.77 (12H, m), 7.62~7.31 (13H, m), 7.20~7.16 (2H, m)
TABLE-US-00022 TABLE 22 Com- Com- pound FD-MS pound FD-MS 1-2 m/z = 579.18 1-11 m/z = 580.17 (C.sub.40H.sub.25N.sub.3S = 579.72) (C.sub.39H.sub.24N.sub.4S = 580.71) 1-12 m/z = 656.20 1-17 m/z = 552.17 (C.sub.45H.sub.28N4S = 656.81) (C.sub.39H.sub.24N.sub.2S = 552.69) 1-23 m/z = 655.21 1-27 m/z = 656.20 (C.sub.46H.sub.29N.sub.3S = 655.81) (C.sub.45H.sub.28N.sub.4S = 656.80) 1-33 m/z = 655.21 1-36 m/z = 656.20 (C.sub.46H.sub.29N.sub.3S = 655.81) (C.sub.45H.sub.28N.sub.4S = 656.80) 1-39 m/z = 656.20 1-40 m/z = 732.23 (C.sub.45H.sub.28N4S = 656.80) (C.sub.51H.sub.32N.sub.4S = 732.90) 1-41 m/z = 732.23 1-42 m/z = 732.23 (C.sub.51H.sub.32N.sub.4S = 732.89) (C.sub.51H.sub.32N.sub.4S = 732.89) 1-46 m/z = 732.23 1-64 m/z = 656.20 (C.sub.51H.sub.32N.sub.4S = 732.89) (C.sub.45H.sub.28N.sub.4S = 656.80) 1-65 m/z = 695.24 1-66 m/z = 695.24 (C.sub.49H.sub.33N.sub.3S = 695.87) (C.sub.49H.sub.33N.sub.3S = 695.87) 1-67 m/z = 696.23 1-68 m/z = 669.22 (C.sub.48H.sub.32N.sub.4S = 696.86) (C.sub.47H.sub.31N.sub.3S = 669.83) 1-69 m/z = 771.27 1-70 m/z = 772.27 (C.sub.55H.sub.37N.sub.3S = 771.97) (C.sub.54H.sub.36N.sub.4S = 772.96) 1-71 m/z = 733.26 1-72 m/z = 772.27 (C.sub.52H.sub.35N.sub.3S = 733.93) (C.sub.54H.sub.36N.sub.4S = 772.96) 1-78 m/z = 696.23 1-82 m/z = 744.23 (C.sub.48H.sub.32N.sub.4S = 696.86) (C.sub.52H.sub.32N.sub.4S = 744.90) 1-83 m/z = 744.23 1-84 m/z = 745.23 (C.sub.52H.sub.32N.sub.4S = 744.90) (C.sub.51H.sub.31N.sub.5S = 745.89) 1-93 m/z = 745.23 1-96 m/z = 685.16 (C.sub.51H.sub.31N.sub.5S = 745.89) (C.sub.46H.sub.27N.sub.3S.sub.2 = 685.86) 1-98 m/z = 686.16 1-99 m/z = 761.20 (C.sub.45H.sub.26N.sub.4S.sub.2 = 686.84) (C.sub.51H.sub.31N.sub.3S.sub.2 = 761.95) 1-100 m/z = 762.19 1-110 m/z = 686.16 (C.sub.51H.sub.30N.sub.4S.sub.2 = 762.94) (C.sub.45H.sub.26N.sub.4S.sub.2 = 686.84) 1-117 m/z = 670.18 1-119 m/z = 746.21 (C.sub.45H.sub.26N.sub.4OS = 670.78) (C.sub.51H.sub.30N.sub.4OS = 746.88) 1-126 m/z = 670.18 1-163 m/z = 656.80 (C.sub.45H.sub.26N.sub.4OS = 670.78) (C.sub.45H.sub.28N.sub.4S = 656.20) 1-170 m/z = 762.19 1-172 m/z = 746.21 (C.sub.51H.sub.30N.sub.4S.sub.2 = 762.94) (C.sub.51H.sub.30N.sub.4OS = 746.88) 1-176 m/z = 643.17 1-177 m/z = 659.15 (C.sub.44H.sub.25N.sub.3OS = 643.75) (C.sub.44H.sub.25N.sub.3S.sub.2 = 659.82) 1-178 m/z = 669.22 1-179 m/z = 669.22 (C.sub.47H.sub.31N.sub.3S = 669.83) (C.sub.47H.sub.31N.sub.3S = 669.83) 1-180 m/z = 659.15 1-181 m/z = 643.17 (C.sub.44H.sub.25N.sub.3S.sub.2 = 659.82) (C.sub.44H.sub.25N.sub.3OS = 643.75)
TABLE-US-00023 TABLE 23 Compound FD-Mass Compound FD-Mass 2-1 m/z = 742.94 (C54H34N2S = 742.24) 2-2 m/z = 666.84 (C48H30N2S = 666.21) 2-3 m/z = 742.94 (C54H34N2S = 742.24) 2-4 m/z = 742.94 (C54H34N2S = 742.24) 2-5 m/z = 716.90 (C52H32N2S = 716.23) 2-6 m/z = 716.90 (C52H32N2S = 716.23) 2-7 m/z = 783.00 (C57H38N2S = 782.28) 2-8 m/z = 783.00 (C57H38N2S = 782.28) 2-9 m/z = 772.98 (C54H32N2S2 = 772.20) 2-10 m/z = 772.98 (C54H32N2S2 = 772.20) 2-11 m/z = 756.92 (C54H32N2OS = 756.22) 2-12 m/z = 756.92 (C54H32N2OS = 756.22) 2-13 m/z = 666.84 (C48H30N2S = 666.21) 2-14 m/z = 742.94 (C54H34N2S = 742.24) 2-15 m/z = 742.94 (C54H34N2S = 742.24) 2-16 m/z = 716.90 (C52H32N2S = 716.23) 2-17 m/z = 716.90 (C52H32N2S = 716.23) 2-18 m/z = 783.00 (C57H38N2S = 782.28) 2-19 m/z = 783.00 (C57H38N2S = 782.28) 2-20 m/z = 772.98 (C54H32N2S2 = 772.20) 2-21 m/z = 756.92 (C54H32N2OS = 756.22) 2-22 m/z = 756.92 (C54H32N2OS = 756.22) 2-23 m/z = 772.98 (C54H32N2S2 = 772.20) 2-24 m/z = 666.84 (C48H30N2S = 666.21) 2-25 m/z = 742.94 (C54H34N2S = 742.24) 2-26 m/z = 742.94 (C54H34N2S = 742.24) 2-27 m/z = 783.00 (C57H38N2S = 782.28) 2-28 m/z = 772.98 (C54H32N2S2 = 772.20) 2-29 m/z = 756.92 (C54H32N2OS = 756.22) 2-30 m/z = 772.98 (C54H32N2S2 = 772.20) 2-31 m/z = 783.00 (C57H38N2S = 782.28) 2-32 m/z = 756.92 (C54H32N2OS = 756.22) 2-33 m/z = 666.84 (C48H30N2S = 666.21) 2-34 m/z = 742.94 (C54H34N2S = 742.24) 2-35 m/z = 742.94 (C54H34N2S = 742.24) 2-36 m/z = 772.98 (C54H32N2S2 = 772.20) 2-37 m/z = 756.92 (C54H32N2OS = 756.22) 2-38 m/z = 772.98 (C54H32N2S2 = 772.20) 2-39 m/z = 783.00 (C57H38N2S = 782.28) 2-40 m/z = 756.92 (C54H32N2OS = 756.22) 2-41 m/z = 666.84 (C48H30N2S = 666.21) 2-42 m/z = 742.94 (C54H34N2S = 742.24) 2-43 m/z = 742.94 (C54H34N2S = 742.24) 2-44 m/z = 772.98 (C54H32N2S2 = 772.20) 2-45 m/z = 756.92 (C54H32N2OS = 756.22) 2-46 m/z = 772.98 (C54H32N2S2 = 772.20) 2-47 m/z = 783.00 (C57H38N2S = 782.28) 2-48 m/z = 756.92 (C54H32N2OS = 756.22) 2-49 m/z = 666.84 (C48H30N2S = 666.21) 2-50 m/z = 742.94 (C54H34N2S = 742.24) 2-51 m/z = 742.94 (C54H34N2S = 742.24) 2-52 m/z = 772.98 (C54H32N2S2 = 772.20) 2-53 m/z = 756.92 (C54H32N2OS = 756.22) 2-54 m/z = 772.98 (C54H32N2S2 = 772.20) 2-55 m/z = 783.00 (C57H38N2S = 782.28) 2-56 m/z = 756.92 (C54H32N2OS = 756.22) 2-57 m/z = 666.84 (C48H30N2S = 666.21) 2-58 m/z = 742.94 (C54H34N2S = 742.24) 2-59 m/z = 742.94 (C54H34N2S = 742.24) 2-60 m/z = 772.98 (C54H32N2S2 = 772.20) 2-61 m/z = 756.92 (C54H32N2OS = 756.22) 2-62 m/z = 772.98 (C54H32N2S2 = 772.20) 2-63 m/z = 783.00 (C57H38N2S = 782.28) 2-64 m/z = 756.92 (C54H32N2OS = 756.22) 2-65 m/z = 666.84 (C48H30N2S = 666.21) 2-66 m/z = 742.94 (C54H34N2S = 742.24) 2-67 m/z = 742.94 (C54H34N2S = 742.24) 2-68 m/z = 772.98 (C54H32N2S2 = 772.20) 2-69 m/z = 756.92 (C54H32N2OS = 756.22) 2-70 m/z = 772.98 (C54H32N2S2 = 772.20) 2-71 m/z = 783.00 (C57H38N2S = 782.28) 2-72 m/z = 756.92 (C54H32N2OS = 756.22) 2-73 m/z = 666.84 (C48H30N2S = 666.21) 2-74 m/z = 742.94 (C54H34N2S = 742.24) 2-75 m/z = 742.94 (C54H34N2S = 742.24) 2-76 m/z = 772.98 (C54H32N2S2 = 772.20) 2-77 m/z = 756.92 (C54H32N2OS = 756.22) 2-78 m/z = 772.98 (C54H32N2S2 = 772.20) 2-79 m/z = 783.00 (C57H38N2S = 782.28) 2-80 m/z = 756.92 (C54H32N2OS = 756.22) 2-81 m/z = 666.84 (C48H30N2S = 666.21) 2-82 m/z = 742.94 (C54H34N2S = 742.24) 2-83 m/z = 742.94 (C54H34N2S = 742.24) 2-84 m/z = 772.98 (C54H32N2S2 = 772.20) 2-85 m/z = 756.92 (C54H32N2OS = 756.22) 2-86 m/z = 772.98 (C54H32N2S2 = 772.20) 2-87 m/z = 783.00 (C57H38N2S = 782.28) 2-88 m/z = 756.92 (C54H32N2OS = 756.22) 2-89 m/z = 666.84 (C48H30N2S = 666.21) 2-90 m/z = 742.94 (C54H34N2S = 742.24) 2-91 m/z = 742.94 (C54H34N2S = 742.24) 2-92 m/z = 772.98 (C54H32N2S2 = 772.20) 2-93 m/z = 756.92 (C54H32N2OS = 756.22) 2-94 m/z = 772.98 (C54H32N2S2 = 772.20) 2-95 m/z = 783.00 (C57H38N2S = 782.28) 2-96 m/z = 756.92 (C54H32N2OS = 756.22) 2-97 m/z = 666.84 (C48H30N2S = 666.21) 2-98 m/z = 742.94 (C54H34N2S = 742.24) 2-99 m/z = 742.94 (C54H34N2S = 742.24) 2-100 m/z = 772.98 (C54H32N2S2 = 772.20) 2-101 m/z = 756.92 (C54H32N2OS = 756.22) 2-102 m/z = 772.98 (C54H32N2S2 = 772.20) 2-103 m/z = 783.00 (C57H38N2S = 782.28) 2-104 m/z = 756.92 (C54H32N2OS = 756.22)
Table 21 shows NMR values, and Tables 22 and 23 show measured values by field desorption mass spectrometry (FD-MS).
FIG. 4 illustrates a measurement graph of LTPL of Compound 1-2 at a wavelength of 363 nm.
FIG. 5 illustrates a measurement graph of PL of Compound 1-2 at a wavelength of 238 nm.
FIG. 6 illustrates a UV absorption spectrum of Compound 1-2.
FIG. 7 illustrates a measurement graph of LTPL of Compound 1-11 at a wavelength of 339 nm.
FIG. 8 illustrates a measurement graph of PL of Compound 1-11 at a wavelength of 234 nm.
FIG. 9 illustrates a UV absorption spectrum of Compound 1-11.
FIG. 10 illustrates a measurement graph of LTPL of Compound 1-23 at a wavelength of 241 nm.
FIG. 11 illustrates a measurement graph of PL of Compound 1-23 at a wavelength of 241 nm.
FIG. 12 illustrates a UV absorption spectrum of Compound 1-23.
FIG. 13 illustrates a measurement graph of LTPL of Compound 1-27 at a wavelength of 340 nm.
FIG. 14 illustrates a measurement graph of PL of Compound 1-27 at a wavelength of 241 nm.
FIG. 15 illustrates a UV absorption spectrum of Compound 1-27.
FIG. 16 illustrates a measurement graph of LTPL of Compound 1-33 at a wavelength of 291 nm.
FIG. 17 illustrates a measurement graph of PL of Compound 1-33 at a wavelength of 239 nm.
FIG. 18 illustrates a UV absorption spectrum of Compound 1-33.
FIG. 19 illustrates a measurement graph of LTPL of Compound 1-39 at a wavelength of 259 nm.
FIG. 20 illustrates a measurement graph of PL of Compound 1-39 at a wavelength of 259 nm.
FIG. 21 illustrates a UV absorption spectrum of Compound 1-39.
FIG. 22 illustrates a measurement graph of LTPL of Compound 1-41 at a wavelength of 260 nm.
FIG. 23 illustrates a measurement graph of PL of Compound 1-41 at a wavelength of 260 nm.
FIG. 24 illustrates a UV absorption spectrum of Compound 1-41.
FIG. 25 illustrates a measurement graph of LTPL of Compound 1-65 at a wavelength of 361 nm.
FIG. 26 illustrates a measurement graph of PL of Compound 1-65 at a wavelength of 235 nm.
FIG. 27 illustrates a UV absorption spectrum of Compound 1-65.
FIG. 28 illustrates a measurement graph of LTPL of Compound 1-66 at a wavelength of 360 nm.
FIG. 29 illustrates a measurement graph of PL of Compound 1-66 at a wavelength of 307 nm.
FIG. 30 illustrates a UV absorption spectrum of Compound 1-66.
FIG. 31 illustrates a measurement graph of LTPL of Compound 1-67 at a wavelength of 361 nm.
FIG. 32 illustrates a measurement graph of PL of Compound 1-67 at a wavelength of 266 nm.
FIG. 33 illustrates a UV absorption spectrum of Compound 1-67.
FIG. 34 illustrates a measurement graph of LTPL of Compound 1-69 at a wavelength of 344 nm.
FIG. 35 illustrates a measurement graph of PL of Compound 1-69 at a wavelength of 308 nm.
FIG. 36 illustrates a UV absorption spectrum of Compound 1-69.
FIG. 37 illustrates a measurement graph of LTPL of Compound 1-70 at a wavelength of 344 nm.
FIG. 38 illustrates a measurement graph of PL of Compound 1-70 at a wavelength of 267 nm.
FIG. 39 illustrates a UV absorption spectrum of Compound 1-70.
FIG. 40 illustrates a measurement graph of LTPL of Compound 1-71 at a wavelength of 344 nm.
FIG. 41 illustrates a measurement graph of PL of Compound 1-71 at a wavelength of 241 nm.
FIG. 42 illustrates a UV absorption spectrum of Compound 1-71.
FIG. 43 illustrates a measurement graph of LTPL of Compound 1-78 at a wavelength of 361 nm.
FIG. 44 illustrates a measurement graph of PL of Compound 1-78 at a wavelength of 263 nm.
FIG. 45 illustrates a UV absorption spectrum of Compound 1-78.
FIG. 46 illustrates a measurement graph of LTPL of Compound 1-82 at a wavelength of 344 nm.
FIG. 47 illustrates a measurement graph of PL of Compound 1-82 at a wavelength of 307 nm.
FIG. 48 illustrates a UV absorption spectrum of Compound 1-82.
FIG. 49 illustrates a measurement graph of LTPL of Compound 1-84 at a wavelength of 363 nm.
FIG. 50 illustrates a measurement graph of PL of Compound 1-84 at a wavelength of 298 nm.
FIG. 51 illustrates a UV absorption spectrum of Compound 1-84.
FIG. 52 illustrates a measurement graph of LTPL of Compound 1-99 at a wavelength of 355 nm.
FIG. 53 illustrates a measurement graph of PL of Compound 1-99 at a wavelength of 355 nm.
FIG. 54 illustrates a UV absorption spectrum of Compound 1-99.
Experimental Example
Experimental Example 1
1) Manufacture of Organic Light Emitting Device
A glass substrate, in which ITO was thinly coated to have a thickness of 1,500 .ANG., was ultrasonically washed with distilled water. When the washing with distilled water is finished, the glass substrate was ultrasonically washed with a solvent such as acetone, methanol, and isopropyl alcohol, dried and then was subjected to UVO treatment for 5 minutes by using UV in a UV washing machine. Thereafter, the substrate was transferred to a plasma washing machine (PT), and then was subjected to plasma treatment in a vacuum state for an ITO work function and in order to remove a residual film, and was transferred to a thermal deposition equipment for organic deposition.
As the common layers, the hole injection layer 4,4',4''-tris[2-naphthyl(phenyl)amino]triphenylamine (2-TNATA) and the hole transport layer N,N'-di(1-naphthyl)-N,N'-diphenyl-(1,1'-biphenyl)-4,4'-diamine (NPB) were formed on the ITO transparent electrode (positive electrode).
A light emitting layer was thermally vacuum deposited thereon as follows. The light emitting layer was deposited to have a thickness of 400 .ANG. by using a compound described in the following Table 24 as a host and tris(2-phenylpyridine)iridium (Ir(ppy).sub.3) as a green phosphorescent dopant to dope the host with Ir(ppy).sub.3 in an amount of 7%. Thereafter, BCP as a hole blocking layer was deposited to have a thickness of 60 .ANG., and Alq.sub.3 as an electron transport layer was deposited to have a thickness of 200 .ANG. thereon. Finally, an organic electroluminescence device was manufactured by depositing lithium fluoride (LiF) to have a thickness of 10 .ANG. on the electron transport layer to form an electron injection layer, and then depositing an aluminum (Al) negative electrode to have a thickness of 1,200 .ANG. on the electron injection layer to form a negative electrode.
Meanwhile, all the organic compounds required for manufacturing an OLED device were subjected to vacuum sublimed purification under 10.sup.-6 to 10.sup.-8 torr for each material, and used for the manufacture of OLED.
2) Driving Voltage and Light Emitting Efficiency of Organic Electroluminescence Device
For the organic electroluminescence device manufactured as described above, electroluminescence (EL) characteristics were measured by M7000 manufactured by McScience Inc., and based on the measurement result thereof, T.sub.90 was measured by a lifetime measurement equipment (M6000) manufactured by McScience Inc. when the reference brightness was 6,000 cd/m.sup.2. Characteristics of the organic electroluminescence device of the present invention are as shown in the following Table 24.
TABLE-US-00024 TABLE 24 Driving voltage Efficiency Color coordinate (x, Lifetime Compound (V) (cd/A) y) (T.sub.90) Example 1 1-2 4.05 67.2 (0.294, 0.654) 142 Example 2 1-11 3.90 68.9 (0.293, 0.653) 151 Example 3 1-17 3.82 60.5 (0.283, 0.643) 186 Example 4 1-23 4.39 64.1 (0.296, 0.654) 123 Example 5 1-27 4.19 66.1 (0.297, 0.653) 133 Example 6 1-33 4.40 63.2 (0.305, 0.654) 120 Example 7 1-39 3.88 69.9 (0.296, 0.652) 154 Example 8 1-41 4.11 76.6 (0.296, 0.675) 137 Example 9 1-64 4.00 75.6 (0.296, 0.674) 134 Example 10 1-65 3.83 68.3 (0.272, 0.683) 149 Example 11 1-66 4.02 67.1 (0.293, 0.654) 145 Example 12 1-67 3.80 70.2 (0.284, 0.682) 160 Example 13 1-69 4.26 65.4 (0.295, 0.693) 127 Example 14 1-70 4.39 67.0 (0.296, 0.701) 140 Example 15 1-71 4.25 64.7 (0.295, 0.675) 130 Example 16 1-76 4.22 68.7 (0.294, 0.676) 133 Example 17 1-77 3.91 75.5 (0.280, 0.678) 148 Example 18 1-78 3.81 72.5 (0.281, 0.679) 158 Example 19 1-79 3.96 67.2 (0.273, 0.688) 137 Example 20 1-80 3.79 73.1 (0.295, 0.659) 169 Example 21 1-82 3.91 68.2 (0.274, 0.684) 147 Example 22 1-84 3.70 70.1 (0.293, 0.656) 156 Example 23 1-85 3.99 69.2 (0.273, 0.687) 147 Example 24 1-86 3.71 78.1 (0.295, 0.658) 170 Example 25 1-91 3.98 61.2 (0.274, 0.683) 158 Example 26 1-92 3.73 76.1 (0.293, 0.655) 159 Example 27 1-93 3.81 67.6 (0.286, 0.650) 137 Example 28 1-94 4.20 66.7 (0.293, 0.664) 152 Example 29 1-95 3.90 79.2 (0.294, 0.662) 199 Example 30 1-96 3.79 69.3 (0.272, 0.683) 149 Example 31 1-98 3.71 65.3 (0.271, 0.681) 179 Example 32 1-99 4.23 62.7 (0.293, 0.654) 145 Example 33 1-100 3.93 74.2 (0.294, 0.652) 190 Example 34 1-109 3.82 69.4 (0.301, 0.693) 187 Example 35 1-110 4.12 64.4 (0.302, 0.693) 227 Example 36 1-111 3.88 68.9 (0.286, 0.685) 177 Example 37 1-112 3.93 64.9 (0.272, 0.673) 169 Example 38 1-113 4.25 67.7 (0.293, 0.654) 195 Example 39 1-114 3.11 70.2 (0.314, 0.692) 178 Example 40 1-117 3.80 67.5 (0.276, 0.651) 190 Example 41 1-118 3.79 69.3 (0.272, 0.683) 149 Example 42 1-119 4.29 65.1 (0.285, 0.695) 180 Example 43 1-125 3.73 67.0 (0.276, 0.689) 190 Example 44 1-126 4.09 72.5 (0.301, 0.685) 198 Example 45 1-127 3.81 67.6 (0.286, 0.650) 137 Example 46 1-128 3.91 68.2 (0.274, 0.684) 147 Example 47 1-138 3.70 70.1 (0.293, 0.656) 156 Example 48 1-139 4.11 76.6 (0.296, 0.675) 137 Example 49 1-140 4.00 75.6 (0.296, 0.674) 134 Example 50 1-155 3.83 68.3 (0.272, 0.683) 149 Example 51 1-156 4.05 67.2 (0.294, 0.654) 142 Example 52 1-157 3.90 68.9 (0.293, 0.653) 151 Example 53 1-158 3.82 60.5 (0.283, 0.643) 186 Example 54 1-160 3.73 67.0 (0.276, 0.689) 190 Example 55 1-162 3.71 78.1 (0.295, 0.658) 170 Example 56 1-164 3.78 68.9 (0.286, 0.685) 177 Example 57 1-165 4.29 65.1 (0.285, 0.695) 180 Example 58 1-170 4.21 68.2 (0.294, 0.644) 217 Example 59 1-172 3.81 70.1 (0.313, 0.706) 186 Example 60 1-174 3.73 67.0 (0.276, 0.689) 190 Example 61 1-176 3.78 68.9 (0.286, 0.685) 177 Example 62 1-177 3.83 64.9 (0.272, 0.673) 169 Example 63 1-178 4.25 67.7 (0.293, 0.654) 195 Example 64 1-179 3.81 70.2 (0.314, 0.692) 178 Example 65 1-180 3.89 65.4 (0.295, 0.693) 167 Example 66 1-181 3.73 67.0 (0.276, 0.689) 190 Comparative Example 1 CBP 5.23 41.1 (0.285, 0.681) 50 Comparative Example 2 ref 4 4.82 58.8 (0.294, 0.654) 55 Comparative Example 3 ref 5 4.20 62.3 (0.284, 0.695) 31 Comparative Example 4 ref 6 4.66 61.2 (0.296, 0.676) 80 Comparative Example 5 ref 7 4.10 59.7 (0.276, 0.684) 82 Comparative Example 6 ref 8 4.92 52.2 (0.286, 0.644) 65 Comparative Example 7 ref 9 4.91 56.3 (0.286, 0.644) 73
