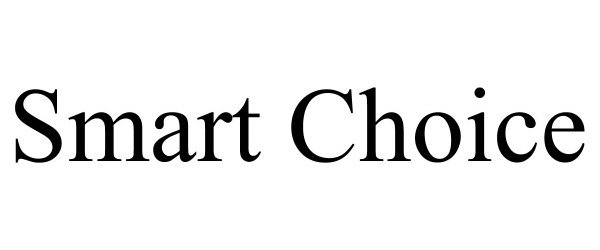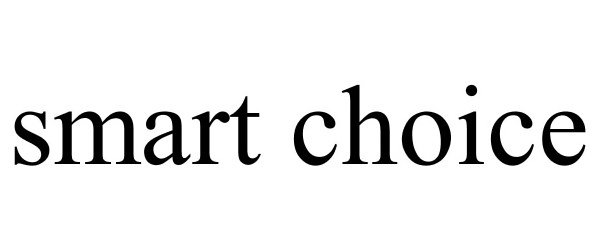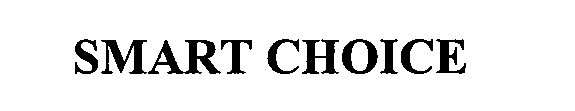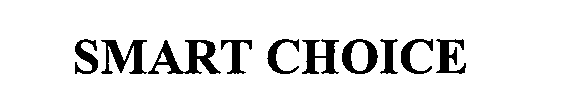SMART CHOICE
American Pharmaceutical Company
| Research |
|
| Serial Number | 75069392 |
| Mark Drawing Code | 3000: Illustration: Drawing or design which also includes word(s)/ letter(s)/number(s) |
| Attorney Name | David L. Bieber |
| Law Office Assigned | L60 |
| Employee Name | COWARD, JEFFERY |
Timeline
| 1993-08-20 | Date of First Use |
| 1996-03-08 | Application Filed |
| 1999-06-11 | Abandon |
| 1999-09-11 | Status: Dead/Abandoned |
| 1999-09-17 | Location: FILE REPOSITORY (FRANCONIA) |
| 2018-07-08 | Transaction Date |
Trademark Applicants & Owners
| Owner: | |
| Address | 200 Webro Road Parsippany NJ 07054 |
| Legal Entity Type | Corporation |
| Legal Entity State | NJ |
Documents
Click the blue "Refresh" button to load certificates, specimines, application, and other documents.
Design Search Codes
| 261127 | Oblongs not used as carriers for words, letters or designs |
Attorney of Record
Good, Services, and Codes
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| Type Code | Type |
|---|---|
| GS0051 | vitamins, minerals, non-prescription medicines, and pharmaceutical preparations, namely, aspirin tablets; children's chewable aspirin tablet; tri-buffered aspirin tablets; ibuprofen tablets and caplets; non-aspirin pain relief tablets, caplets and gelatin caplets; non-aspirin pain relief cold tablets; non-aspirin pain relief PM caplets; children's non-aspirin pain relief chew tabs and elixir; infant's non-aspirin pain relief drops; adult strength pain relief tablets; antihistamine nasal decongestant tablets; total allergy medication tablets, capsules and oral liquid; chlorpheniramine tablets; allergy and cold elixir and tablets; sinus aid tablets; nasal decongestant tablets; saline nasal spray; sore throat lozenges; sore throat lozenges; sore throat spray; cough and cold formula tablets; timed cold capsules; night time cold medicine; cough control syrups; tristine cold syrup; tristine expectorant; bacitracin ointment; corticreme hydrocortisone creme; triple antibiotic ointment; hemor aid suppositories; tolnaftate antifungal creme; cool gel; benzoyl peroxide gel; bisacodyl tablets; natural vegetable laxative powder; women's gentle laxative tablets; stool softener softgels; chocolate laxative tablets; laxative pills; pink bismuth liquid; infant gas relief drops; antacid suspension; milk of magnesia antacid liquid; chewable antacid tablets; anti-diarrheal caplets; travel sickness tablets; amphetrim timed diet caplets; alertness tablets; medicated cleansing pads w/aloe vera; ear wax removal drops; calcitab tablets; ferrous sulfate tablets; natural oyster shell calcium tablets; odor free garlic tablets; ginseng caplets; stress free vitamins; anti-oxidant vitamin and mineral softgels; one a day natural formula vitamins; circus chews children vitamins; thera M vitamins; vitrum vitamins; chromium picolinate capsules; vitamin C and rose hips tablets; cod liver oil softgels; niacin tablets; ultra-vite vitamin tablets; beta carotene softgels; vitamin C tablets; selenium tablets; zinc gluconate tablets; super B tablets; lecithin softgels; natural prenatal forumla tablets; vitamin B-6 tablets; potassium gluconate tablets; acidophilus and pectin caplets; vitamin E softgels; vitamin B-1 tablets |
Trademark Filing History
| Description | Date | Event Coding |
|---|---|---|
| ASSIGNED TO EXAMINER | 1996-08-12 | 1 DOCK D:Assigned to Examiner |
| ASSIGNED TO EXAMINER | 1996-08-22 | 2 DOCK D:Assigned to Examiner |
| NON-FINAL ACTION MAILED | 1996-09-19 | 3 CNRT F:First Action |
| LETTER OF SUSPENSION MAILED | 1997-06-06 | 4 CNSL F:First Action |
| ASSIGNED TO EXAMINER | 1997-12-08 | 5 DOCK D:Assigned to Examiner |
| NON-FINAL ACTION MAILED | 1998-12-10 | 6 CNRT F:First Action |
| ABANDONMENT - FAILURE TO RESPOND OR LATE RESPONSE | 1999-09-11 | 7 ABN2 O:Outgoing Correspondence |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.