Electrophoretic dispersion including charged pigment particles, uncharged additive nanoparticles, and uncharged neutral density particles
Sprague , et al.
U.S. patent number 10,288,975 [Application Number 15/803,996] was granted by the patent office on 2019-05-14 for electrophoretic dispersion including charged pigment particles, uncharged additive nanoparticles, and uncharged neutral density particles. This patent grant is currently assigned to E INK CALIFORNIA, LLC. The grantee listed for this patent is E Ink California, LLC. Invention is credited to Peter B. Laxton, Robert A. Sprague, Bo-Ru Yang.

| United States Patent | 10,288,975 |
| Sprague , et al. | May 14, 2019 |
Electrophoretic dispersion including charged pigment particles, uncharged additive nanoparticles, and uncharged neutral density particles
Abstract
The present invention is directed to an electrophoretic dispersion comprising charged pigment particles, in particular, an electrophoretic dispersion including both uncharged or lightly charged neutral buoyancy particles and uncharged or lightly charged additive particles. The combination improves the performance of an electrophoretic display, in particular the color state stability over time.
| Inventors: | Sprague; Robert A. (Saratoga, CA), Yang; Bo-Ru (Banqiao, TW), Laxton; Peter B. (Marshfield, MA) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Applicant: |
|
||||||||||
| Assignee: | E INK CALIFORNIA, LLC (Fremont,
CA) |
||||||||||
| Family ID: | 61240481 | ||||||||||
| Appl. No.: | 15/803,996 | ||||||||||
| Filed: | November 6, 2017 |
Prior Publication Data
| Document Identifier | Publication Date | |
|---|---|---|
| US 20180059500 A1 | Mar 1, 2018 | |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | Issue Date | ||
|---|---|---|---|---|---|
| 14715379 | May 18, 2015 | 9835926 | |||
| 13847888 | Jun 9, 2015 | 9052564 | |||
| 61613833 | Mar 21, 2012 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G02F 1/167 (20130101); G02F 2001/1678 (20130101) |
| Current International Class: | G02B 26/00 (20060101); G02F 1/167 (20190101); G09G 3/34 (20060101); G02F 1/1675 (20190101) |
| Field of Search: | ;359/296 |
References Cited [Referenced By]
U.S. Patent Documents
| 5961804 | October 1999 | Jacobson |
| 6693620 | February 2004 | Herb |
| 6751008 | June 2004 | Liang |
| 6859302 | February 2005 | Liang |
| 6867898 | March 2005 | Liang |
| 6930818 | August 2005 | Liang |
| 7170670 | January 2007 | Webber |
| 7679814 | March 2010 | Paolini, Jr. |
| 7869118 | January 2011 | Wang |
| 7933062 | April 2011 | Masuzawa |
| 8462423 | June 2013 | Farrand |
| 8610998 | December 2013 | Baisch |
| 8665513 | March 2014 | Hsiao |
| 9052564 | June 2015 | Sprague |
| 9835926 | December 2017 | Sprague |
| 2005/0012979 | January 2005 | Minami |
| 2011/0168558 | July 2011 | Fransaer |
| 2002021201 | Mar 2002 | WO | |||
Other References
|
Ho, Andrew, "Embedding e-Paper in Smart Cards, Pricing Labels & Indicators", Presentation conducted at Smart Paper Conference, Atlanta, GA, USA (Nov. 15-16, 2006). cited by applicant . Zang, H.M., "Developments in Microcup Flexible Displays", Presentation conducted at the 6th Annual Flexible Display and Microelectronics Conference, Phoenix, AZ, Feb. 6-8, 2007. cited by applicant . Zang, H.M. et al., "Microcup e-Paper for Embedded and Flexible Designs", IDMC'07, Taipei International Convention Center, Taiwan, (Jul. 2007). cited by applicant . Sprague, R.A., "SiPix Microcup Electrophoretic Epaper for Ebooks", NIP 25, 2009, pp. 460-462; (Sep. 23, 2009). (Presentation conducted on Sep. 23, 2009 at the 25th Int'l Conference on Digital Printing Technologies,Louisville, Kentucky, USA.). cited by applicant . Seigou Kawaguchi et al., "Synthesis of polyethylene macromonomers and their radical copolymerizations with methyl methacrylate in homogeneous and oligoethylene melts media" Designed Monomers and Polymers 2000, vol. 3, No. 3, p. 263-277. cited by applicant . Liang, R.C. and Tseng, Scott (2003) Microcup LCD, A New Type of Dispersed LCD by Roll-to-Roll Manufacturing Process, Paper presented at the IDMC, Taipei, Taiwan, dated Feb. 2003. cited by applicant . Korean Intellectual Property Office; PCT/US2013/033063; International Search Report and Written Opinion; dated Jul. 4, 2013. cited