As can be seen from the results in Table 24, the organic electroluminescence device using a light emitting layer material of the organic electroluminescence device of the present invention had a low driving voltage, an enhanced light emitting efficiency, and a significantly improved lifetime compared to those in Comparative Examples 1 to 7.
Meanwhile, when phenylene is positioned between carbazole and triazine as in Comparative Example 2, the lifetime is reduced because electrons in the LUMO region fail to be stabilized. Further, when there is no carbazole as in Comparative Example 3, the hole mobility deteriorates, and the equilibrium between holes and electrons in the light emitting layer collapses, thereby leading to a decrease in lifetime. In addition, in the case of a compound including dibenzofuran as in Comparative Example 4, the lifetime is reduced because electrons in the LUMO region fail to be stabilized. Furthermore, when a substituent is bonded to the 2nd and 6th positions of dibenzothiophene as in Comparative Example 5, the equilibrium between holes and electrons in the light emitting layer collapses, thereby leading to a decrease in lifetime. Further, as in Comparative Examples 6 and 7, when a heteroaryl group including at least one N is not bonded to the position of Ar1 of Chemical Formula 1 of the present invention, the equilibrium between holes and electrons collapses because there is no substituent which stabilizes electrons, and as a result, a result of reducing the efficiency or lifetime is obtained.
<Experimental Example 2> Manufacture of Organic Light Emitting Device
A glass substrate, in which ITO was thinly coated to have a thickness of 1,500 .ANG., was ultrasonically washed with distilled water. When the washing with distilled water is finished, the glass substrate was ultrasonically washed with a solvent such as acetone, methanol, and isopropyl alcohol, dried and then was subjected to UVO treatment for 5 minutes by using UV in a UV washing machine. Thereafter, the substrate was transferred to a plasma washing machine (PT), and then was subjected to plasma treatment in a vacuum state for an ITO work function and in order to remove a residual film, and was transferred to a thermal deposition equipment for organic deposition.
As the common layers, the hole injection layer 4,4',4''-tris[2-naphthyl(phenyl)amino]triphenylamine (2-TNATA) and the hole transport layer N,N'-di(1-naphthyl)-N,N'-diphenyl-(1,1'-biphenyl)-4,4'-diamine (NPB) were formed on the ITO transparent electrode (positive electrode).
A light emitting layer was thermally vacuum deposited thereon as follows. The light emitting layer was deposited to have a thickness of 400 .ANG. by using a compound described in Chemical Formula 1 and a compound described in Chemical Formula 2 as hosts from the individual supply source, and was deposited by doping the host with Ir(ppy).sub.3 as a green phosphorescent dopant in an amount of 7%. Thereafter, BCP as a hole blocking layer was deposited to have a thickness of 60 .ANG., and Alq.sub.3 as an electron transport layer was deposited to have a thickness of 200 .ANG. thereon. Finally, an organic electroluminescence device was manufactured by depositing lithium fluoride (LiF) to have a thickness of 10 .ANG. on the electron transport layer to form an electron injection layer, and then depositing an aluminum (Al) negative electrode to have a thickness of 1,200 .ANG. on the electron injection layer to form a negative electrode.
Meanwhile, all the organic compounds required for manufacturing an OLED device were subjected to vacuum sublimed purification under 10.sup.-6 to 10.sup.-8 torr for each material, and used for the manufacture of OLED.
<Experimental Example 3> Manufacture of Organic Light Emitting Device
A glass substrate, in which ITO was thinly coated to have a thickness of 1,500 .ANG., was ultrasonically washed with distilled water. When the washing with distilled water is finished, the glass substrate was ultrasonically washed with a solvent such as acetone, methanol, and isopropyl alcohol, dried and then was subjected to UVO treatment for 5 minutes by using UV in a UV washing machine. Thereafter, the substrate was transferred to a plasma washing machine (PT), and then was subjected to plasma treatment in a vacuum state for an ITO work function and in order to remove a residual film, and was transferred to a thermal deposition equipment for organic deposition.
As the common layers, the hole injection layer 4,4',4''-tris[2-naphthyl(phenyl)amino]triphenylamine (2-TNATA) and the hole transport layer N,N'-di(1-naphthyl)-N,N'-diphenyl-(1,1'-biphenyl)-4,4'-diamine (NPB) were formed on the ITO transparent electrode (positive electrode).
A light emitting layer was thermally vacuum deposited thereon as follows. The light emitting layer was deposited to have a thickness of 400 .ANG. by pre-mixing a compound described in Chemical Formula 1 and a compound described in Chemical Formula 2 as hosts, and then was deposited from one supply source by doping the host with Ir(ppy).sub.3 as a green phosphorescent dopant in an amount of 7%. Thereafter, BCP as a hole blocking layer was deposited to have a thickness of 60 .ANG., and Alq.sub.3 as an electron transport layer was deposited to have a thickness of 200 .ANG. thereon. Finally, an organic electroluminescence device was manufactured by depositing lithium fluoride (LiF) to have a thickness of 10 .ANG. on the electron transport layer to form an electron injection layer, and then depositing an aluminum (Al) negative electrode to have a thickness of 1,200 .ANG. on the electron injection layer to form a negative electrode.
Meanwhile, all the organic compounds required for manufacturing an OLED device were subjected to vacuum sublimed purification under 10.sup.-6 to 10.sup.-8 torr for each material, and used for the manufacture of OLED.
The driving voltages and light emitting efficiencies of the organic electroluminescence devices according to Experimental Examples 2 and 3 are as follows.
For the organic electroluminescence device manufactured as described above, electroluminescence (EL) characteristics were measured by M7000 manufactured by McScience Inc., and based on the measurement result thereof, T.sub.90 was measured by a lifetime measurement equipment (M6000) manufactured by McScience Inc. when the reference brightness was 6,000 cd/m.sup.2.
Characteristics of the organic electroluminescence device of the present invention are as shown in the following Tables 25 to 27. For reference, Table 25 is an example in which the two host compounds in Experimental Example 2 were simultaneously deposited by using an individual supply source, Table 26 is an example in which the two light emitting compounds in Experimental Example 3 were pre-mixed, and then deposited by using one supply source, and Table 27 is an example in which the single host material in Experimental Example 2 was applied.
##STR00295##
TABLE-US-00025 TABLE 25 Driving Light emitting layer Mixture weight voltage Efficiency Color coordinate Lifetime compound ratio (V) (cd/A) (x, y) (T.sub.90) Example 67 1-39:2-2 1:8 4.74 53.2 (0.251, 0.714) 201 Example 68 1:5 4.72 58.2 (0.241, 0.711) 224 Example 69 1:2 4.33 77.2 (0.236, 0.717) 384 Example 70 1:1 4.42 76.8 (0.247, 0.727) 321 Example 71 2:1 4.62 70.2 (0.233, 0.714) 280 Example 72 5:1 4.37 69.3 (0.243, 0.714) 178 Example 73 8:1 4.24 69.0 (0.267, 0.712) 164 Example 74 1-39:2-3 1:2 4.37 73.2 (0.241, 0.711) 373 Example 75 1:1 4.48 72.8 (0.257, 0.729) 319 Example 76 2:1 4.63 71.2 (0.231, 0.714) 271 Example 77 1-40:2-2 1:2 4.36 76.2 (0.241, 0.711) 381 Example 78 1:1 4.49 74.8 (0.247, 0.727) 323 Example 79 2:1 4.64 72.1 (0.253, 0.694) 290 Example 80 1-40:2-3 1:2 4.34 72.2 (0.241, 0.711) 331 Example 81 1:1 4.41 70.8 (0.241, 0.714) 291 Example 82 2:1 4.61 70.2 (0.231, 0.711) 259 Example 83 1-67:2-2 1:2 4.34 77.2 (0.246, 0.717) 379 Example 84 1:1 4.41 76.7 (0.251, 0.714) 311 Example 85 2:1 4.66 73.1 (0.241, 0.711) 279 Example 86 1-67:2-3 1:2 4.44 76.2 (0.246, 0.697) 340 Example 87 1:1 4.51 75.7 (0.247, 0.727) 311 Example 88 2:1 4.96 73.2 (0.223, 0.714) 262 Example 89 1-46:2-2 1:2 4.46 71.2 (0.261, 0.711) 301 Example 90 1:1 4.59 70.7 (0.241, 0.711) 289 Example 91 2:1 4.61 69.1 (0.256, 0.717) 261 Example 92 1-46:2-3 1:2 4.41 70.8 (0.221, 0.691) 291 Example 93 1:1 4.53 69.3 (0.247, 0.727) 269 Example 94 2:1 4.59 68.2 (0.243, 0.714) 252 Example 95 1-41:2-2 1:2 4.46 70.8 (0.267, 0.712) 296 Example 96 1:1 4.60 70.4 (0.241, 0.721) 284 Example 97 2:1 4.61 68.8 (0.252, 0.737) 255 Example 98 1-41:2-3 1:2 4.41 69.3 (0.221, 0.691) 285 Example 99 1:1 4.53 68.8 (0.247, 0.687) 261 Example 100 2:1 4.61 67.2 (0.242, 0.714) 247 Comparative Ref 3:2-2.sup. 1:2 4.31 65.2 (0.251, 0.711) 210 Example 8 Comparative 1:1 4.63 63.3 (0.266, 0.707) 192 Example 9 Comparative 2:1 4.79 61.1 (0.241, 0.691) 182 Example 10 Comparative Ref 3:2-3.sup. 1:2 4.31 64.7 (0.231, 0.721) 199 Example 11 Comparative 1:1 4.63 62.3 (0.266, 0.687) 189 Example 12 Comparative 2:1 4.79 61.2 (0.241, 0.711) 172 Example 13 Comparative .sup. 1-39:Ref 1 1:2 4.33 70.3 (0.246, 0.717) 182 Example 14 Comparative 1:1 4.42 69.6 (0.247, 0.727) 179 Example 15 Comparative 2:1 4.62 68.1 (0.253, 0.694) 162 Example 16 Comparative .sup. 1-39:Ref 2 1:2 4.32 69.7 (0.243, 0.714) 178 Example 17 Comparative 1:1 4.44 68.3 (0.241, 0.711) 174 Example 18 Comparative 2:1 4.45 66.2 (0.241, 0.711) 159 Example 19 Comparative .sup. 1-40:Ref 1 1:2 4.36 69.7 (0.241, 0.691) 177 Example 20 Comparative 1:1 4.49 69.1 (0.257, 0.727) 171 Example 21 Comparative 2:1 4.64 68.3 (0.243, 0.714) 167 Example 22 Comparative .sup. 1-40:Ref 2 1:2 4.44 68.8 (0.241, 0.714) 166 Example 23 Comparative 1:1 4.45 67.9 (0.241, 0.711) 165 Example 24 Comparative 2:1 4.36 66.6 (0.246, 0.697) 161 Example 25 Comparative .sup. 1-67:Ref 1 1:2 4.46 69.3 (0.266, 0.717) 185 Example 26 Comparative 1:1 4.59 69.2 (0.241, 0.714) 179 Example 27 Comparative 2:1 4.61 68.1 (0.241, 0.711) 171 Example 28 Comparative .sup. 1-67:Ref 2 1:2 4.41 68.6 (0.247, 0.727) 183 Example 29 Comparative 1:1 4.53 67.2 (0.223, 0.694) 175 Example 30 Comparative 2:1 4.59 66.1 (0.241, 0.714) 161 Example 31 Comparative .sup. 1-46:Ref 1 1:2 4.46 69.3 (0.241, 0.711) 180 Example 32 Comparative 1:1 4.59 68.2 (0.251, 0.691) 177 Example 33 Comparative 2:1 4.61 67.1 (0.246, 0.717) 163 Example 34 Comparative .sup. 1-46:Ref 2 1:2 4.61 68.2 (0.251, 0.711) 172 Example 35 Comparative 1:1 4.41 66.4 (0.247, 0.727) 168 Example 36 Comparative 2:1 4.53 65.1 (0.243, 0.694) 161 Example 37 Comparative .sup. 1-41:Ref 1 1:2 4.47 69.1 (0.241, 0.721) 175 Example 38 Comparative 1:1 4.44 67.9 (0.251, 0.693) 172 Example 39 Comparative 2:1 4.63 67.0 (0.246, 0.711) 158 Example 40 Comparative .sup. 1-41:Ref 2 1:2 4.61 67.7 (0.251, 0.721) 167 Example 41 Comparative 1:1 4.35 66.1 (0.237, 0.737) 163 Example 42 Comparative 2:1 4.56 64.7 (0.243, 0.699) 155 Example 43
TABLE-US-00026 TABLE 26 Driving Light emitting layer Mixture weight voltage Efficiency Color coordinate Lifetime compound ratio (V) (cd/A) (x, y) (T.sub.90) Example 101 1-39:2-2 1:8 4.72 53.7 (0.253, 0.714) 234 Example 102 1:5 4.68 58.9 (0.241, 0.691) 252 Example 103 1:2 4.33 77.2 (0.256, 0.717) 513 Example 104 1:1 4.42 76.8 (0.247, 0.727) 451 Example 105 2:1 4.62 70.2 (0.243, 0.714) 413 Example 106 5:1 4.37 69.3 (0.257, 0.737) 286 Example 107 8:1 4.24 69.0 (0.243, 0.732) 262 Example 108 1-39:2-3 1:2 4.37 73.2 (0.241, 0.691) 401 Example 109 1:1 4.48 72.8 (0.257, 0.729) 449 Example 110 2:1 4.63 71.2 (0.241, 0.714) 401 Example 111 1-40:2-2 1:2 4.36 76.2 (0.241, 0.711) 507 Example 112 1:1 4.49 74.8 (0.247, 0.727) 453 Example 113 2:1 4.64 72.1 (0.233, 0.714) 423 Example 114 1-40:2-3 1:2 4.34 72.2 (0.241, 0.691) 461 Example 115 1:1 4.41 70.8 (0.241, 0.714) 423 Example 116 2:1 4.61 70.2 (0.231, 0.711) 381 Example 117 1-67:2-2 1:2 4.34 77.2 (0.246, 0.717) 503 Example 118 1:1 4.41 76.7 (0.241, 0.694) 440 Example 119 2:1 4.66 73.1 (0.231, 0.711) 409 Example 120 1-67:2-3 1:2 4.44 76.2 (0.246, 0.717) 470 Example 121 1:1 4.51 75.7 (0.247, 0.727) 455 Example 122 2:1 4.96 73.2 (0.253, 0.714) 396 Example 123 1-46:2-2 1:2 4.46 71.2 (0.241, 0.691) 430 Example 124 1:1 4.59 70.7 (0.241, 0.711) 413 Example 125 2:1 4.61 69.1 (0.236, 0.717) 396 Example 126 1-46:2-3 1:2 4.41 70.8 (0.241, 0.711) 421 Example 127 1:1 4.53 69.3 (0.257, 0.727) 393 Example 128 2:1 4.59 68.2 (0.243, 0.694) 382 Example 129 1-41:2-2 1:2 4.46 70.7 (0.241, 0.693) 425 Example 130 1:1 4.62 70.2 (0.241, 0.721) 408 Example 131 2:1 4.65 68.8 (0.236, 0.717) 391 Example 132 1-14:2-3 1:2 4.41 70.2 (0.241, 0.711) 415 Example 133 1:1 4.55 69.0 (0.257, 0.737) 387 Example 134 2:1 4.60 67.9 (0.243, 0.732) 377 Example 135 1-39:2-7 1:2 4.32 70.2 (0.263, 0.704) 425 Example 136 1:1 4.46 72.5 (0.251, 0.683) 386 Example 137 1-39:2-9 1:2 4.42 65.4 (0.271, 0.701) 395 Example 138 1:1 4.63 67.2 (0.266, 0.697) 369 Example 139 1-39:2-11 1:2 4.72 67.5 (0.271, 0.705) 415 Example 140 1:1 4.85 69.4 (0.267, 0.682) 387 Example 141 1-46:2-7 1:2 4.46 64.2 (0.263, 0.662) 432 Example 142 1:1 4.69 65.5 (0.253, 0.672) 385 Comparative Ref 3:2-2.sup. 1:2 4.31 65.2 (0.241, 0.711) 250 Example 44 Comparative 1:1 4.63 63.3 (0.256, 0.697) 241 Example 45 Comparative 2:1 4.79 61.1 (0.241, 0.711) 234 Example 46 Comparative Ref 3:2-3.sup. 1:2 4.31 64.7 (0.231, 0.711) 243 Example 47 Comparative 1:1 4.63 62.3 (0.246, 0.717) 233 Example 48 Comparative 2:1 4.79 61.2 (0.251, 0.691) 220 Example 49 Comparative .sup. 1-39:Ref 1 1:2 4.33 70.3 (0.246, 0.717) 249 Example 50 Comparative 1:1 4.42 69.6 (0.247, 0.727) 229 Example 51 Comparative 2:1 4.62 68.1 (0.243, 0.694) 221 Example 52 Comparative .sup. 1-39:Ref 2 1:2 4.32 69.7 (0.233, 0.714) 231 Example 53 Comparative 1:1 4.44 68.3 (0.241, 0.711) 221 Example 54 Comparative 2:1 4.45 66.2 (0.241, 0.691) 213 Example 55 Comparative .sup. 1-40:Ref 1 1:2 4.36 69.7 (0.231, 0.711) 235 Example 56 Comparative 1:1 4.49 69.1 (0.247, 0.727) 220 Example 57 Comparative 2:1 4.64 68.3 (0.243, 0.714) 210 Example 58 Comparative .sup. 1-40:Ref 2 1:2 4.44 68.8 (0.251, 0.694) 234 Example 59 Comparative 1:1 4.45 67.9 (0.241, 0.711) 221 Example 60 Comparative 2:1 4.36 66.6 (0.246, 0.717) 215 Example 61 Comparative .sup. 1-67:Ref 1 1:2 4.46 69.3 (0.236, 0.697) 229 Example 62 Comparative 1:1 4.59 69.2 (0.241, 0.714) 215 Example 63 Comparative 2:1 4.61 68.1 (0.251, 0.711) 210 Example 64 Comparative .sup. 1-67:Ref 2 1:2 4.41 68.6 (0.247, 0.727) 245 Example 65 Comparative 1:1 4.53 67.2 (0.243, 0.694) 234 Example 66 Comparative 2:1 4.59 66.1 (0.241, 0.714) 229 Example 67 Comparative .sup. 1-46:Ref 1 1:2 4.46 69.3 (0.251, 0.711) 241 Example 68 Comparative 1:1 4.59 68.2 (0.241, 0.691) 235 Example 69 Comparative 2:1 4.61 67.1 (0.246, 0.717) 221 Example 70 Comparative .sup. 1-46:Ref 2 1:2 4.61 68.2 (0.241, 0.711) 231 Example 71 Comparative 1:1 4.41 66.4 (0.237, 0.727) 227 Example 72 Comparative 2:1 4.53 65.1 (0.243, 0.714) 219 Example 73 Comparative .sup. 1-41:Ref 1 1:2 4.49 69.0 (0.221, 0.721) 236 Example 74 Comparative 1:1 4.62 67.9 (0.231,0.621) 230 Example 75 Comparative 2:1 4.63 66.6 (0.256, 0.727) 216 Example 76 Comparative .sup. 1-41:Ref 2 1:2 4.64 67.7 (0.251, 0.731) 225 Example 77 Comparative 1:1 4.43 66.0 (0.257, 0.697) 222 Example 78 Comparative 2:1 4.55 64.7 (0.243, 0.694) 214 Example 79
TABLE-US-00027 TABLE 27 Driving Light emitting layer voltage Efficiency Color coordinate Lifetime compound (V) (cd/A) (x, y) (T.sub.90) Comparative 2-2 4.75 51.2 (0.254, 0.724) 121 Example 80 Comparative 2-3 4.83 50.9 (0.233, 0.703) 112 Example 81 Comparative Ref 1 4.83 52.4 (0.258, 0.727) 95 Example 82 Comparative Ref 2 4.93 51.5 (0.247, 0.737) 83 Example 83 Comparative Ref 3 4.81 55.9 (0.246, 0.727) 104 Example 84
The organic light emitting device of the present invention includes a light emitting layer which uses a host and a phosphorescent dopant, and the host is composed of a host compound (p-n type) in which two or more compounds are mixed, and as a result, the organic light emitting device of the present invention has better lifetime characteristics than an organic light emitting device including a host compound composed of a single compound in the related art.
In particular, the p-n type host of the present invention has an advantage in that the ratio of the host may be adjusted to increase the light emitting characteristics, and the advantage is a result which can be achieved by appropriately combining a P host having a good hole mobility and an n host having a good electron mobility.
Further, in the present invention, the light emitting host composed of plural species of compounds was deposited by pre-mixing mixtures, and then forming the host by one deposition supply source. In this case, since the deposition is not conducted several times, the uniformity and thin film characteristics of the thin film may be improved, the process procedures may be simplified, the costs may be reduced, and a device in which the efficiency and lifetime have been improved may be formed.
* * * * *
C00001

