GOFEXII
CSL Behring GmbH
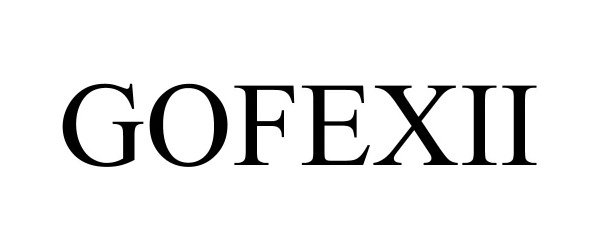
Mark For: GOFEXII® trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 90398211 |
| Registration Number | 6618401 |
| Mark Literal Elements | GOFEXII |
| Mark Drawing Type | 4 - STANDARD CHARACTER MARK |
| Mark Type | Trademark |
| Current Location | NEW APPLICATION PROCESSING 2020-12-24 |
| Basis | 44(d) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | No |
| Intent To Use | No |
| Filed ITU | No |
| 44D Filed | Yes |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Domestic Representative | Scott D. Woldow |
| Attorney Name | Scott D. Woldow |
| Attorney Docket Number | 034845.255 |
Timeline
| 2020-12-21 | Application Filed |
| 2020-12-24 | Location: NEW APPLICATION PROCESSING |
| 2020-12-24 | Status: Live/Pending |
| 2020-12-24 | Status: New application will be assigned to an examining attorney approximately 3 months after filing date. |
| 2020-12-24 | Transaction Date |
| 2022-01-18 | Trademark Registered |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | Emil-von-Behring Str. 76 Marburg GERMANY D-35041 |
| Legal Entity Type | Limited Liability Company |
| Legal Entity State | GERMANY |
Documents
| MULTI | 2020-12-21 | |
| JPEG | 2020-12-21 |
Attorney of Record
Good, Services, and Codes
| International Codes: | 5 |
| U.S. Codes: | 005,006,018,044,046,051,052 |
| Type Code | Type |
|---|---|
| GS0051 | Pharmaceutical preparations; antibodies; blood products; blood plasma; blood plasma fractions; blood plasma products for medical purposes; recombinant proteins for medical purposes; cell cultures for medical purposes |
| GS0051 | Pharmaceutical preparations for treating von Willebrand disease, hemophilia, primary immunodeficiency, hereditary angioedema, fibrinogen deficiency, Alpha 1 antitrypsin deficiency, and inflammatory demyelinating polyneuropathy; blood plasma; blood plasma fractions; recombinant protein arrays for medical diagnosis purposes; Pharmaceutical preparations for the treatment and prevention of blood and bleeding disorders; blood products for the treatment of hemophilia, namely, pharmaceutical preparations for the treatment of hemophilia; pharmaceutical products derived from recombinant DNA technology for the treatment of hemophilia, namely, pharmaceutical preparations derived from recombinant DNA technology for the treatment of hemophilia; blood products for the treatment of hemophilia, namely, a blood clotting aid and delivery system for use in human and veterinary medicine; pharmaceutical preparations and substances for the treatment of blood disorders; antibodies, namely, diagnostic kits consisting primarily of monoclonal antibodies, buffers, and reagents for use in disease testing; blood for medical purposes; cell cultures for medical purposes, namely, cells for medical or clinical use; cell cultures for medical purposes, namely, living cells for medical use; cell cultures for medical purposes, namely, stem cells for medical or veterinary use; pharmaceutical preparations for the treatment of blood conditions, respiratory conditions, pneumonia, covid-19, and influenza; Pharmaceutical preparations, namely, antibody for the treatment of acute respiratory distress syndrome, ARDS; pharmaceutical preparations, namely, antibody for the treatment of Idiopathic Pulmonary Fibrosis, IPF and Interstitial Lung Diseases, ILD; Pharmaceutical preparations, namely, antibody for the treatment of Hereditary Angioedema, HAE |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| NEW APPLICATION ENTERED IN TRAM | 2020-12-24 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.