by applicant . Liang, R.C., "BreakThrough-Newly-Developed Color Electronic Paper Promises--Unbeatable Production Efficiency", Nikkei Microdevices, p. 3 (Dec. 2002). (in Japanese, with English translation). cited by applicant . Liang, R.C. et al., "Microcup Electrophoretic Displays by Roll-to-Roll Manufacturing Processes", IDW '02, Paper EP2-2, pp. 1337-1340, (Dec. 2002). cited by applicant . Liang, R.C., "Microcup Electrophoretic and Liquid Crystal Displays by Roll-to-Roll Manufacturing Processes", Presented at Flexible Microelectronics & Displays Conference of U.S. Display Consortium, Phoenix, Arizona, USA (Feb. 2003). cited by applicant . Liang, R.C. et al., "Passive Matrix Microcup Electrophoretic Displays", IDMC'03, Paper FR-17-5, p. 1-4, Taipei, Taiwan, (Feb. 2003). cited by applicant . Liang, R.C.et al., "Microcup Displays: Electronic Paper by Roll-to-Roll Manufacturing Processes" Journal of the SID, 11(4), pp. 621-628, (Feb. 2003). cited by applicant . Chen, S.M., "Revolution electronic paper: The New Application and the Dynamics of Companies", Topology Research Institute, pp. 1-10, (May 2003). (In Chinese, English abstract attached). cited by applicant . Liang, R.C. et al., "Microcup Active and Passive Matrix Electrophoretic Displays by a Roll-to-Roll Manufacturing Processes", SID 03 Digest, Paper 20.1, pp. 838-841 (May 2003). cited by applicant . Lee, H. et al., "SiPix Microcup Electronic Paper--An Introduction" Advanced Display, Issue 37, pp. 4-9, (Jun. 2003). (in Chinese, English abstract attached). cited by applicant . Chen, S.M., "The Applications for the Revolutionary Electronic Paper Technology", OPTO News & Letters, 102, pp. 37-41 (Jul. 2003). (in Chinese, English abstract attached). cited by applicant . Zang, H.M. et al., "Microcup Electronic Paper by Roll-to-Roll Manufacturing Processes", The Spectrum, 16(2), pp. 16-21 (2003). cited by applicant . Allen, K., "Electrophoretics Fulfilled. Emerging Displays Review: Emerging Display Technologies, Monthly Report", Stanford Resources Display Insight, pp. 9-14 (Oct. 2003). cited by applicant . Kleper, M. et al., "An Investigation of the Emerging and Developing Technologies Related to the Generation Beyond Print-on-Paper", Advanced Display Technologies, Rochester Institute of Technology, pp. 13-15, (Oct. 2003). cited by applicant . Zang, H.M., "Microcup Electronic Paper by Roll-to-Roll Manufacturing Processes", Presentation at the Advisory Board Meeting, Bowling Green State Univ., Ohio, USA (Oct. 2003). cited by applicant . Chung, J. et al., "Microcup Electrophoretic Displays, Grayscale and Color Rendition", IDW, AMD2 & EP1-2, pp. 243-246 (Dec. 2003). cited by applicant . Ho, C. et al., "Microcup Electronic Paper by Roll-to-Roll Manufacturing Processes", Presentation conducted at FEG, Nei-Li, Taiwan, (Dec. 2003). cited by applicant . Zang, H.M. et al., "Threshold and Grayscale Stability of Microcup Electronic Paper", SPIE vol. 5289, pp. 102-108, (Jan. 2004). cited by applicant . Zang, H.M., "Microcup Electronic Paper", Presentation at the Displays & Microelectronics Conference of U.S. Display Consortium, Phoenix, Arizona, USA (Feb. 2004). cited by applicant . Wang, X. et al., "Microcup Electronic Paper and the Converting Processes", ASID, 10.1.2-26, pp. 396-399, Nanjing, China, (Feb. 2004). cited by applicant . Chuag, Y.S. et al., "Roll-to-Roll Processes for the Manufacturing of Patterned Conductive Electrodes on Flexible Substrates", Mat Res. Soc. Symp. Proc., vol. 814, I9.6.1., (Apr. 2004). cited by applicant . Liang, R.C., "Microcup Electronic Paper by Roll-to-Roll Manufacturing Process", Presented at the Flexible Displays & Electronics 2004 of Intertech, San Fransisco, California, USA, (Apr. 2004). cited by applicant . Hou, J. et al., "Reliability and Performance of Flexible Electrophoretic Displays by Roll-to-Roll Manufacturing Processes", SID Digest, 32.3, pp. 1066-1069 (May 2004). cited by applicant . Wang, X. et al., "Microcup Electronic Paper and the Converting Processes", Advanced Display, Issue 43, pp. 48-51 (Jun. 2004). (in Chinese, with English abstract). cited by applicant . Liang, R.C. et al., "Format Flexible Microcup Electronic Paper by Roll-to-Roll Manufacturing Process", Presented at 14th FPD Manufacturing Technology EXPO & Conference, Tokyo, Japan, (Jun. 30, 2004-Jul. 2, 2004). cited by applicant . Liang, R.C. "Flexible and Rollable Displays/Electronic Paper--A Technology Overview", Presented at the METS Conference, Taipei, Taiwan, (Oct. 2004). cited by applicant . Bardsley, J.N. et al., "Microcup Electrophoretic Displays", USDC Flexible Display Report, 3.1.2. pp. 3-12 to 3-16, (Nov. 2004). cited by applicant . Ho, Candice, "Microcup Electronic Paper Device and Application", Presentation conducted at USDC 4th Annual Flexible Display and Microelectronics Conference, Phoenix, Arizona, USA, (Feb. 1, 2005). cited by applicant . Zang, H.M. et al., "Flexible Microcup EPD by RTR Process", Presentation conducted at 2nd Annual Paper-Like Displays Conference, St. Pete Beach, Florida, USA, (Feb. 9-11, 2005). cited by applicant . Liang R.C. "Flexible and Roll-able Displays/Electronic Paper--A Brief Technology Overview", Presentation at Flexible Display Forum, Taiwan (Feb. 2005). cited by applicant . Wang, X. et al., "Inkjet Fabrication of Multi-Color Microcup Electrophorectic Display", 5th Flexible Microelectronics & Displays Conference of U.S. Display Consortium, Phoenix, AZ, USA, (Feb. 2006). cited by applicant . Zang, H.M., et al., "Monochrome and Area Color Microcup EPDs by Roll-to-Roll Manufacturing Processes", ICIS 06 International Congress of Imaging Science Final Program and Proceedings, pp. 362-365, Rochester, New York, USA (May 2006). cited by applicant . Wang, X. et al., "Roll-to-Roll Manufacturing Process for Full Color Electrophoretic Film", SID 06 Digest, vol. 37, Issue 1, pp. 1587-1589, (Jun. 2006). cited by applicant . Zang, H.M., "Monochrome and Area Color Microcup EPDs by Roll-to-Roll Manufacturing Process", Presentation conducted at the Fourth Organic Electronics Conference and Exhibition (OEC-06), Frankfurt, Germany, (Sep. 25-27, 2006). cited by applicant. |
Primary Examiner: Hasan; Mohammed A
Attorney, Agent or Firm: Bean; Brian D.
Parent Case Text
RELATED APPLICATIONS
This application is a continuation-in-part of U.S. application Ser. No. 14/715,379, filed May 18, 2015, which is a continuation-in-part of U.S. application Ser. No. 13/847,888, filed Mar. 20, 2013, now U.S. Pat. No. 9,052,564, which claims priority to U.S. Provisional Application No. 61/613,833, filed Mar. 21, 2012; the contents of both are incorporated herein by reference in their entirety.
Claims
What is claimed is:
1. An electrophoretic dispersion comprising: a solvent or solvent mixture; two different species of charged pigment particles having opposite charge polarities, wherein each species has a different color, and wherein each species of charged pigment particles has an average diameter between 50 nm and 1 .mu.m; uncharged or lightly charged additive particles having an average diameter between 10 nm and 250 nm and being present in the electrophoretic dispersion at a concentration between 5% and 40% by volume of the electrophoretic dispersion; and uncharged or lightly charged neutral buoyancy particles having an average diameter between 100 nm and 5 .mu.m and being present in the electrophoretic dispersion at a concentration between 0.3% and 20% by weight of electrophoretic dispersion.
2. The dispersion of claim 1, wherein the concentration of the additive particles is between 5% and 30% by volume of the electrophoretic dispersion.
3. The dispersion of claim 1, wherein the additive particles are formed from a polymer, metal, or metal oxide.
4. The dispersion of claim 1, wherein the additive particles are formed from polystyrene, poly(2-vinylnapthalene), poly(methyl methacrylate), titanium dioxide, silicon dioxide, silver, or gold.
5. The dispersion of claim 1, wherein the uncharged or lightly charged neutral buoyancy particles comprise a polymeric material.
6. The dispersion of claim 5, wherein the uncharged or lightly charged neutral buoyancy particles are formed from polyacrylate, poly(methacrylate), polystyrene, polyaniline, polypyrrole, or polysiloxane.
7. The dispersion of claim 5, wherein the uncharged or lightly charged neutral buoyancy particles are formed from poly(pentabromophenyl methacrylate), poly(2-vinylnapthalene), poly(naphthyl methacrylate), poly(alpha-methystyrene), poly(N-benzyl methacrylamide), or poly(benzyl methacrylate).