C00002

C00003

C00004

C00005

C00006

C00007

C00008

C00009

C00010

C00011

C00012

C00013
C00014

C00015

C00016

C00017

C00018

C00019

C00020
C00021

C00022

C00023

C00024

C00025

C00026

C00027

C00028

C00029

C00030

C00031

C00032

C00033

C00034

C00035

C00036

C00037

C00038

C00039

C00040

C00041

C00042

C00043

C00044
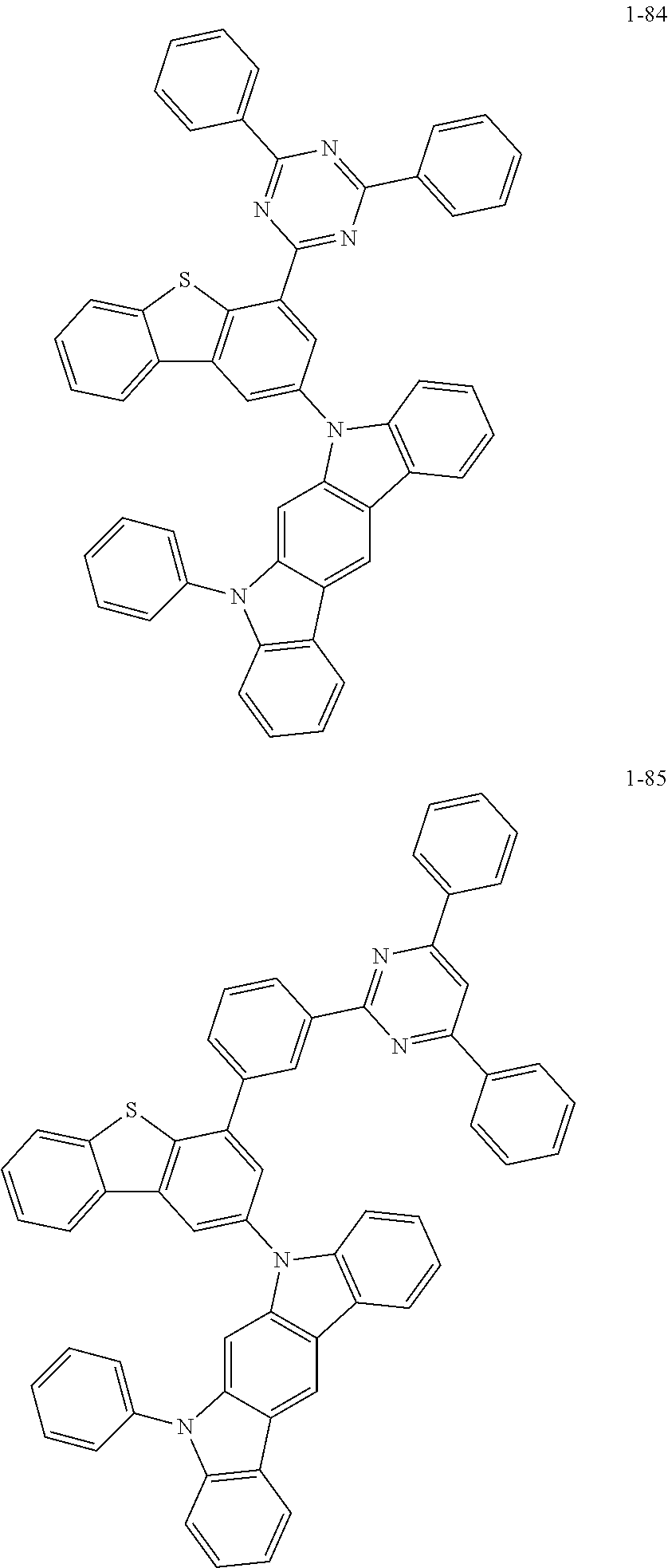
C00045

C00046
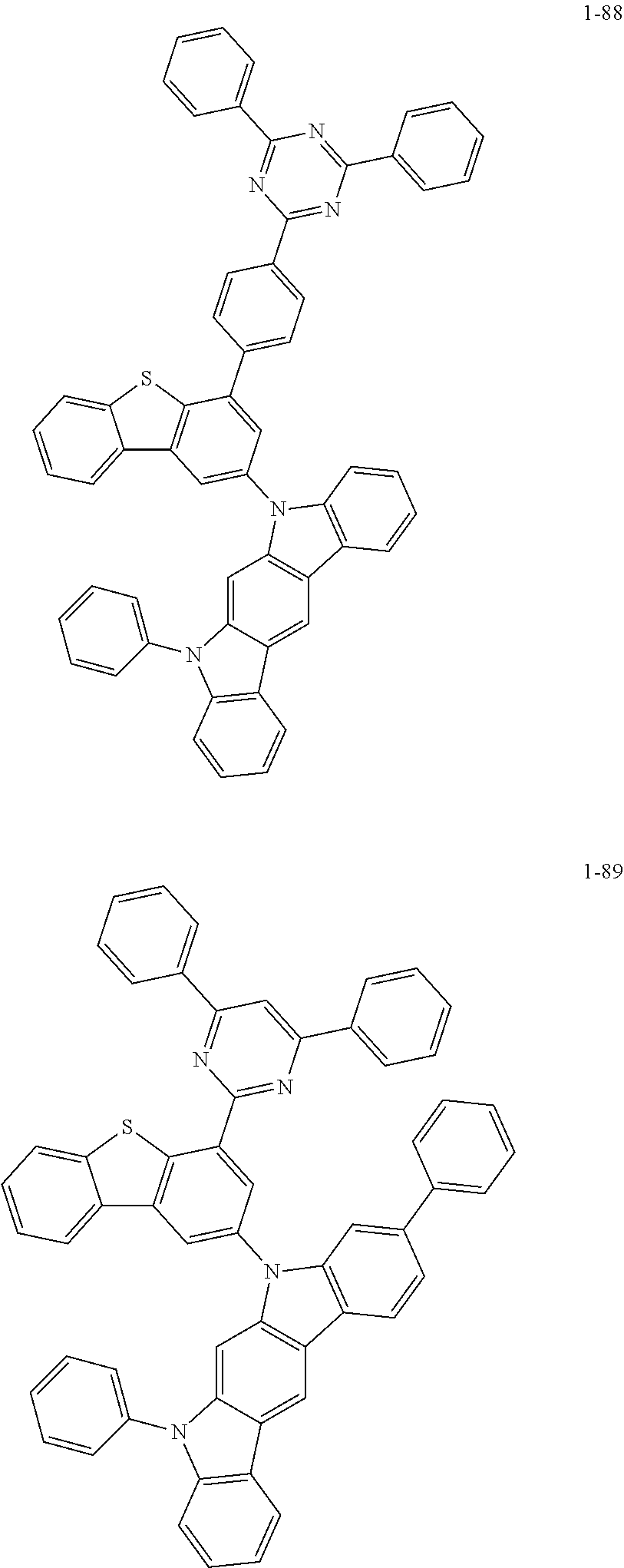
C00047

C00048

C00049

C00050

C00051

C00052

C00053

C00054

C00055
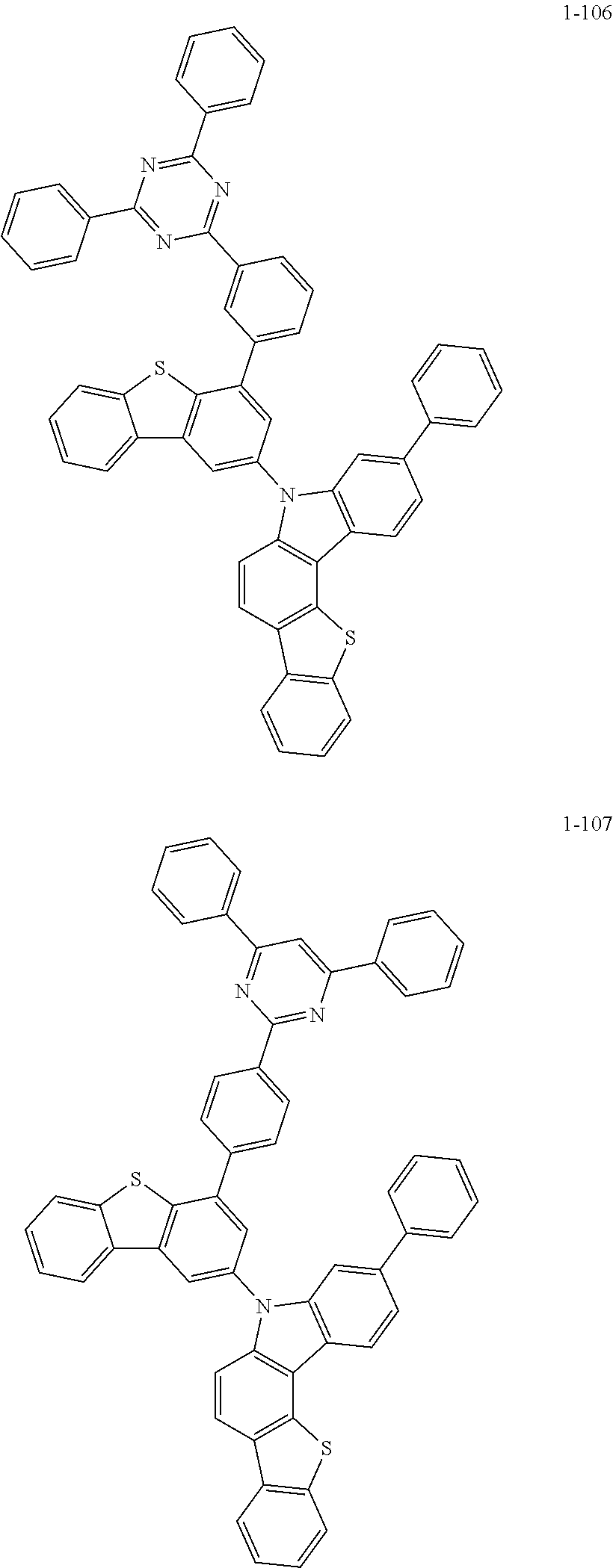
C00056

C00057

C00058

C00059

C00060

C00061
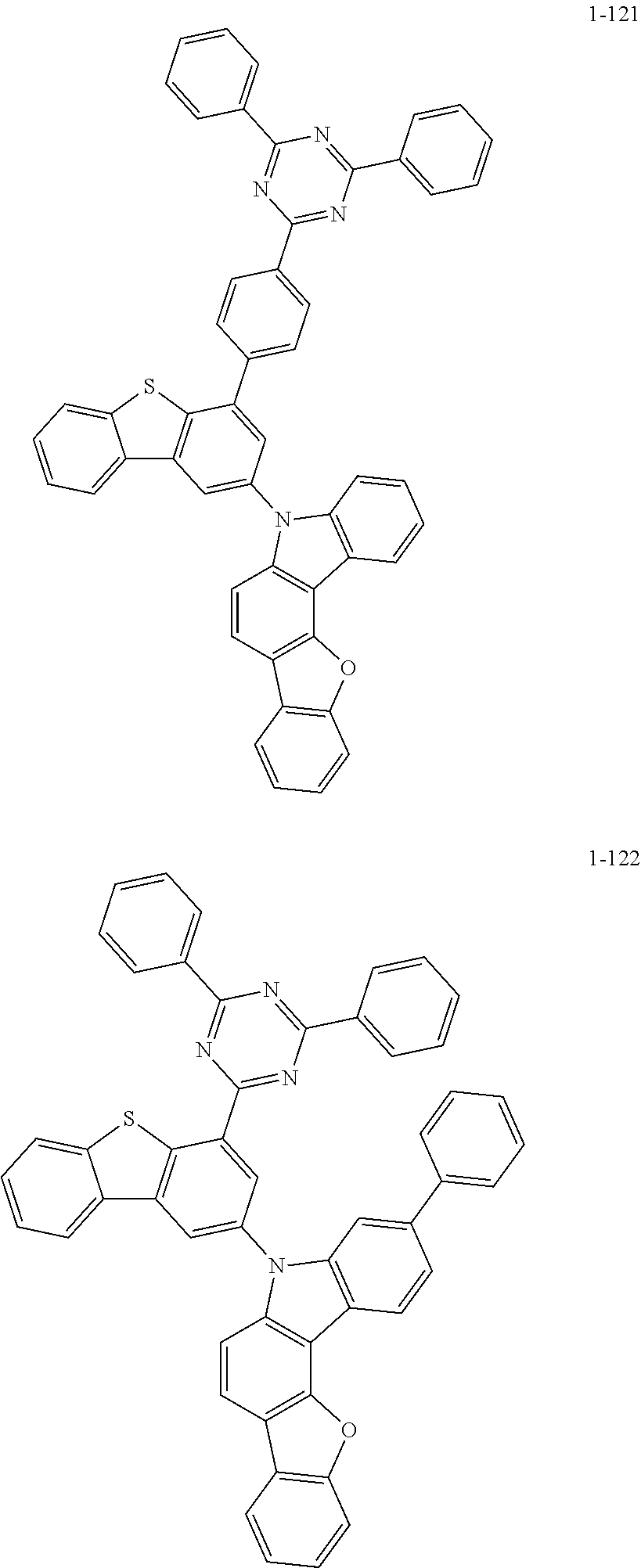
C00062

C00063

C00064

C00065

C00066

C00067

C00068

C00069

C00070

C00071

C00072

C00073
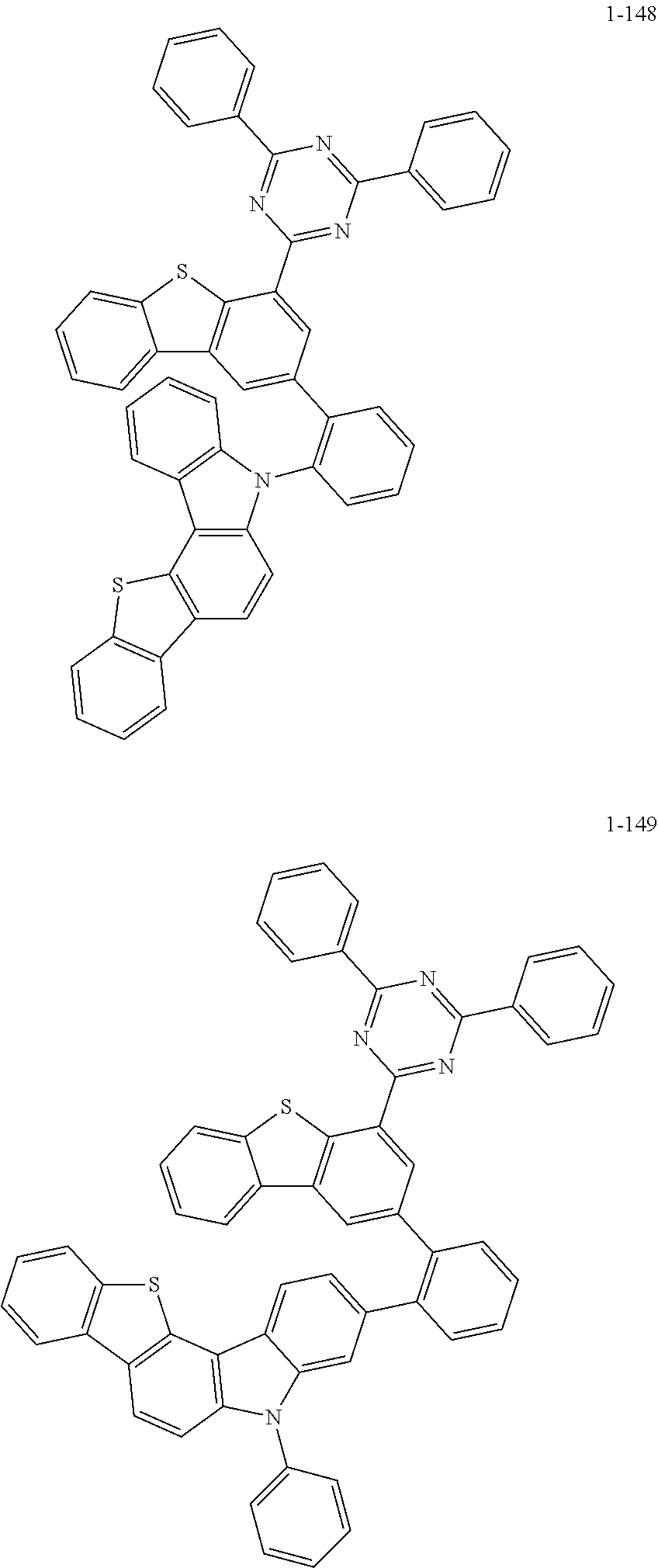
C00074

C00075

C00076
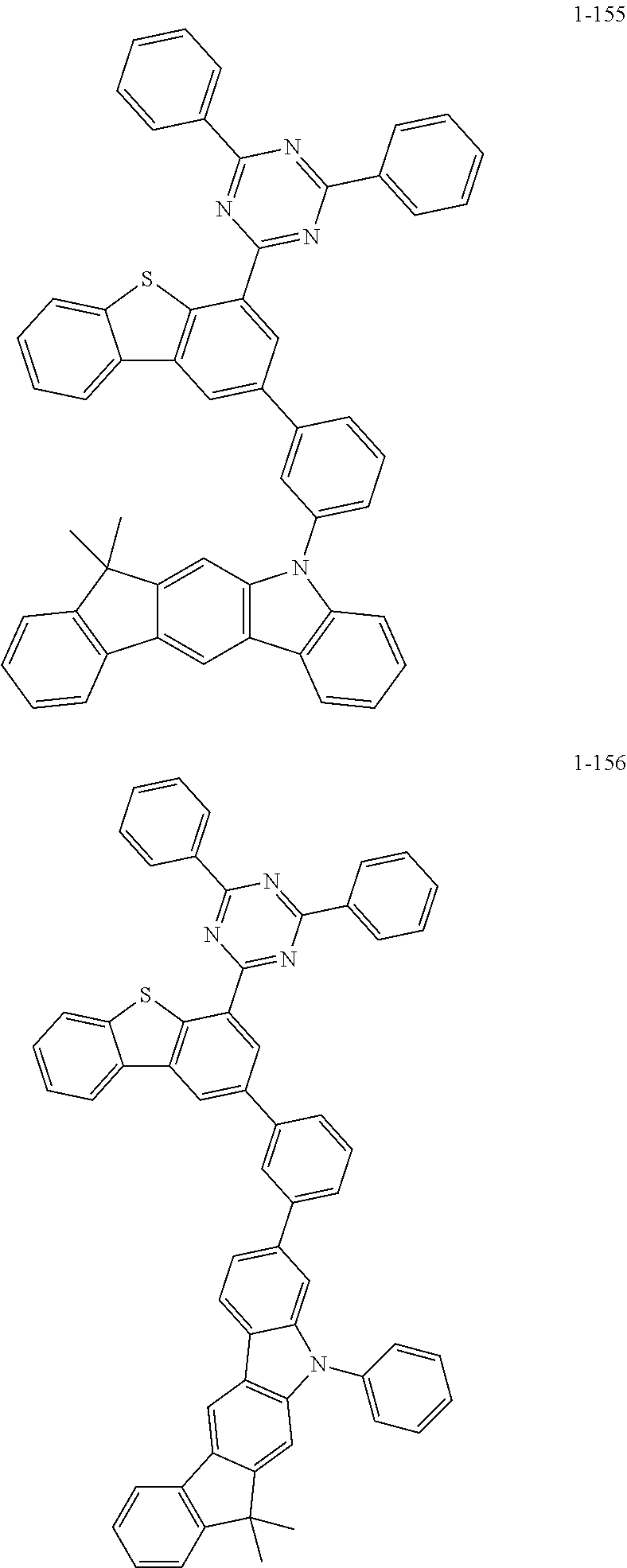
C00077

C00078

C00079

C00080

C00081

C00082

C00083
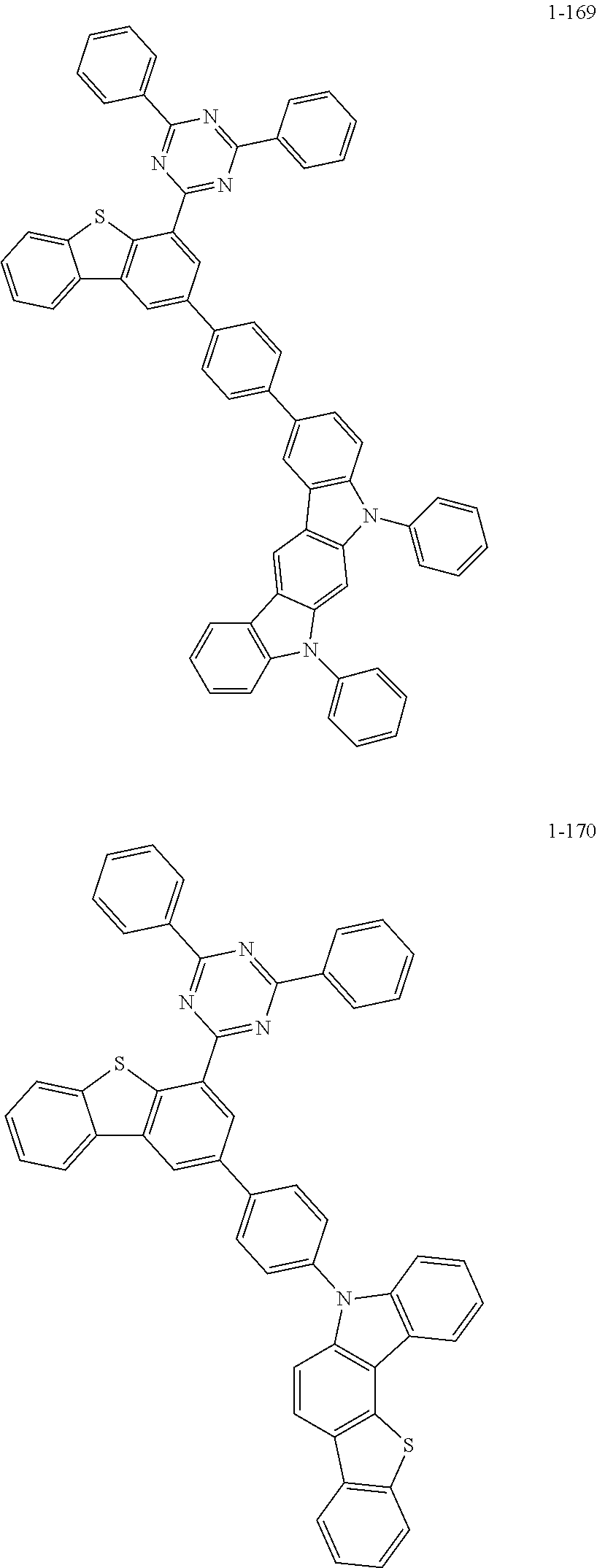
C00084