8. The dispersion of claim 1, wherein the refractive index of the uncharged or lightly charged neutral buoyancy particles is different from that of the solvent or solvent mixture of the electrophoretic dispersion.
9. The dispersion of claim 1, wherein the average diameter of the additive particles is smaller than the average diameter of the neutral buoyancy particles.
10. The dispersion of claim 1, further comprising a charge control agent.
11. The dispersion of claim 1, wherein the uncharged or lightly charged neutral buoyancy particles are white.
Description
FIELD OF THE INVENTION
The present invention is directed to an electrophoretic dispersion, in particular, an electrophoretic dispersion comprising uncharged or lightly charged additive particles for improving bistability of an electrophoretic display.
BACKGROUND OF THE INVENTION
An electrophoretic display (EPD) is a non-emissive device based on the electrophoresis phenomenon influencing charged pigment particles suspended in a dielectric solvent. An EPD typically comprises a pair of spaced-apart plate-like electrodes. At least one of the electrode plates, typically on the viewing side, is transparent. An electrophoretic dispersion composed of a dielectric solvent with charged pigment particles dispersed therein is enclosed between the two electrode plates.
An electrophoretic dispersion may have one type of charged pigment particles dispersed in a solvent or solvent mixture of a contrasting color. In this case, when a voltage difference is imposed between the two electrode plates, the pigment particles migrate by attraction to the plate of polarity opposite that of the pigment particles. Thus, the color showing at the transparent plate can be either the color of the solvent or the color of the pigment particles. Reversal of plate polarity will cause the particles to migrate to the opposite plate, thereby reversing the color.
Alternatively, an electrophoretic dispersion may comprise two types of pigment particles of contrasting colors and carrying opposite charges and the two types of pigment particles are dispersed in a clear solvent or solvent mixture. In this case, when a voltage difference is imposed between the two electrode plates, the two types of pigment particles would move to opposite ends. Thus one of the colors of the two types of the pigment particles would be seen at the viewing side.
For all types of electrophoretic display, the dispersion contained within the individual display cells of the display is one of the most crucial parts of the device. The composition of the dispersion determines, to a large extent, the lifetime, contrast ratio, switching rate and bistability of the device.
BRIEF DESCRIPTION OF THE DRAWINGS
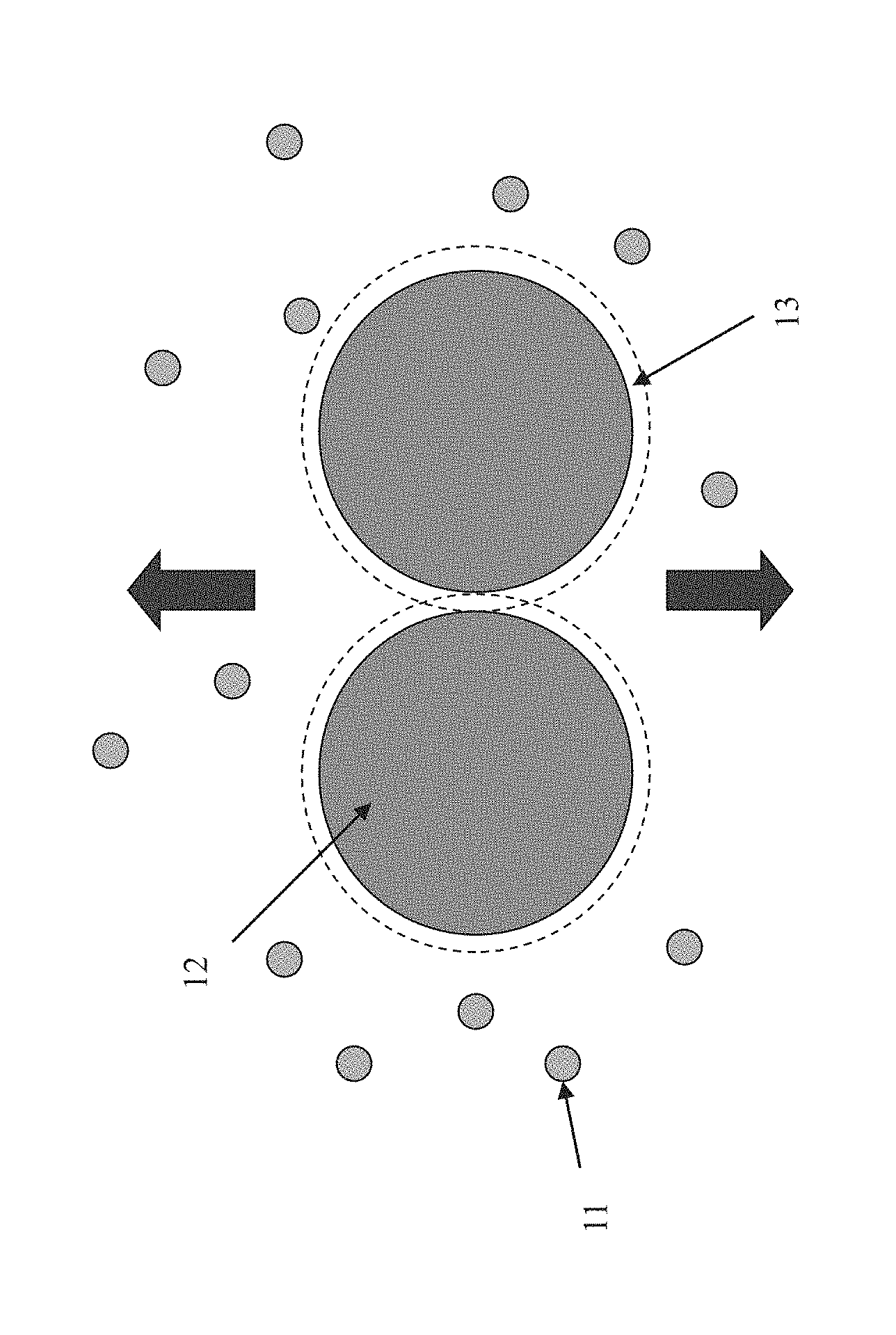
FIG. 1 depicts how the additive nanoparticles may induce flocculation or weak agglomeration of the charged pigment particles.