C00085
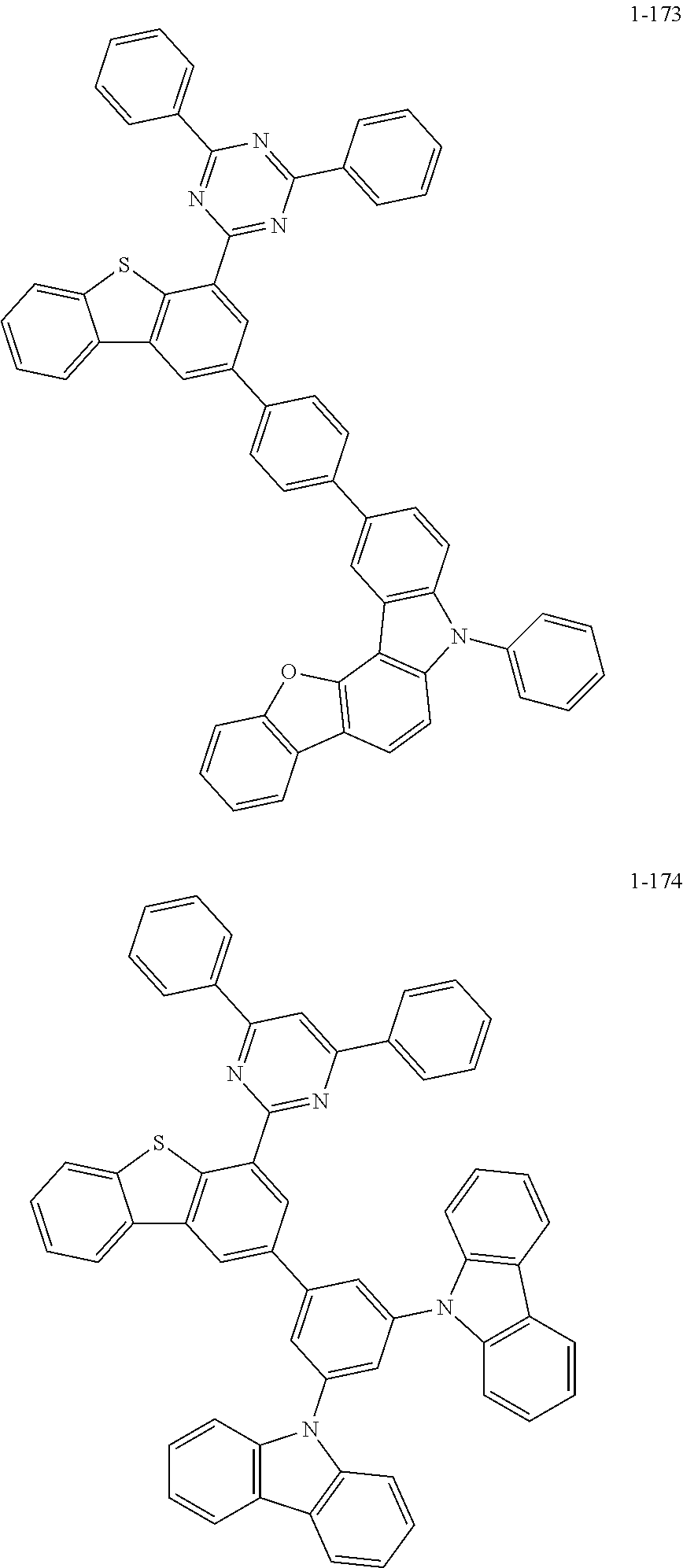
C00086

C00087

C00088
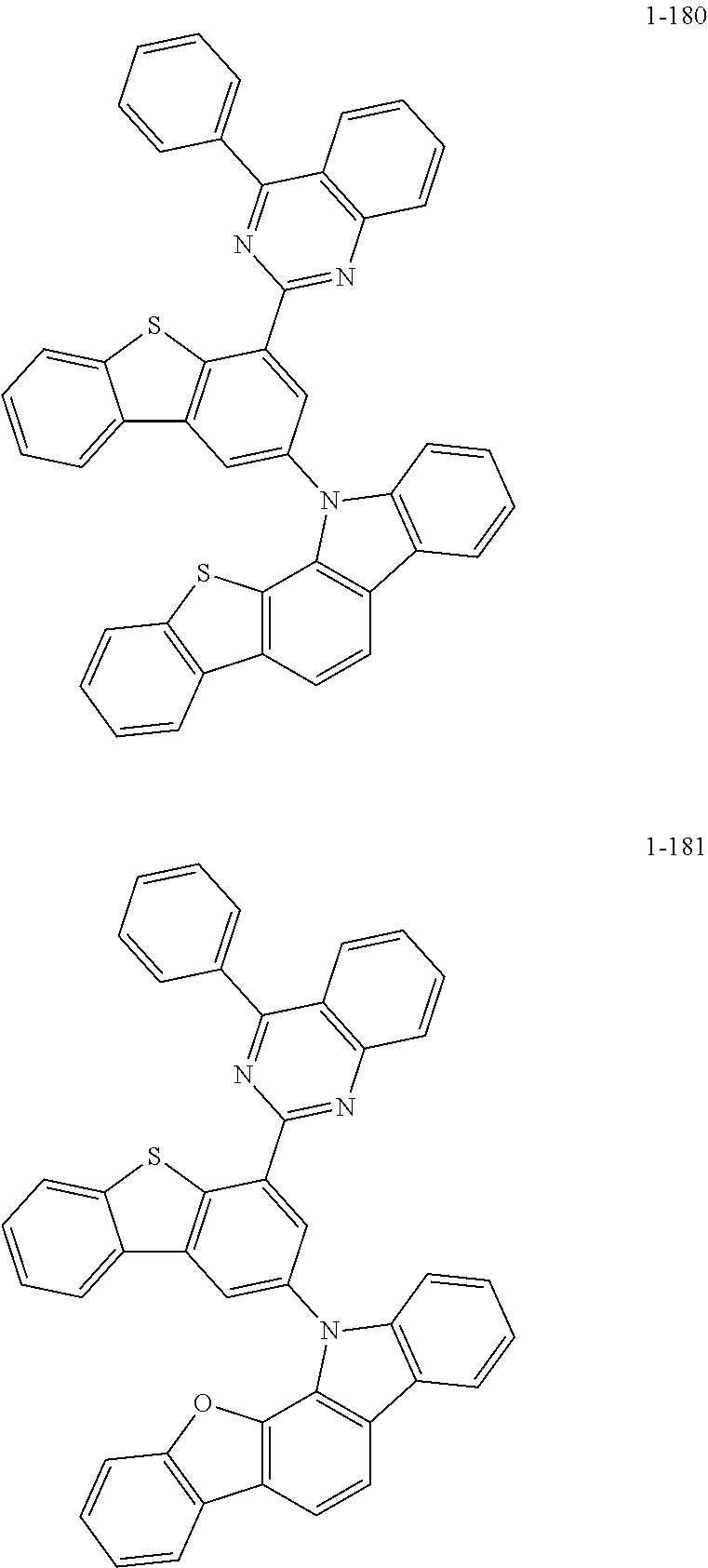
C00089

C00090

C00091

C00092

C00093

C00094

C00095

C00096

C00097

C00098

C00099

C00100

C00101

C00102

C00103

C00104

C00105

C00106

C00107

C00108

C00109

C00110

C00111

C00112

C00113

C00114

C00115

C00116

C00117

C00118

C00119

C00120
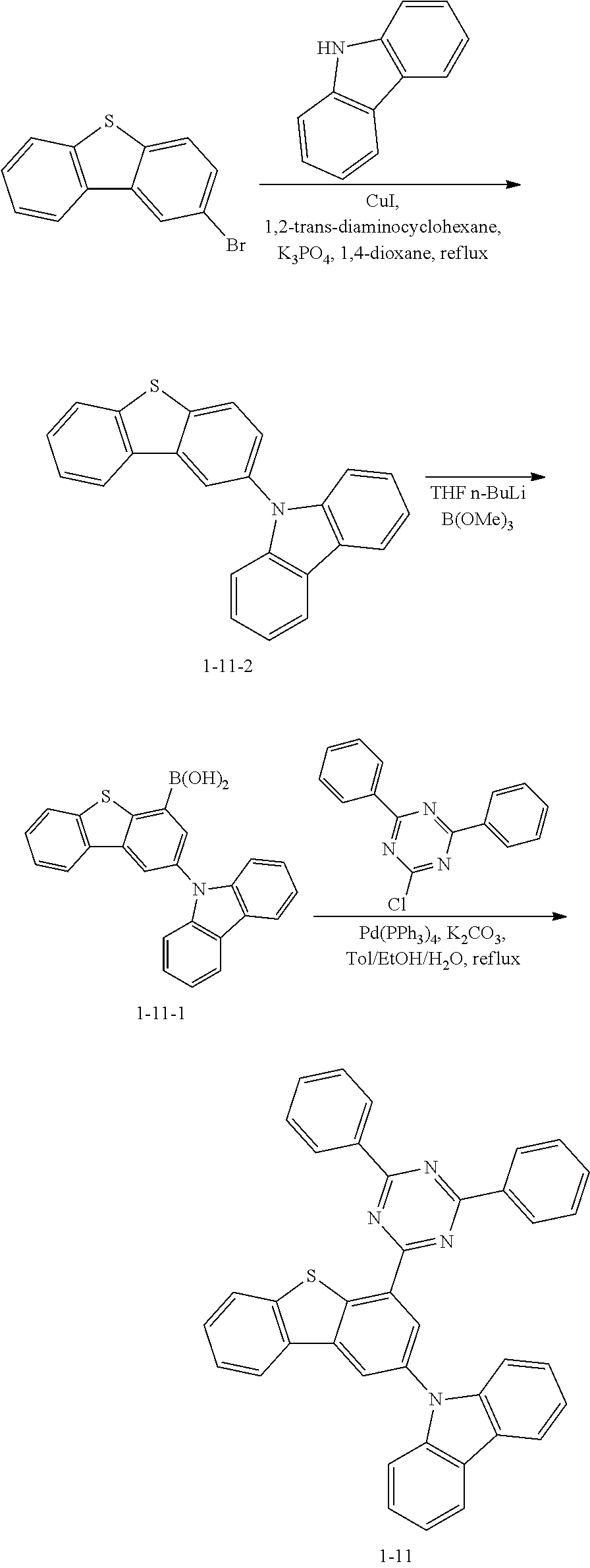
C00121

C00122

C00123
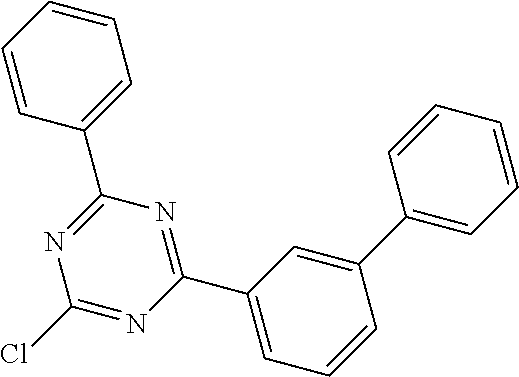
C00124

C00125

C00126

C00127

C00128
C00129

C00130

C00131

C00132

C00133

C00134

C00135

C00136

C00137

C00138

C00139

C00140

C00141

C00142

C00143

C00144

C00145
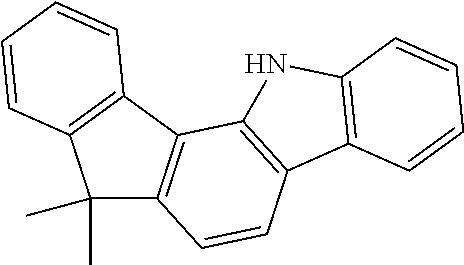
C00146

C00147
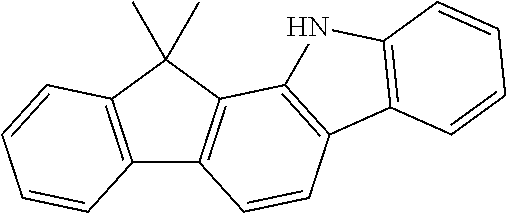
C00148

C00149

C00150

C00151

C00152

C00153

C00154

C00155

C00156

C00157

C00158

C00159

C00160
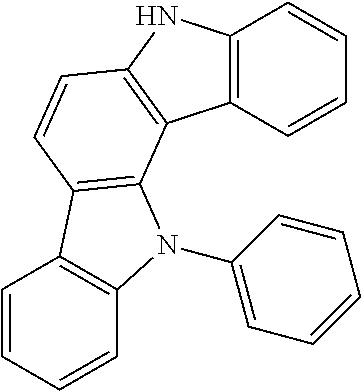
C00161
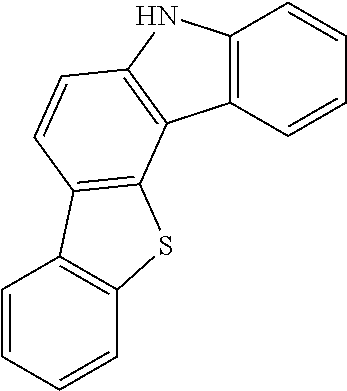
C00162

C00163

C00164

C00165

C00166

C00167

C00168

C00169

C00170

C00171

C00172

C00173

C00174

C00175

C00176

C00177

C00178

C00179

C00180

C00181

C00182

C00183

C00184

C00185

C00186

C00187

C00188

C00189

C00190

C00191

C00192

C00193

C00194

C00195

C00196

C00197

C00198

C00199

C00200
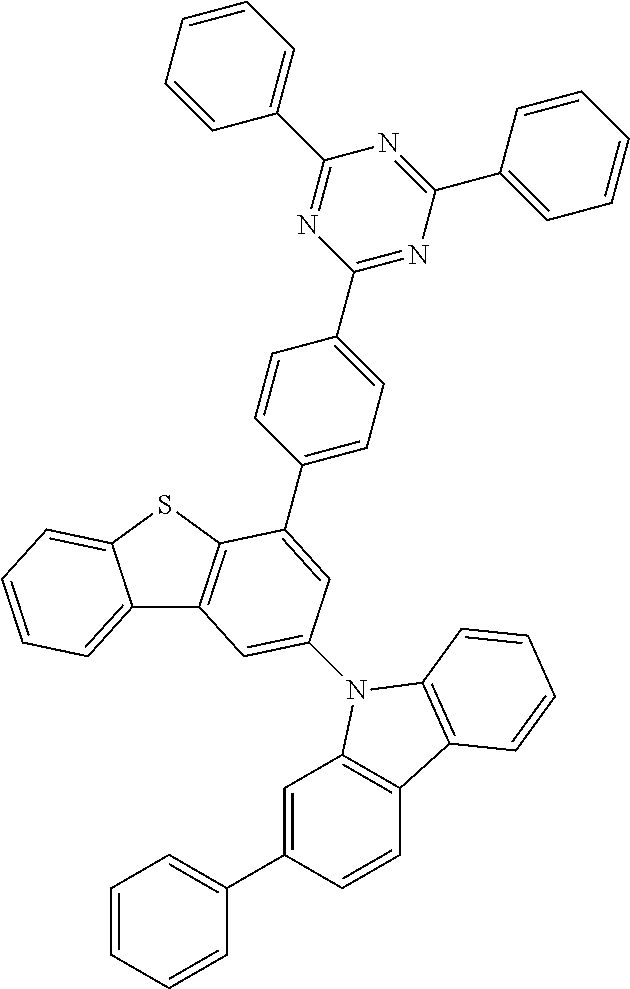
C00201

C00202

C00203

C00204

C00205

C00206

C00207

C00208

C00209

C00210

C00211
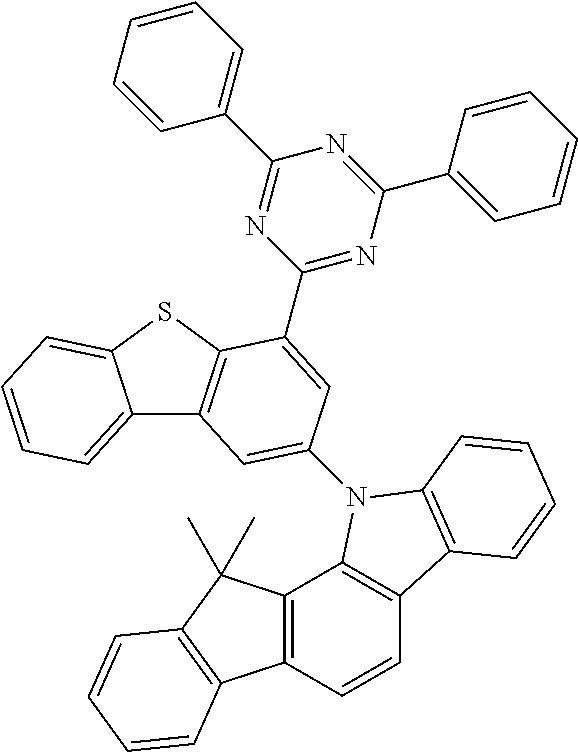
C00212

C00213
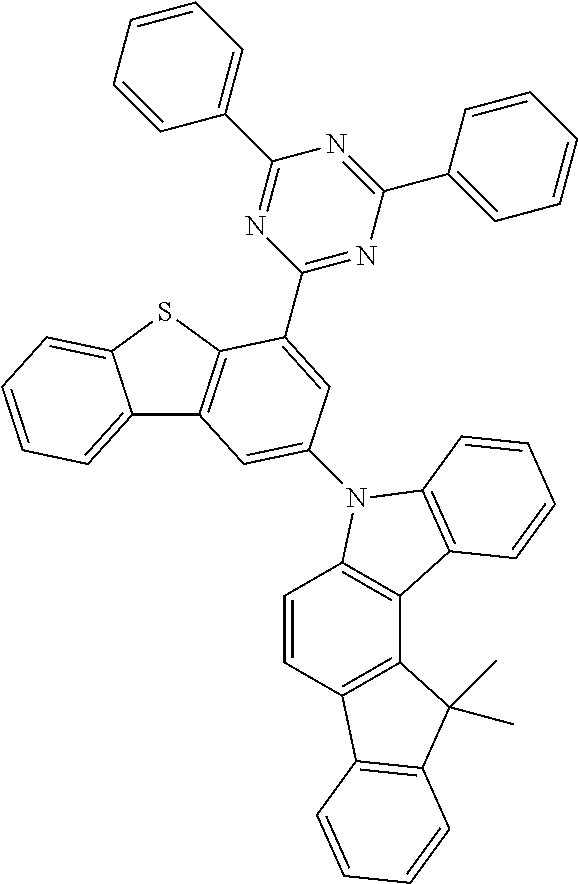
C00214

C00215

C00216

C00217

C00218

C00219

C00220

C00221

C00222

C00223

C00224

C00225
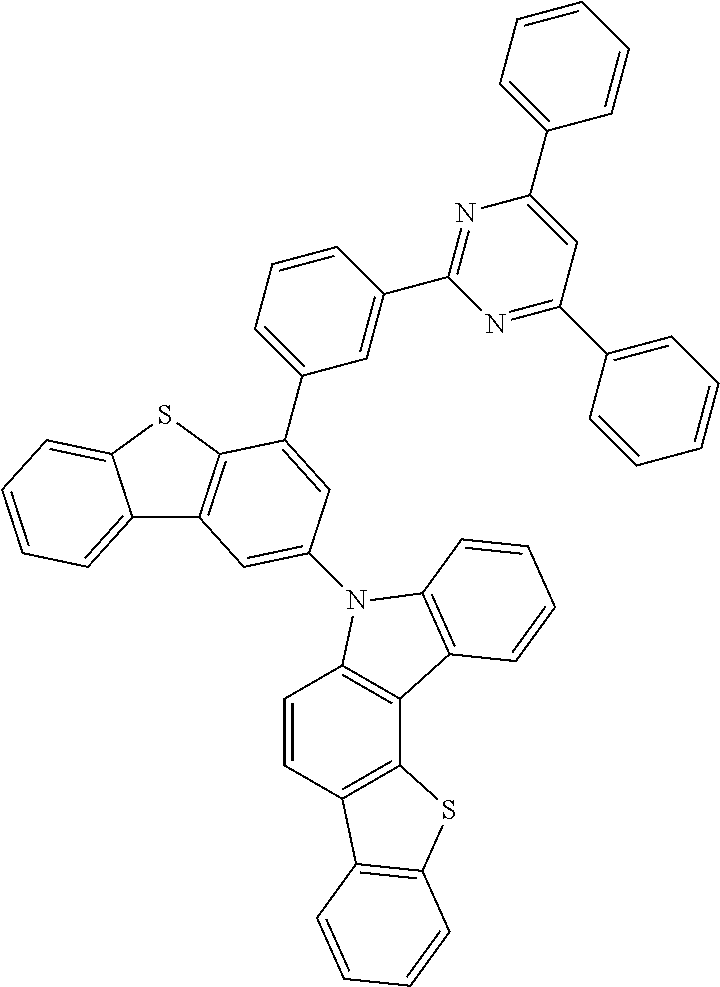
C00226

C00227
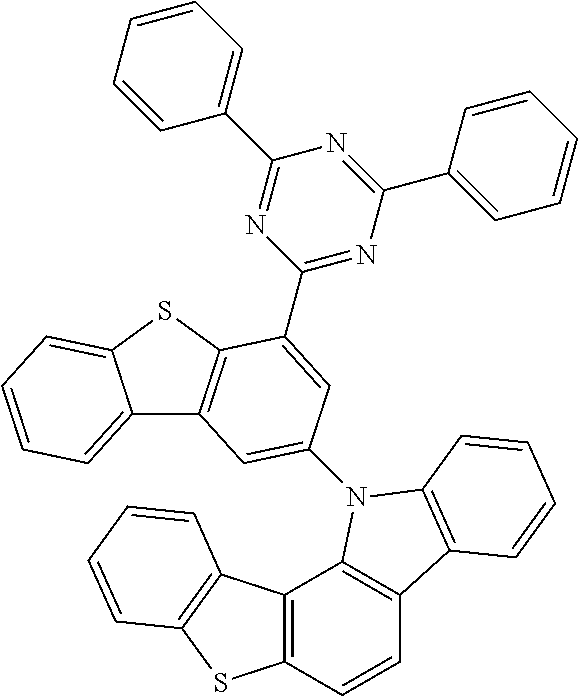
C00228

C00229

C00230

C00231
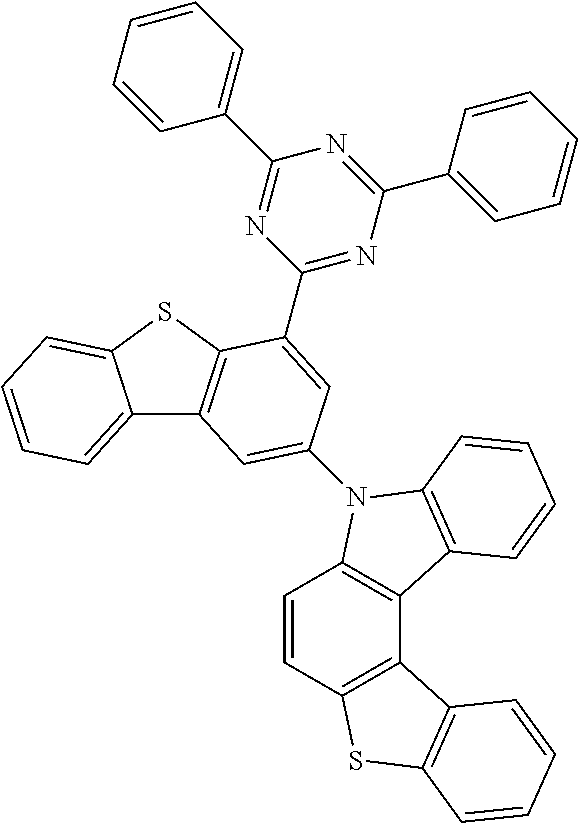
C00232

C00233

C00234

C00235

C00236

C00237