SUMMARY OF THE PRESENT INVENTION
The present invention is directed to an electrophoretic dispersion comprising charged pigment particles and uncharged or lightly charged additive particles dispersed in a solvent or solvent mixture wherein the average diameter of the additive particles is between 10 nm and 250 nm.
In one embodiment, the dispersion comprises two types of charged pigment particles having opposite charge polarities and of contrasting colors. In one embodiment, the additive particles are formed from a polymer, metal or metal oxide. In one embodiment, the additive particles are formed from polystyrene, poly(2-vinylnapthalene), polymethylmethacrylate, titanium dioxide, silicon dioxide or gold. In one embodiment, the concentration of the additive particles is from about 5% to about 40% by volume of the dispersion, e.g., between from about 10% to about 30% by volume of the dispersion. In one embodiment, the charged pigment particles have a hydrodynamic diameter in the range of 0.1 .mu.m to 1 .mu.m.
In one embodiment, the dispersion further comprises uncharged or lightly charged neutral buoyancy particles. In one embodiment, the uncharged or lightly charged neutral buoyancy particles are formed from a polymeric material. In one embodiment, the uncharged or lightly charged neutral buoyancy particles are formed from polyacrylate, polymethacrylate, polystyrene, polyaniline, polypyrrole, polyphenol or polysiloxane. In one embodiment, the uncharged or lightly charged neutral buoyancy particles are formed from poly(pentabromophenyl methacrylate), poly(2-vinylnapthalene), poly(naphthyl methacrylate), poly(alpha-methystyrene), poly(N-benzyl methacrylamide) or poly(benzyl methacrylate).
In one embodiment, the refractive index of the uncharged or lightly charged neutral buoyancy particles is different from that of the solvent or solvent mixture in which the particles are dispersed. In one embodiment, the uncharged or lightly charged neutral buoyancy particles are light-scattering. In one embodiment, the average diameter of the uncharged or lightly charged neutral buoyancy particles is in the range of about 100 nanometers to about 5 microns.
In one embodiment, the average diameter of the additive particles is smaller than the average diameter of the neutral buoyancy particles. For example, the average diameter is between 10 nm and 250 nm, e.g., between 20 nm and 100 nm. In one embodiment, the concentration of the uncharged or lightly charged neutral buoyancy particles in the electrophoretic dispersion is between 0.3% and 20% by weight of electrophoretic dispersion, e.g., between 0.5% and 10% by weight of electrophoretic dispersion. Alternatively, the concentration may not exceeding about 30% by weight. For example, the concentration may be at least 2.5% by weight, but not exceeding about 25% by weight.
DETAILED DESCRIPTION OF THE INVENTION
Prior to the present invention, it was proposed that image bistability of an electrophoretic display may be improved by dissolving a polymer into an electrophoretic dispersion. However the usefulness of this approach is limited because dissolving a polymer in the dispersion would increase the viscosity of the dispersion, resulting in an increase in the switching time.
The present inventors now have found that by adding uncharged or lightly charged particles into an electrophoretic dispersion, the bistability of an electrophoretic display may be improved, without significantly affecting the switching speed. For brevity, this type of particles is referred to as "additive particles" throughout this application.
The term "lightly charged" is defined as having a charge which is less than 50%, preferably less than 25% and more preferably less than 10%, of the average charge carried by the positively charged pigment particles or negatively charged pigment particles.
The charge intensity may be measured in terms of zeta potential. In one embodiment, the zeta potential is determined by Colloidal Dynamics AcoustoSizer IIM with a CSPU-100 signal processing unit, flow through cell (K:127). The instrument constants, such as density of the solvent used in the sample, dielectric constant of the solvent, speed of sound in the solvent, viscosity of the solvent, all of which at the testing temperature (25.degree. C.) are entered before testing. Pigment samples are dispersed in the solvent (which is usually a hydrocarbon fluid having less than 12 carbon atoms), and diluted to between 5-10% by weight. The sample also contains a charge control agent (Solsperse 17000.RTM., available from Lubrizol Corporation, a Berkshire Hathaway company; "Solsperse" is a Registered Trade Mark), with a weight ratio of 1:10 of the charge control agent to the particles. The mass of the diluted sample is determined and the sample is then loaded into the flow through cell for determination of the zeta potential.
The additive particles may be prepared from a material such as a polymer, metal, metal oxide or the like. For example, they may be formed from polystyrene, poly(2-vinylnapthalene), polymethylmethacrylate, titanium dioxide, silicon dioxide, silver, or gold.
Spherical polymer particles may be prepared in the usual way. For example, in dispersion polymerization, a stabilizing agent which is compatible with the electrophoretic dispersion may be added. In this case, the size of the polymer particles can be precisely controlled by the recipe of the polymerization reaction. Specifically, a higher ratio of the stabilizing agent to the monomer tends to result in smaller particles.
Silica particles of controlled size may be prepared by the Stober process and its modern variants.
Metal nanoparticles, on the other hand, may be synthesized by reduction of an aqueous metal salt solution, for example, citrate synthesis of gold nanoparticles. In the case of metal particles, any of several techniques for size control may be used. For example, a higher concentration of a reducing agent tends to result in smaller particles. In the case of metal and metal oxide particles, care must also be taken to use an appropriate stabilizing agent for the eletrophoretic dispersion.
FIG. 1 depicts how the additive particles (11) may induce flocculation or weak agglomeration of the charged pigment particles (12). In this FIGURE, the charged pigment particles (12) are shown to have a depletion zone (13) where the additive particles (11) are excluded due to their size. During switching, an electric field forces the charged pigment particles to be packed densely, which squeezes out the additive particles from the zone between the charged pigment particles. In the packed state, the energy state of the system tends to be at a lower level than that when the charged pigment particles are dispersed. This is due to the fact that the high energy depletion zones around the charged pigment particles overlap, thus reducing the total depletion zone volume. The resulting packed state therefore is stable and resists change, for example, spontaneous redistribution of the charged pigment particles by Brownian motion which in an electrophoretic display is manifest as an image state decay, known as bistability loss.
When a change of the display state is desired, a reverse electric field is applied which pulls the charged pigment particles apart, allowing the additive particles to become uniformly dispersed among the charged pigment particles. Because of the depletion flocculation energy, this change in state requires a voltage to separate the charged pigment particles. But once that happens, the charged pigment particles move through the dispersion which has a relatively low viscosity associated with the uniformly dispersed state.
The additive particles have certain characteristics. First of all, the particles must be colloidally stable against agglomeration, sedimentation (or creaming) or adsorption. In other words, they must energetically prefer the dispersed state over being adsorbed onto the charged pigment particles or walls that surround the electrophoretic dispersion.
Secondly, they must maintain a specific size relative to the charged pigment particles. The average diameter of the additive particles is about 1% to about 25% of the average hydrodynamic diameter of the charged pigment particles. For example, the average diameter of the additive particles may between 1 nm and 500 nm, e.g., between 5 nm and 250 nm, e.g., between 10 nm and 100 nm. While the primary particle size of the charged pigment particles is often in the range of 50 nm to 500 nm, their dispersed hydrodynamic size is often substantially larger. The charged pigment particles usually have an average hydrodynamic diameter in the approximate range of 0.1 .mu.m to 1 .mu.m. It is this larger hydrodynamic size that must be considered when additive particles are included. Therefore, the additive particles, in most cases, are nanoparticles.
Thirdly, the concentration of the additive particles should be controlled within from about 0.1% to about 40%, preferably about 0.3% to about 20%, by volume of the dispersion. This concentration range is critical in order for the additive particles to be effective in improving bistability of an electrophoretic display. In addition, the refractive index of the additive particles is preferably similar to that of the solvent or solvent mixture in which they are dispersed. A final consideration unique to an electrophoretic display is that it is preferred that the particles are uncharged or lightly charged (i.e., substantially less charged than the charged pigment particles). The additive particles may be present in an electrophoretic dispersion comprising one type of charged pigment particles, two types of charged pigment particles or multiple types of charged pigment particles.