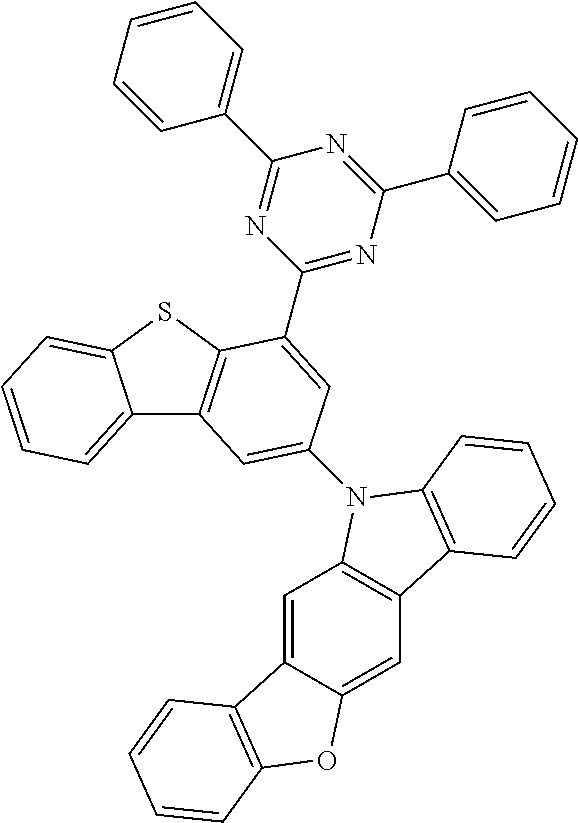
C00238

C00239

C00240
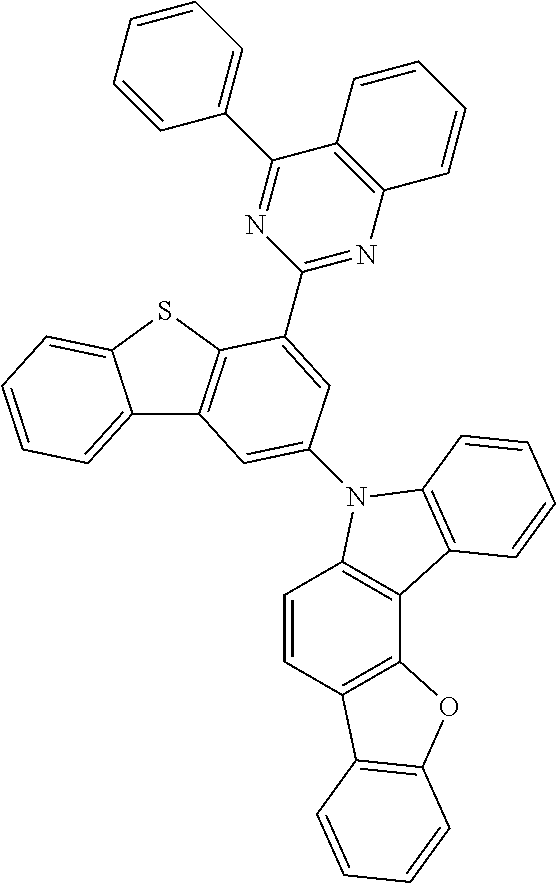
C00241

C00242
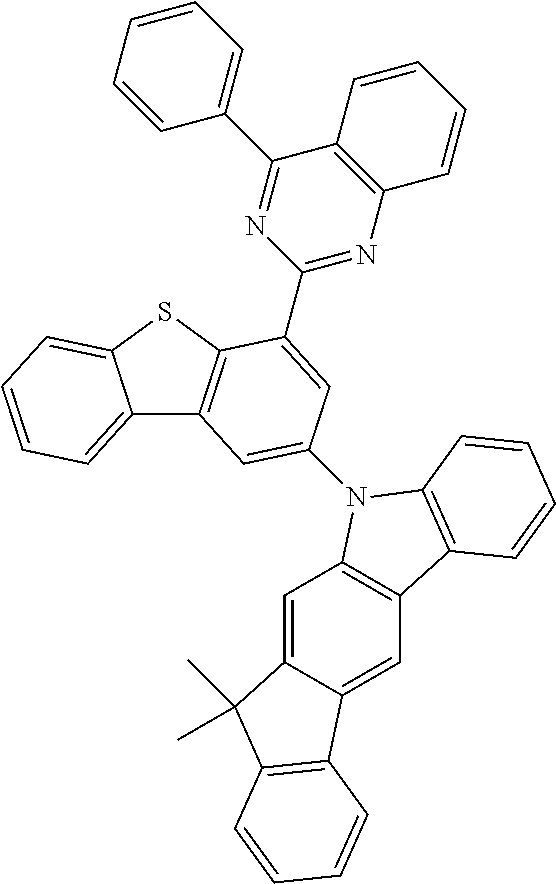
C00243

C00244

C00245

C00246

C00247

C00248

C00249

C00250

C00251

C00252

C00253

C00254

C00255
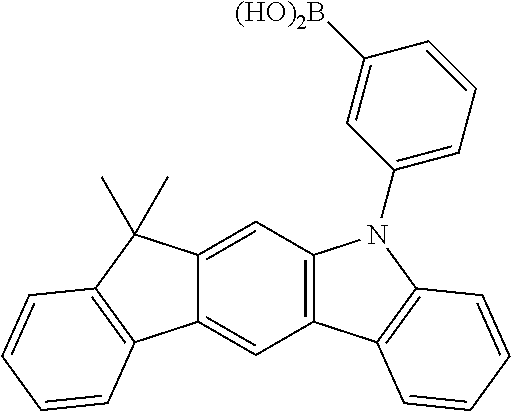
C00256

C00257

C00258

C00259

C00260

C00261

C00262

C00263

C00264

C00265

C00266

C00267

C00268

C00269

C00270
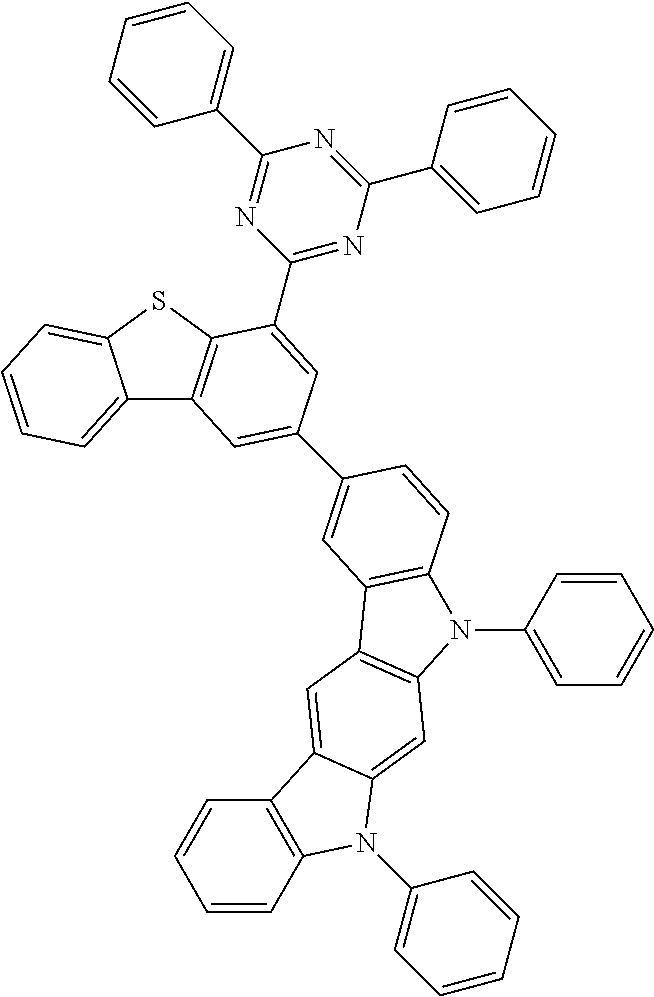
C00271

C00272

C00273
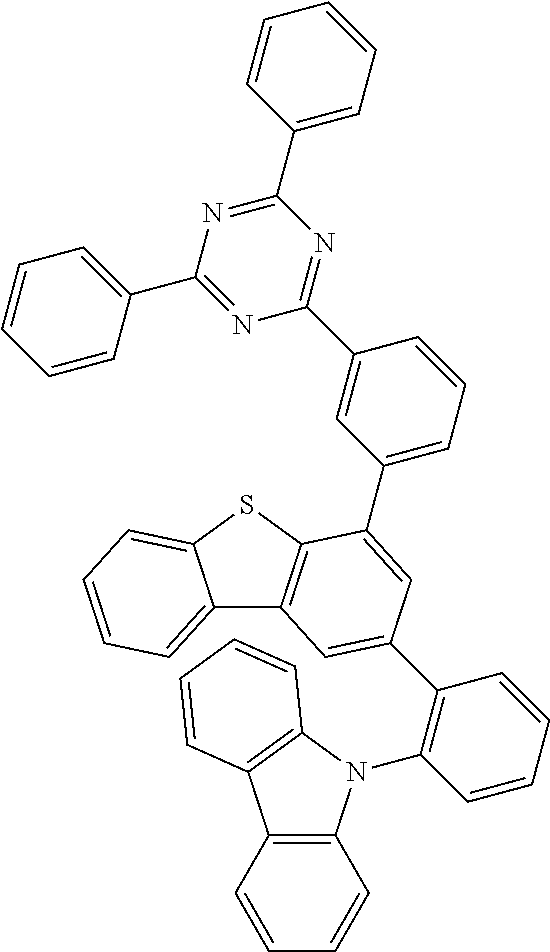
C00274

C00275

C00276
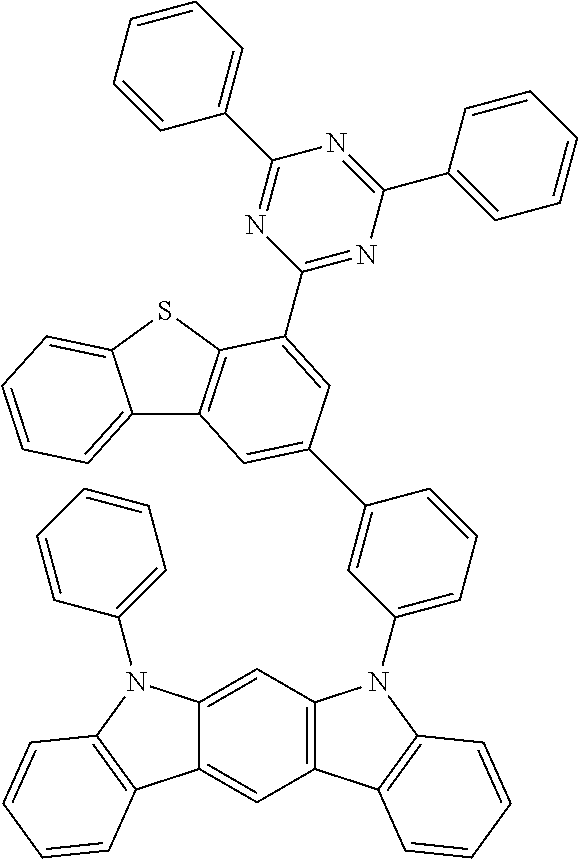
C00277

C00278

C00279

C00280

C00281
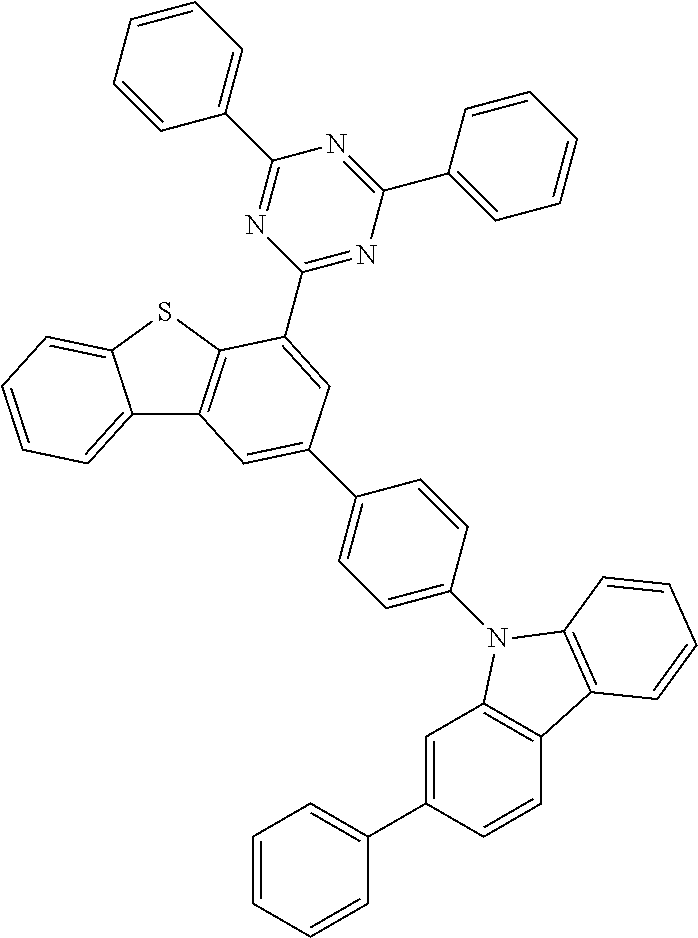
C00282

C00283

C00284

C00285

C00286

C00287

C00288

C00289

C00290

C00291

C00292

C00293

C00294

C00295

C00296

C00297

C00298

C00299

C00300

C00301

C00302

C00303