The charged pigment particles may be formed from an inorganic pigment, such as TiO.sub.2, ZrO.sub.2, ZnO, Al.sub.2O.sub.3, CI pigment black 26 or 28 or the like (e.g., manganese ferrite black spinel or copper chromite black spinel). They may also be formed from an organic pigment such as phthalocyanine blue, phthalocyanine green, diarylide yellow, diarylide AAOT yellow, and quinacridone, azo, rhodamine, perylene pigment series from Sun Chemical, Hansa yellow G particles from Kanto Chemical, and Carbon Lampblack from Fisher. The charged pigment particles may also be particles coated with a polymer layer on their surface, and the polymer coating can be prepared through various conventionally known polymerization techniques. The charged pigment particles may carry a natural charge or are charged through the presence of a charge controlling agent.
The solvent or solvent mixture in which the charged pigment particles are dispersed preferably has a low viscosity and a dielectric constant in the range of about 2 to about 30, preferably about 2 to about 15 for high particle mobility. Examples of suitable dielectric solvent include hydrocarbons such as isopar, decahydronaphthalene (DECALIN), 5-ethylidene-2-norbornene, fatty oils, paraffin oil; silicon fluids; aromatic hydrocarbons such as toluene, xylene, phenylxylylethane, dodecylbenzene and alkylnaphthalene; halogenated solvents such as perfluorodecalin, perfluorotoluene, perfluoroxylene, dichlorobenzotrifluoride, 3,4,5-trichlorobenzotri fluoride, chloropentafluoro-benzene, dichlorononane, pentachlorobenzene; and perfluorinated solvents such as FC-43, FC-70 and FC-5060 from 3M Company, St. Paul Minn., low molecular weight halogen containing polymers such as poly(perfluoropropylene oxide) from TCI America, Portland, Oreg., poly(chlorotrifluoro-ethylene) such as Halocarbon Oils from Halocarbon Product Corp., River Edge, N.J., perfluoropolyalkylether such as Galden from Ausimont or Krytox Oils and Greases K-Fluid Series from DuPont, Delaware, polydimethylsiloxane based silicone oil from Dow-coming (DC-200). The solvent or solvent mixture may be colored by a dye or pigment.
In addition to the uncharged or lightly charged additive particles, the dispersion may further comprise uncharged or lightly charged neutral buoyancy particles. The term "neutral buoyancy" refers to particles which do not rise or fall with gravity. In other words, the particles would float in the dispersion between the two electrode plates. In one embodiment, the density of the neutral buoyancy particles may be the same as the density of the solvent or solvent mixture in which they are dispersed.
The uncharged or lightly charged neutral buoyancy particles may be formed from a polymeric material. The polymeric material may be a copolymer or a homopolymer. Examples of the polymeric material for the uncharged or lightly charged neutral buoyancy particles may include, but are not limited to, polyacrylate, polymethacrylate, polystyrene, polyaniline, polypyrrole, polyphenol, polysiloxane or the like. More specific examples of the polymeric material may include, but are not limited to, poly(pentabromophenyl methacrylate), poly(2-vinylnapthalene), poly(naphthyl methacrylate), poly(alpha-methystyrene), poly(N-benzyl methacrylamide) or poly(benzyl methacrylate). These materials are suitable for the neutral buoyancy particles in one particle system, two particle system, and multiple particle system.
More preferably, the uncharged or lightly charged neutral buoyancy particles are formed from a polymer which is not soluble in the dielectric solvent of the display dispersion, and also has a high refractive index. In one embodiment, the refractive index of the uncharged or lightly charged neutral buoyancy particles is different from that of the solvent or solvent mixture in which the particles are dispersed. However, typically the refractive index of the uncharged or lightly charged neutral buoyancy particles is higher than that of the solvent or solvent mixture. In some cases, the refractive index of the uncharged or lightly charged neutral buoyancy particles may be above 1.45.
In one embodiment, the materials for the uncharged or lightly charged neutral buoyancy particles may comprise an aromatic moiety.
The uncharged or lightly charged neutral buoyancy particles may be prepared from monomers through polymerization techniques, such as suspension polymerization, dispersion polymerization, seed polymerization, soap-free polymerization, emulsion polymerization or physical method, including inverse emulsification-evaporation process. The monomers are polymerized in the presence of a dispersant. The presence of the dispersant allows the polymer particles to be formed in a desired size range and the dispersant may also form a layer physically or chemically bonded to the surface of the polymer particles to prevent the particles from agglomeration.
The dispersants preferably has a long chain (of at least eight atoms), which may stabilize the polymer particles in a hydrocarbon solvent. Such dispersants may be an acrylate-terminated or vinyl-terminated macromolecule, which are suitable because the acrylate or vinyl group can co-polymerize with the monomer in the reaction medium.
One specific example of the dispersant is acrylate terminated polysiloxane (Gelest, MCR-M17, MCR-M22),
Another type of suitable dispersants is polyethylene macromonomers, as shown below: CH.sub.3--[-CH.sub.2-].sub.n--CH.sub.2O--C(.dbd.O)--C(CH.sub.3).dbd.CH.su- b.2
The backbone of the macromonomer may be a polyethylene chain and "n" may be 30-200. The synthesis of this type of macromonomers may be found in Seigou Kawaguchi et al, Designed Monomers and Polymers, 2000, 3, 263.
If the dispersion system is fluorinated, the dispersants are then preferably also fluorinated.
Alternatively, the uncharged or lightly charged neutral buoyancy particles may also be formed from a core particle coated with a polymeric shell and the shell may be formed, for example, from any of the polymeric material identified above for the neutral buoyancy particles.
The core particle may be of an inorganic pigment or an organic pigment as described above.
In the case of core-shell uncharged or lightly charged neutral buoyancy particles, they may be formed by a microencapsulation method, such as coacervation, interfacial polycondensation, interfacial cross-linking, in-suit polymerization or matrix polymerization.
The average diameter of the uncharged or lightly charged neutral buoyancy particles is preferably in the range of about 100 nanometers to about 5 micrometers. In one embodiment, the average diameter of the additive particles is smaller than the average diameter of the neutral buoyancy particles.
In one embodiment of this aspect of the present invention, the uncharged or lightly charged neutral buoyancy particles added to the dispersion may have a color substantially the same as the color of one of the two types of charged pigment particles. For example, in a display dispersion, there may be charged black particles, charged white particles, the uncharged or lightly charged additive particles and uncharged or lightly charged neutral buoyancy particles and the uncharged or lightly charged neutral buoyancy particles may be either white or black.
If the uncharged or lightly charged neutral buoyancy particles are white, they may enhance the reflectivity of the display. If they are black, they may enhance the blackness of the display.
In another embodiment, the uncharged or lightly charged neutral buoyancy particles may have a color substantially different from the color of either one of the two types of charged pigment particles.
The presence of the uncharged or lightly charged neutral buoyancy particles, especially if they are formed from a reflective material, increases reflection of the incident light, thus improving the contrast ratio.
In a further embodiment of the present invention, the concentration of the uncharged or lightly charged neutral buoyancy particles in an electrophoretic dispersion is preferably more than 0.1% by weight, e.g., more than 0.3% by weight, e.g., more than 0.5% by weight, e.g., more than 2.5% by weight, but not exceeding about 40% by weight, e.g., not exceeding about 30% by weight, e.g., not exceeding about 20% by weight. In another embodiment, the concentration of the uncharged or lightly charged neutral buoyancy particles is preferably in a range between about 0.3% to about 20% by weight and more preferably in a range between about 0.5% to about 10% by weight.
The electrophoretic dispersion of the present invention, in addition to the uncharged or lightly charged additive particles and uncharged or lightly charged neutral buoyancy particles, may also comprise other additives such as a charge controlling agent. The charge control agent (CCA) used in all embodiments of the present invention is compatible with the solvent in the electrophoretic dispersion and may interact with the surface of the charged particles to effectively generate either positive or negative charge for the particles. Useful charge control agents include, but are not limited to, sodium dodecylbenzenesulfonate, metal soap, polybutene succinimide, maleic anhydride copolymers, vinylpyridine copolymers, vinylpyrrolidone copolymer, (meth)acrylic acid copolymers or N,N-dimethylaminoethyl (meth)acrylate copolymers), Alcolec LV30 (soy lecithin), Petrostep B100 (petroleum sulfonate) or B70 (barium sulfonate), Solsperse 17000 (active polymeric dispersant), Solsperse 9000 (active polymeric dispersant), OLOA 11000 (succinimide ashless dispersant), OLOA 1200 (polyisobutylene succinimides), Unithox 750 (ethoxylates), Petronate L (sodium sulfonate), Disper BYK 101, 2095, 185, 116, 9077 & 220 and ANTI-TERRA series.
A further aspect of the present invention is directed to an electrophoretic display wherein display cells are filled with any of the display dispersions as described in the present application.
The term "display cell" refers to a micro-container filled with a display dispersion. A display cell may be a microcell as described in U.S. Pat. No. 6,930,818, the content of which is incorporated herein by reference in its entirety. A display cell may also be any other micro-containers (e.g., microcapsules or microchannels), regardless of their shapes or sizes. All of these are within the scope of the present application.
While the present invention has been described with reference to the specific embodiments thereof, it should be understood by those skilled in the art that various changes may be made and equivalents may be substituted without departing from the scope of the invention. In addition, many modifications may be made to adapt a particular situation, materials, compositions, processes, process step or steps, to the objective and scope of the present invention. All such modifications are intended to be within the scope of the claims appended hereto.
* * * * *
D00000

D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.