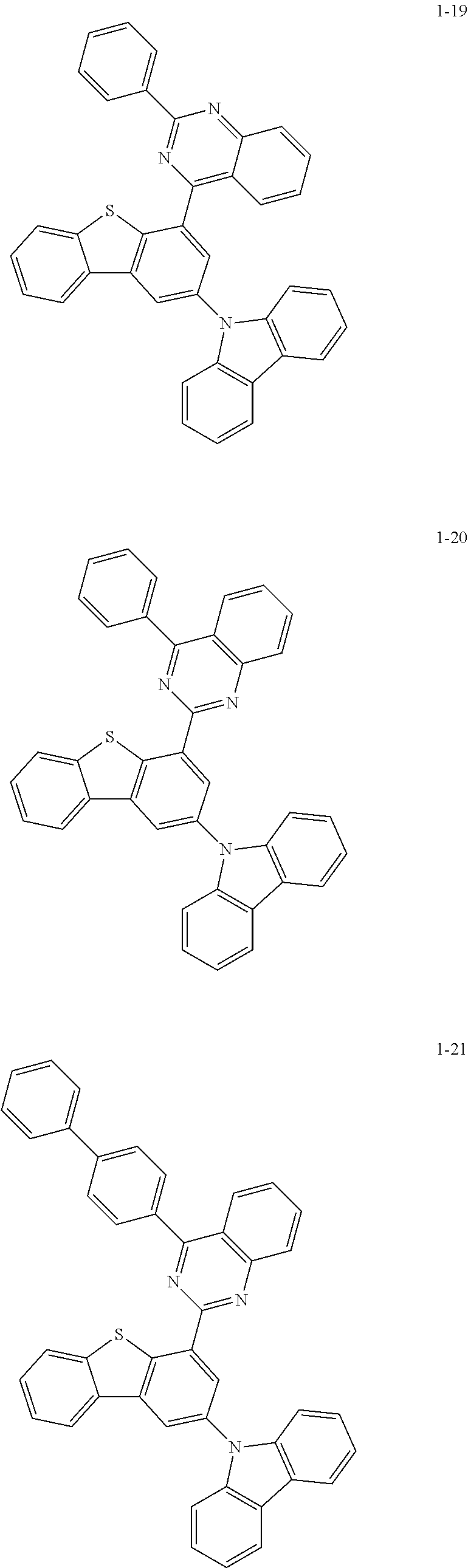
C00304

C00305

C00306

C00307

C00308

C00309

C00310

C00311

C00312

C00313

C00314

C00315
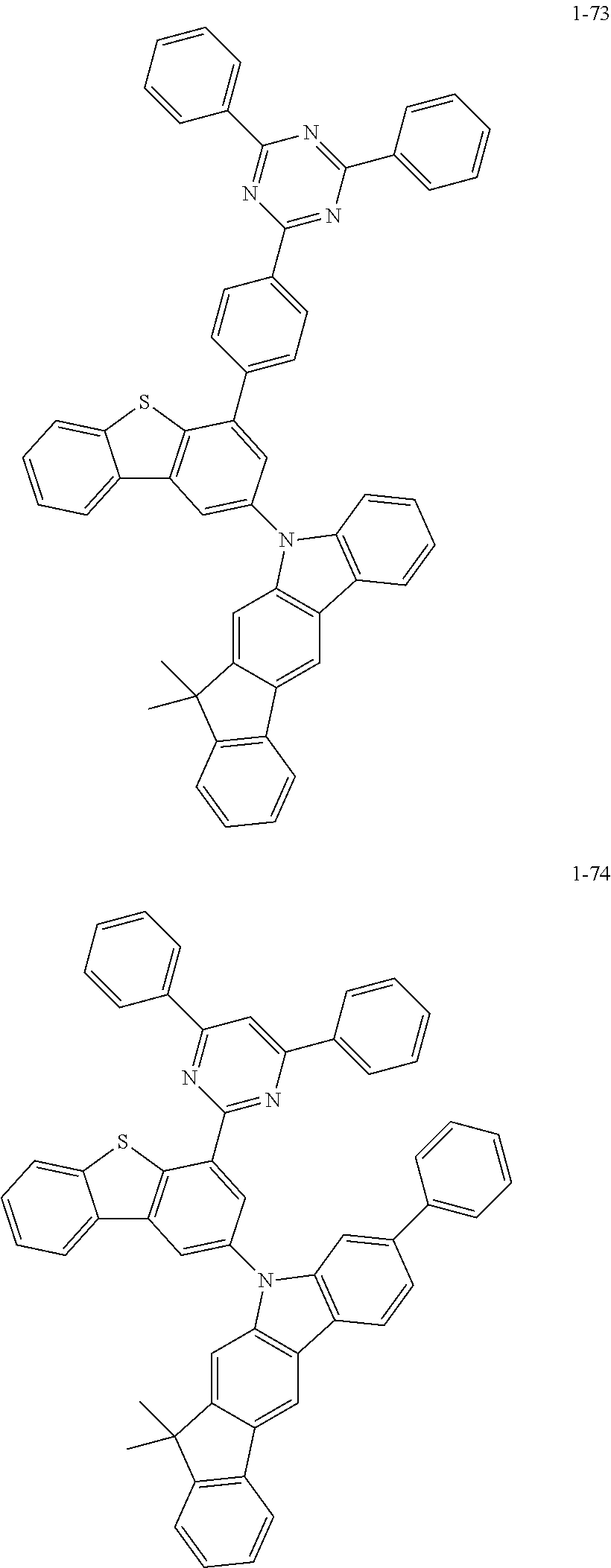
C00316

C00317

C00318

C00319

C00320

C00321

C00322

C00323

C00324

C00325

C00326

C00327

C00328

C00329
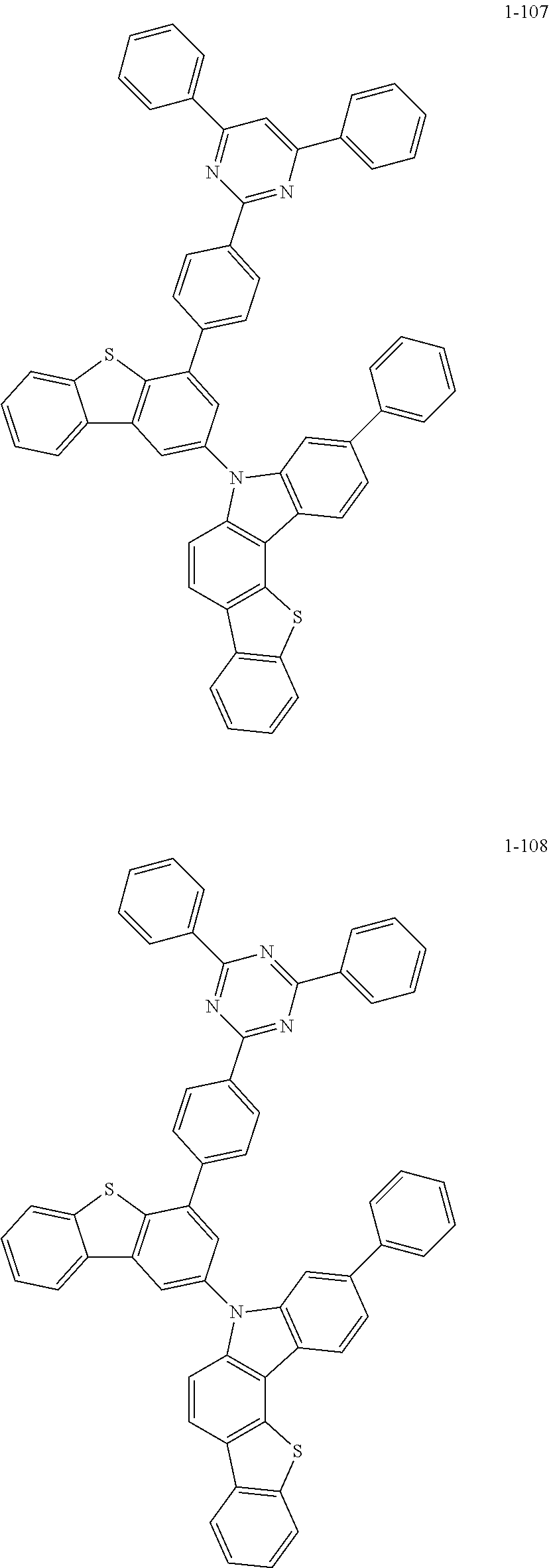
C00330

C00331

C00332

C00333

C00334

C00335

C00336

C00337

C00338

C00339

C00340

C00341

C00342
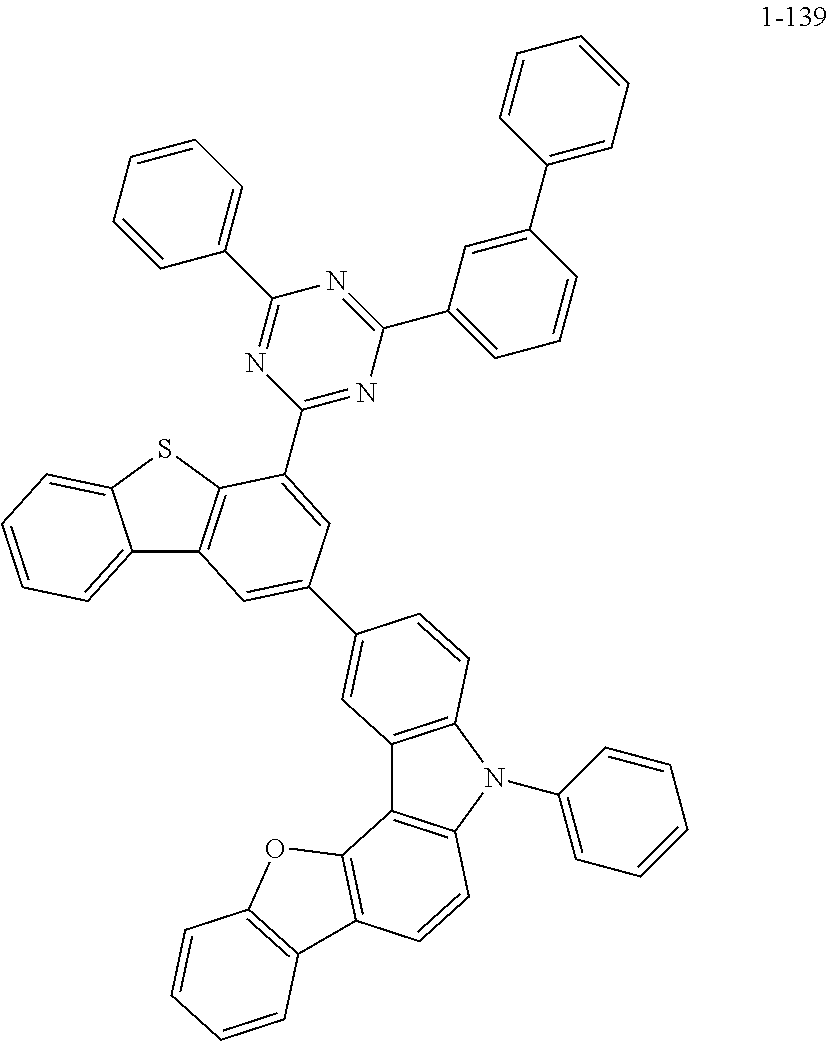
C00343

C00344

C00345
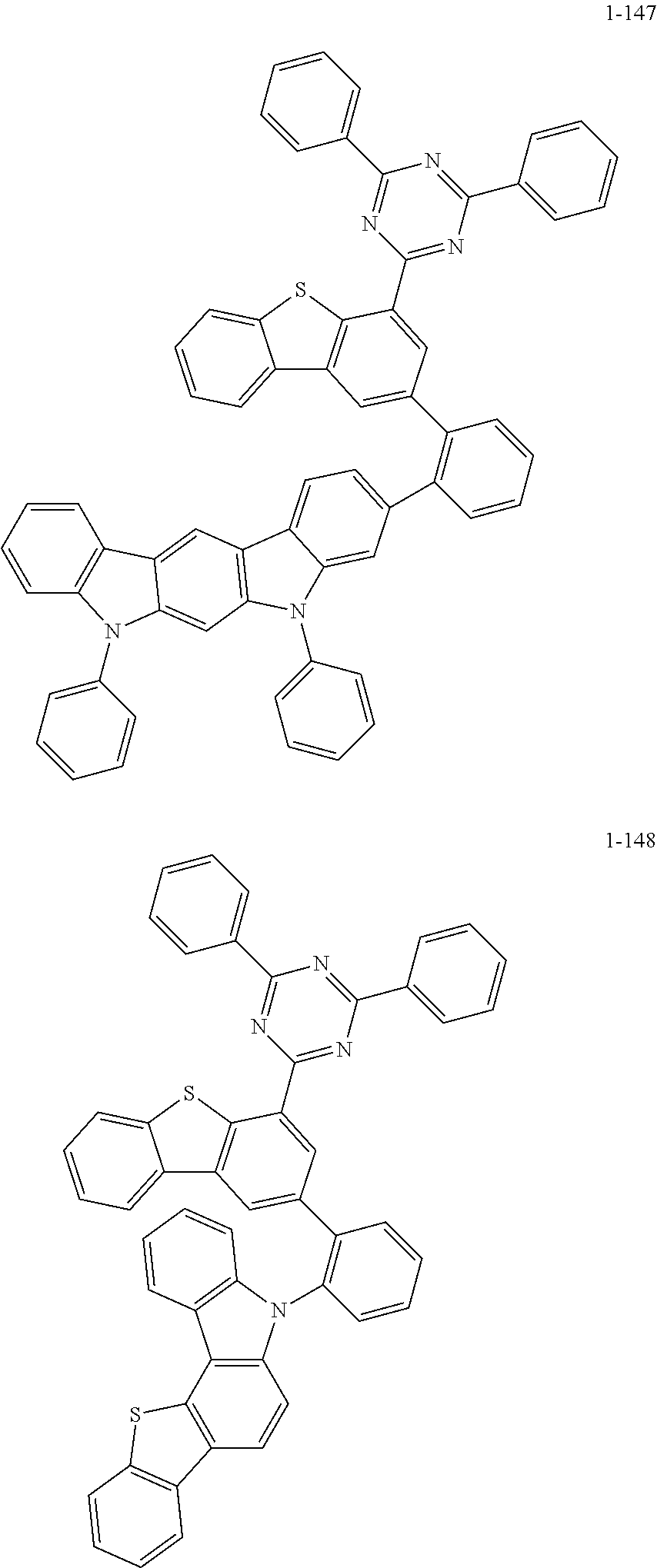
C00346

C00347
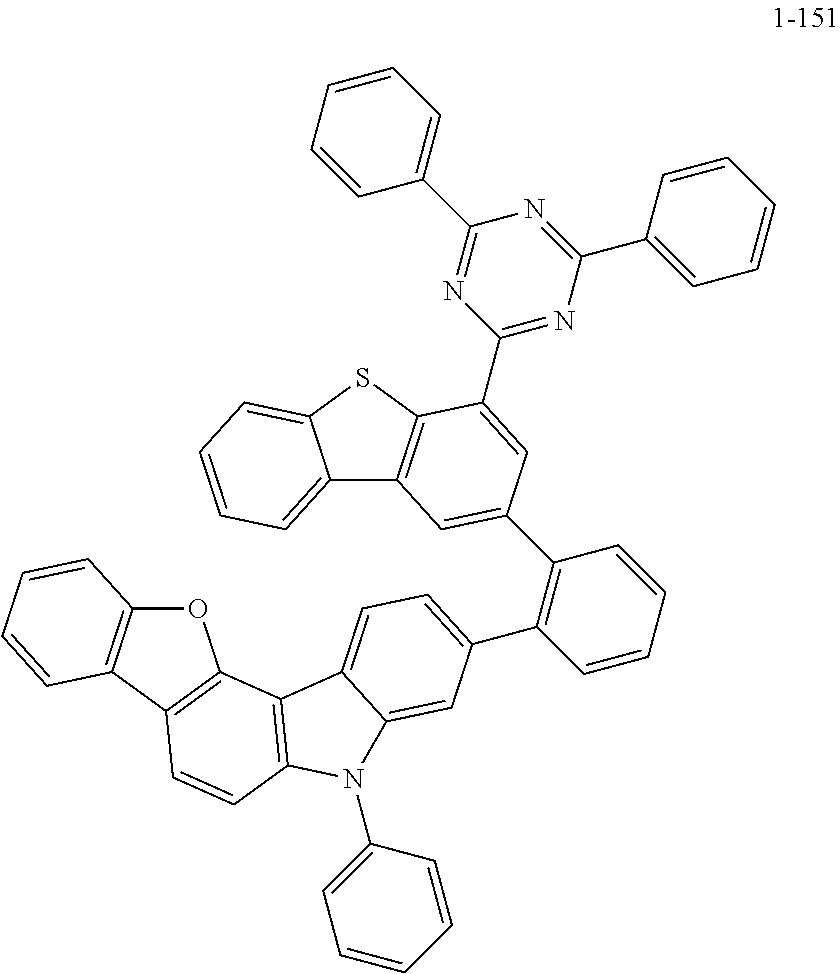
C00348

C00349
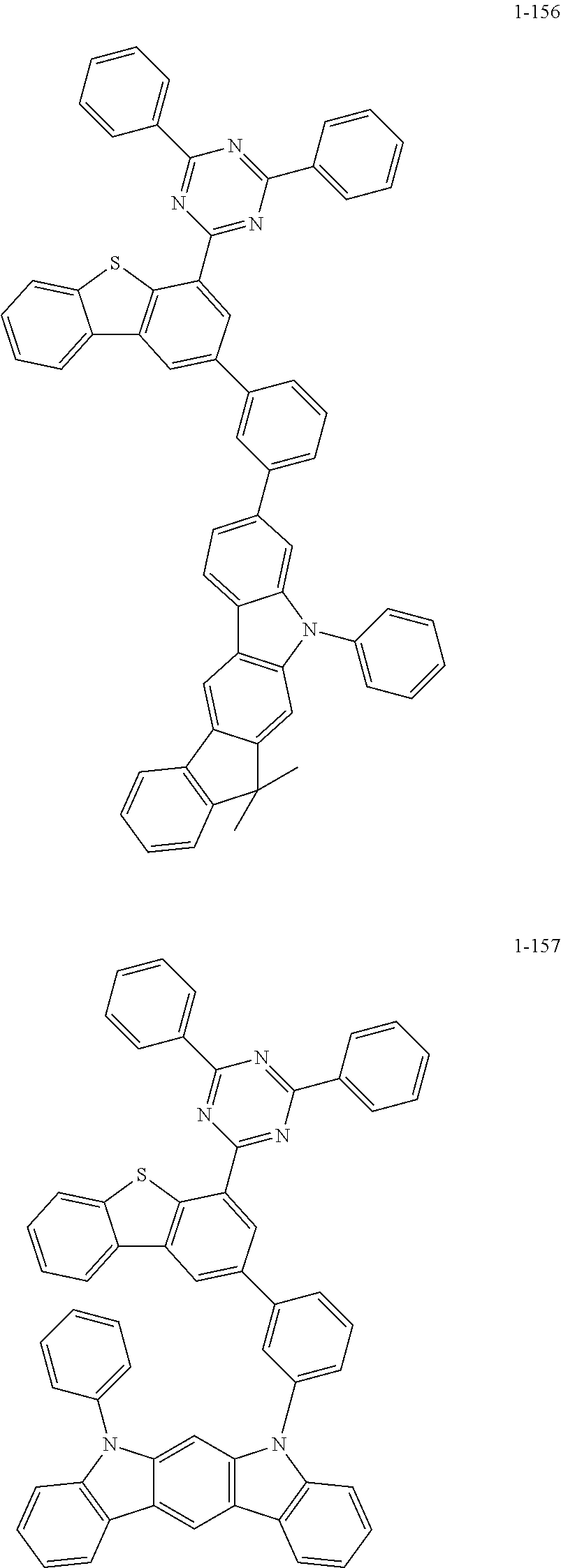
C00350
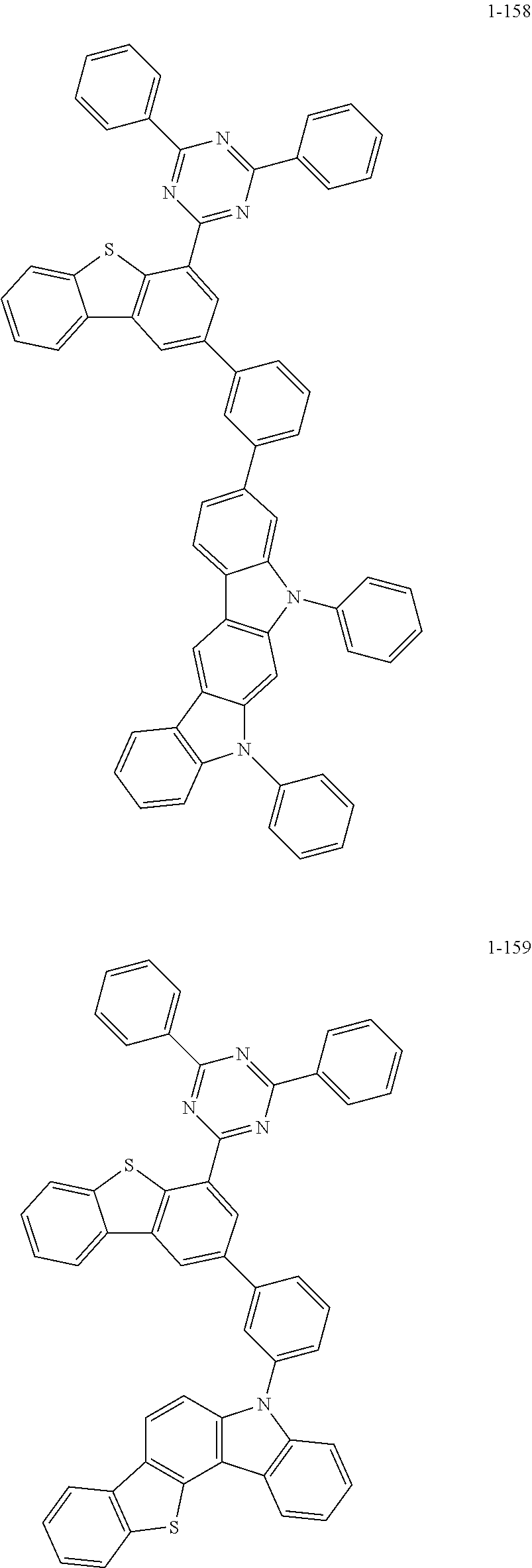
C00351

C00352

C00353

C00354

C00355

C00356

C00357
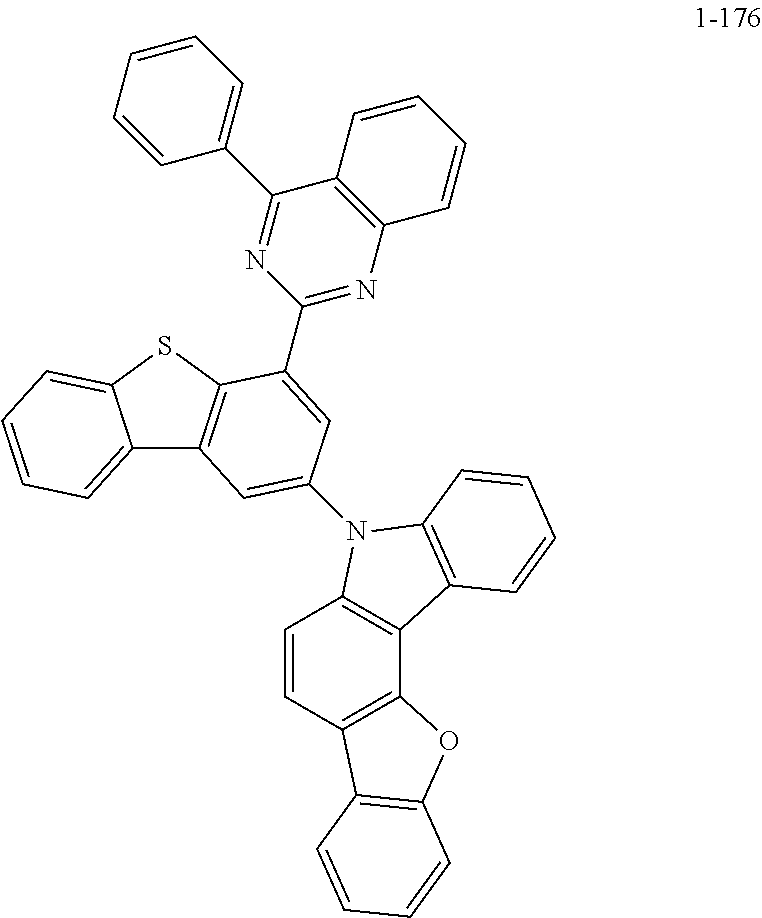
C00358
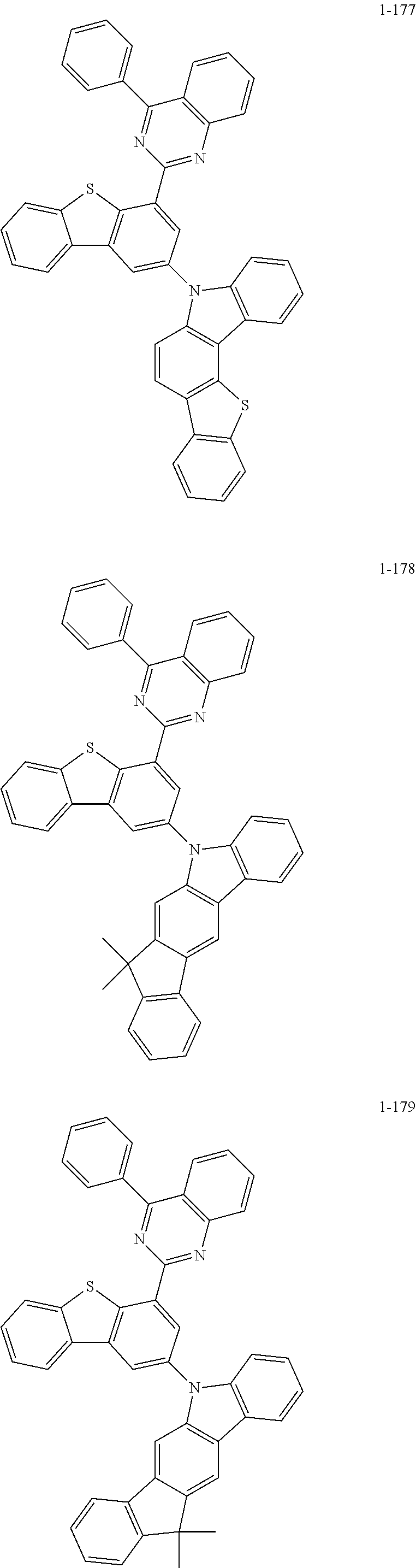
C00359

C00360

C00361

C00362

C00363
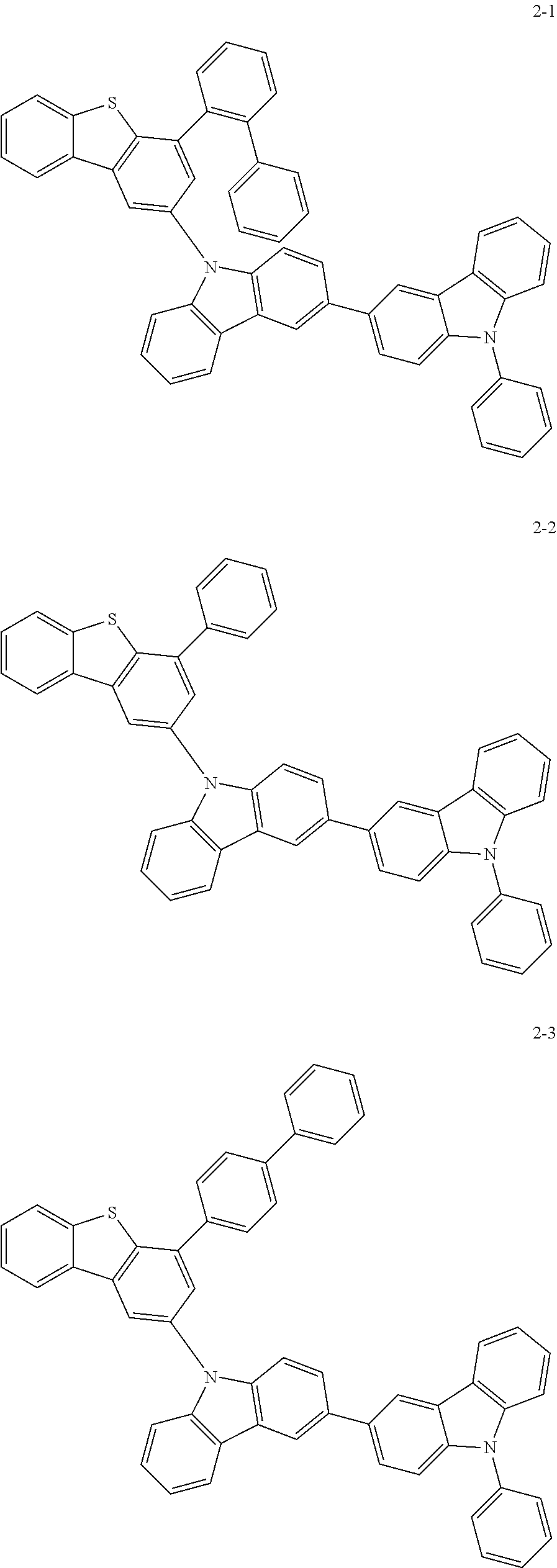
C00364

C00365

C00366

C00367

C00368
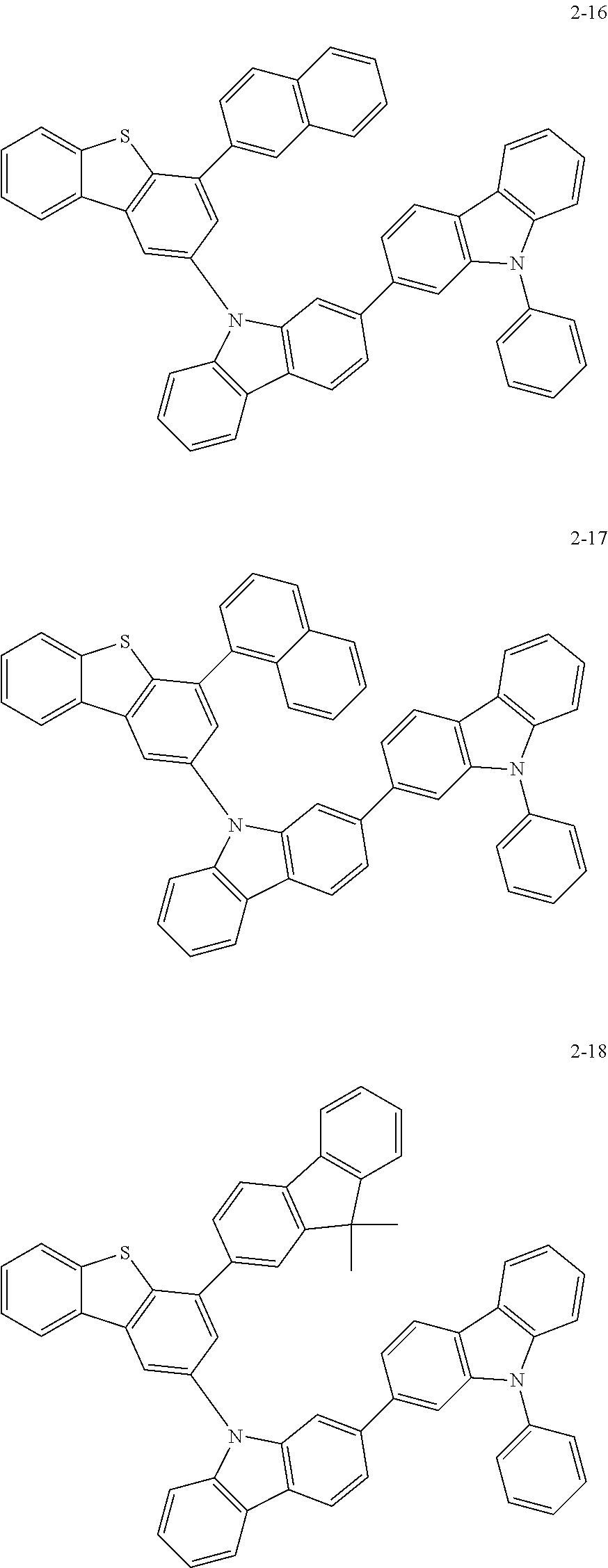
C00369

C00370

C00371
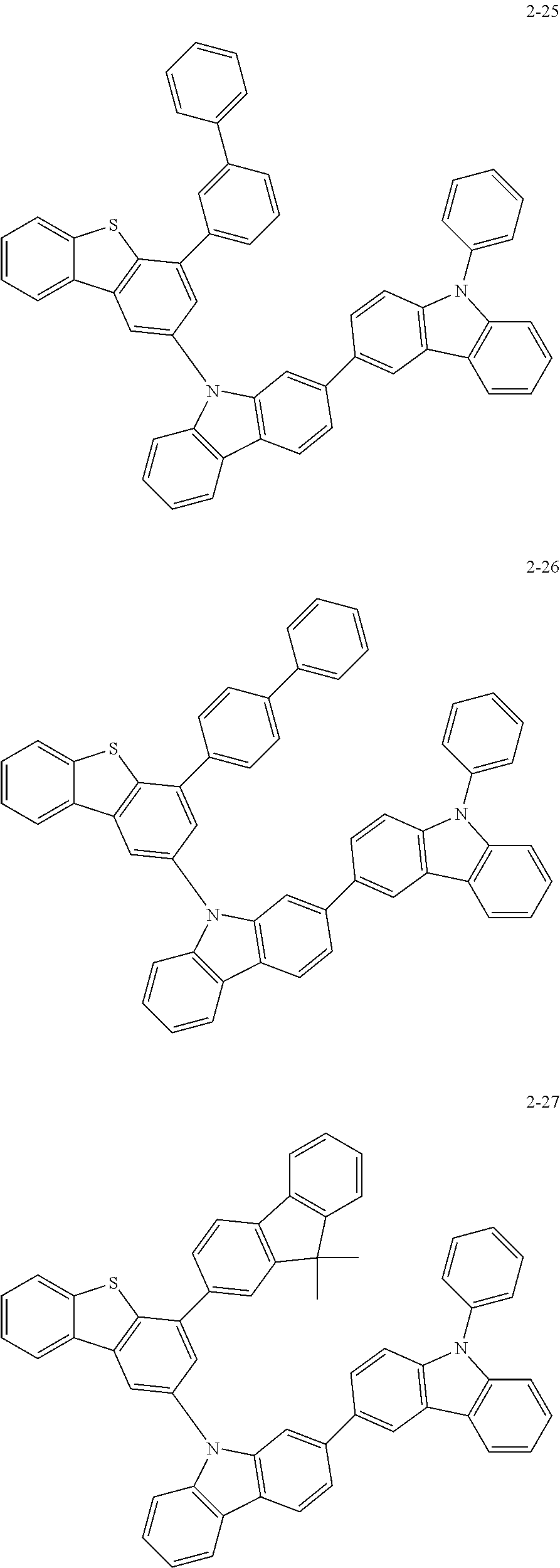
C00372
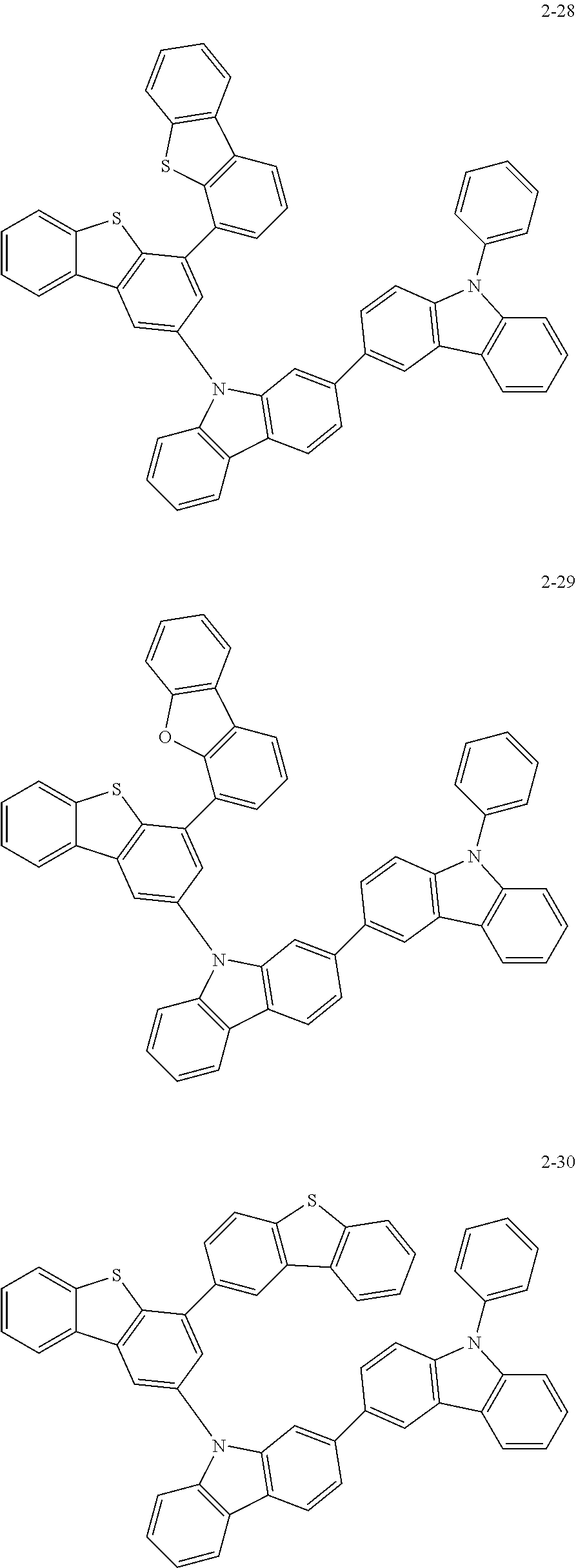
C00373

C00374

C00375

C00376
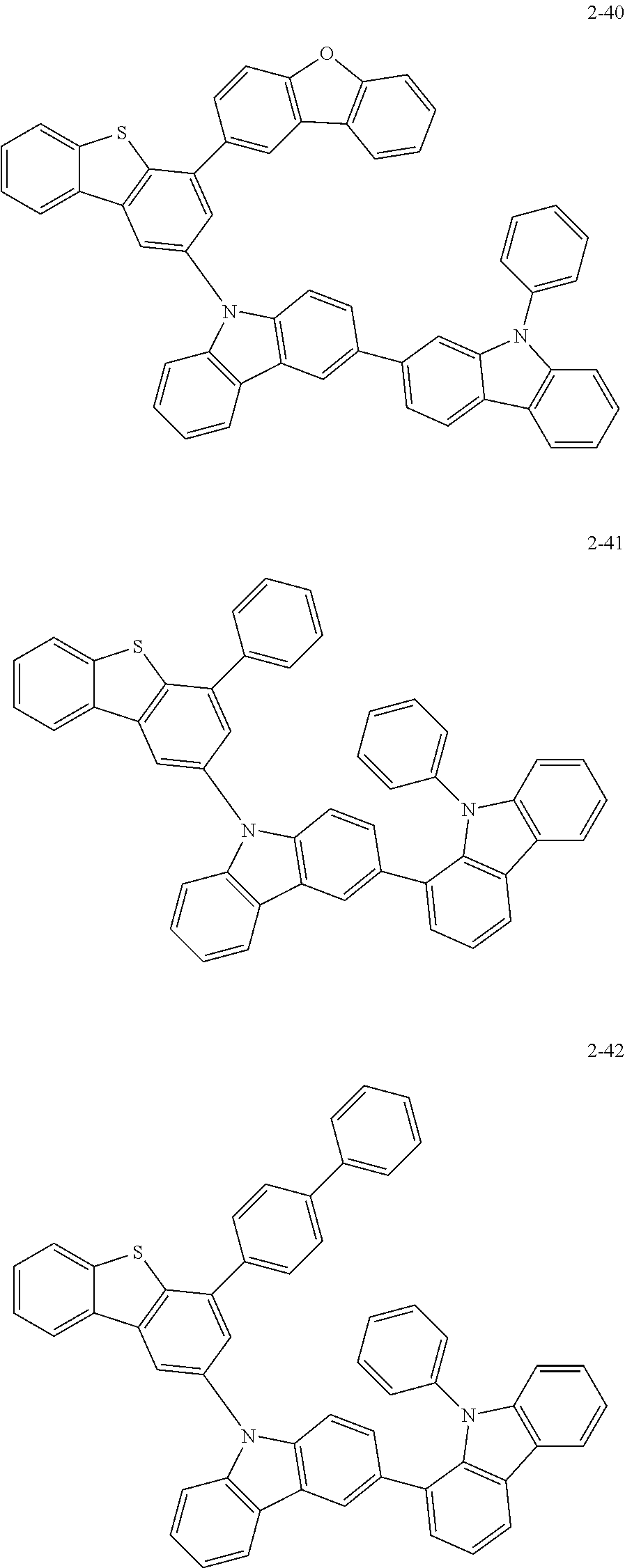
C00377

C00378

C00379

C00380

C00381

C00382

C00383

C00384

C00385

C00386

C00387

C00388

C00389

C00390

C00391

C00392

C00393

C00394

C00395

C00396
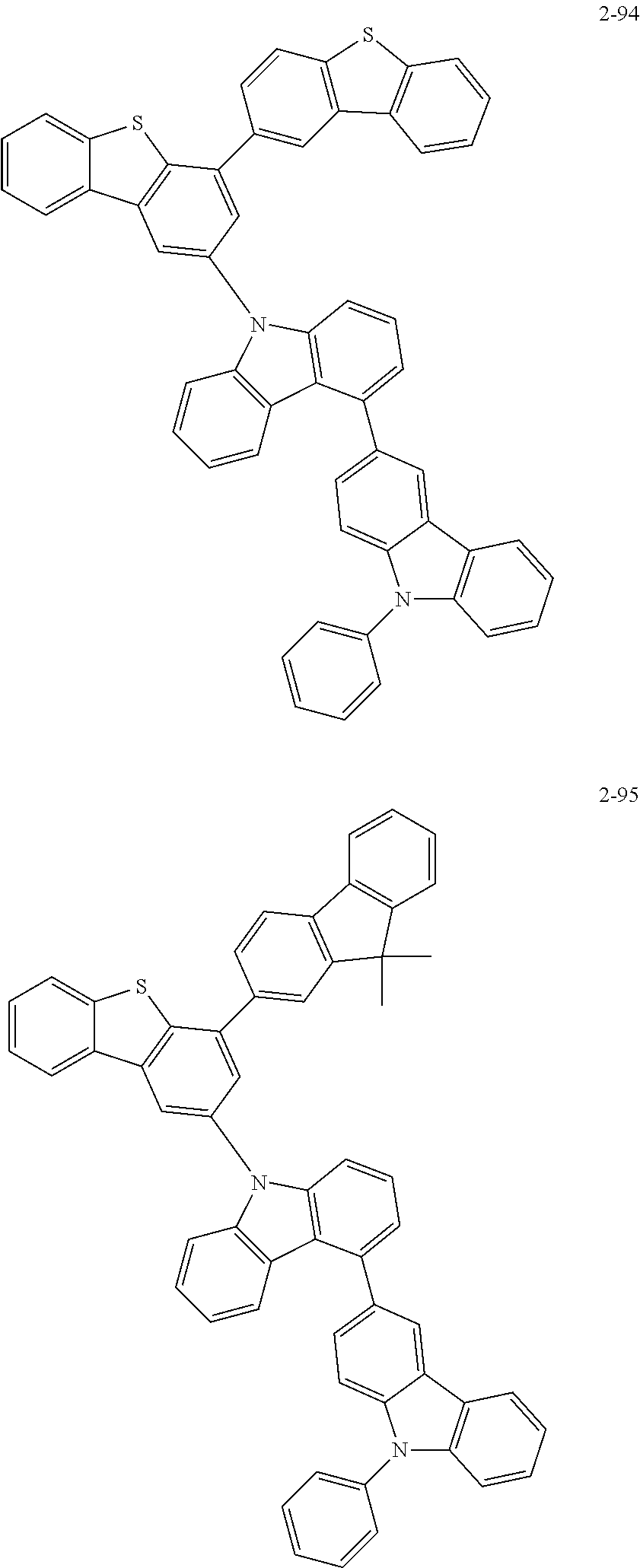
C00397

C00398

C00399

C00400

C00401

C00402

D00000

D00001

D00002

D00003

D00004
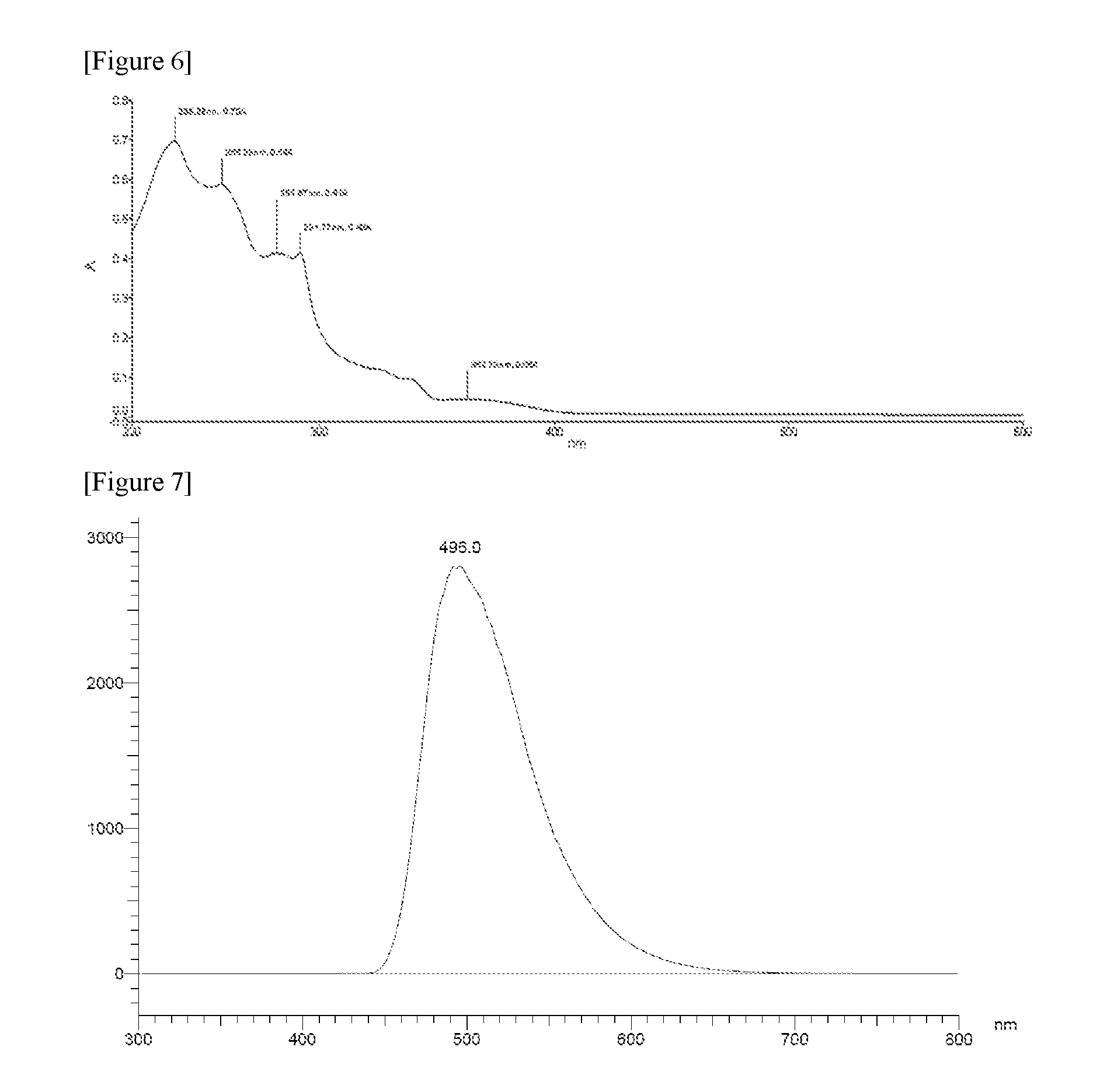
D00005

D00006

D00007

D00008

D00009

D00010
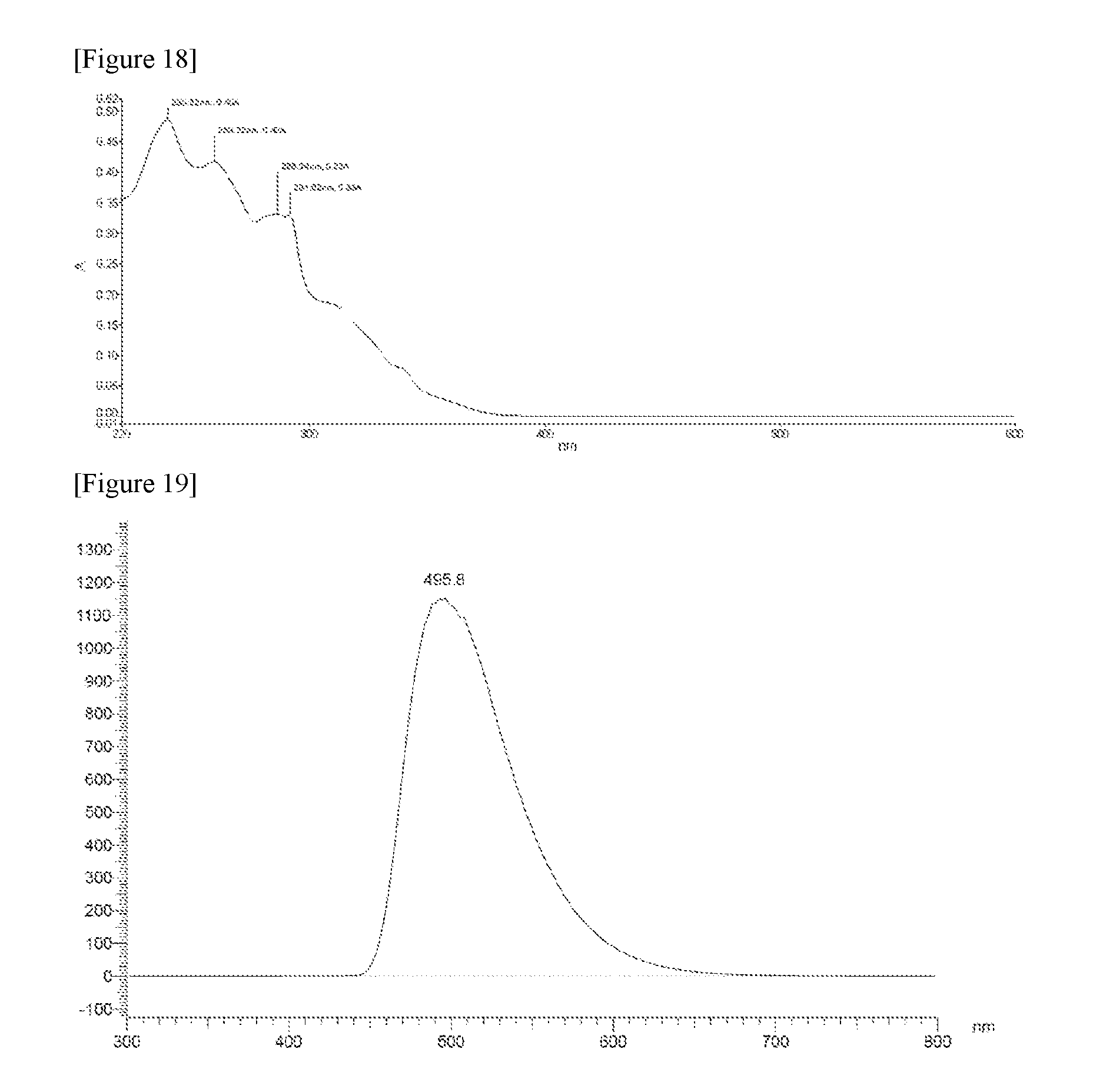
D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025

D00026

D00027

D00028

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.